Acidifying salts - Nonmercurial diuretics
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Diuretics
Some inorganic cations produce weak acidiotic diuresis, such as ammonium chloride, ammonium nitrate, and calcium chloride.
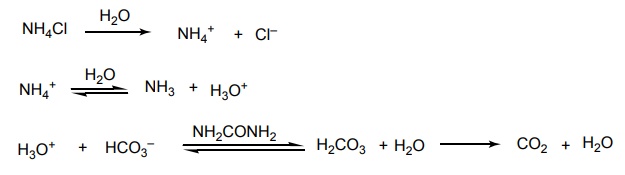
Some inorganic cations produce weak acidiotic diuresis, such as ammonium chloride, ammonium nitrate, and calcium chloride. The mechanism of acidification is represented by the following equation. Ammonium chloride is metabolized to urea in the liver with the formation of H+. The excess acid (H+) is buffered by HCO3 to form carbon dioxide. This leads to an acidification of the urine. An excess Cl– also occurs in the tubular lumen and takes Na+ along with it to maintain electrical neutrality.SYNTHESIS AND DRUG PROFILE
Nonmercurial diuretics
Acidifying salts
Related Topics
