Barbiturates
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Sedatives and Hypnotics
Barbiturates are derivatives of barbituric acid. Their hypnotic activity is conferred by the replacement of H-atom attached to the C-5 position by aryl or alkyl radicals.
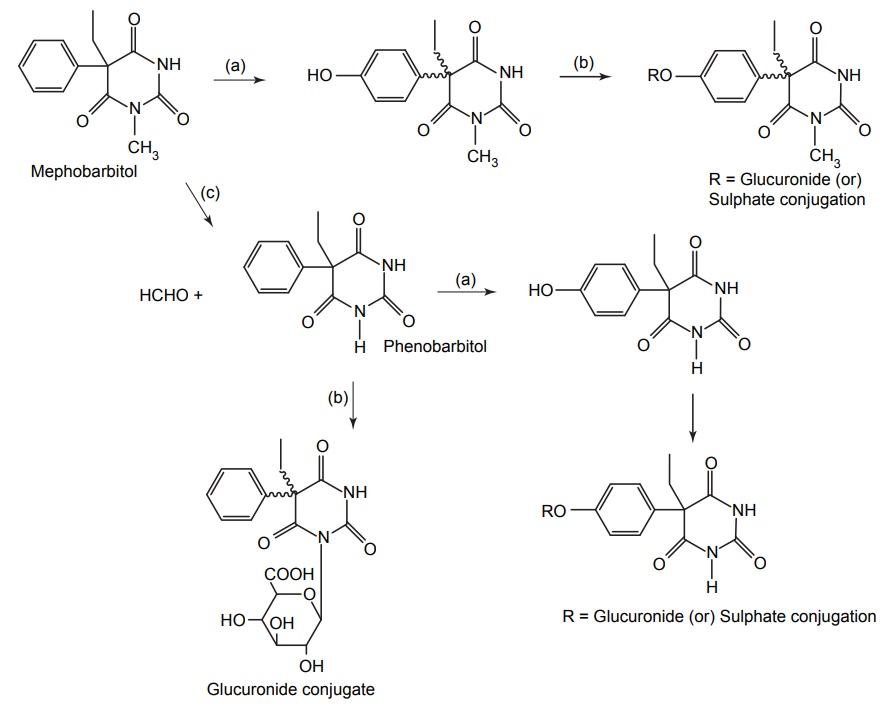
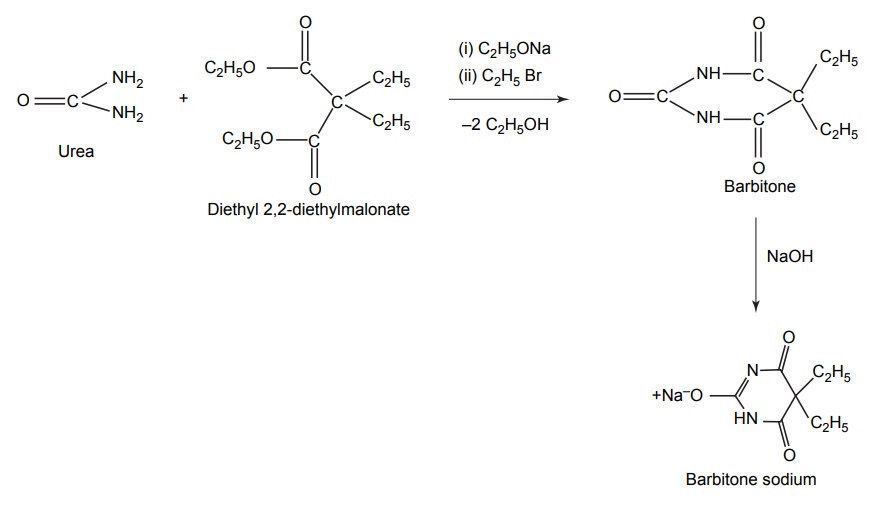
SYNTHESIS AND DRUG PROFILE Barbiturates are derivatives of barbituric acid. Their hypnotic activity is conferred by the replacement of H-atom attached to the C-5 position by aryl or alkyl radicals. They are generally synthesized by adopting the following route of synthesis. Mode of action: Barbiturates primarily act on GABA: benzodiazepin receptor Cl– channel complex and potentiate GABA ergic inhibitory action by increasing the lifetime of Cl– channel opening induced by GABA. Barbiturates do not bind to benzodiazepine receptor promptly, but it binds to another site on the same macromolecular complex to exert the GABA ergic facilitator actions. The barbiturate site appears to be located on a and ß subunit. At high concentrations, barbiturates directly increases Cl– conductance and inhibit Ca2+ dependent release of neurotransmitters and they also depress glutamate-induced neuronal depolarization. Synthesis Route I: From urea and malonic acid Route II: From chloroacetic acid Metabolism of barbiturates: These drugs are metabolized in the liver and forms less lipophilic compounds. These are mediated through glucuronide or sulphate conjugation. Oxidation of a substituent at C-5 forms alcohols or phenols, and these undergo further oxidation to form ketones or carboxylic acids. The barbiturates containing a propene at the fifth position inactivates CYP450 by alkylation of the porphyrin ring of CYP450. The conjugation of heterocyclic nitrogen with glucuronic acid is due to the oxidative metabolism in the biotransformation of 5,5-disubstituted barbiturates (phenobarbital and amobarbital). It undergoes oxidative N-dealkylation at nitrogen. Oxidative desulphation of 2-thio barbiturates yields more hydrophilic barbiturates, which is excreted through urine. Uses: They are used as sedative, hypnotic and anticonvulsant. They are administered through the intravenous (IV) route for inducing anaesthesia. Properties and uses: Baritone sodium exists as white, crystalline powder or colourless crystals that is soluble in boiling water and in alcohol, but only slightly soluble in water. It forms water-soluble salts with sodium hydroxide. It is a powerful hypnotic drug and generally used in the treatment of epileptic seizures. Assay: Dissolve the substance in pyridine and to this add thymolphthalein solution and silver nitrate solution in pyridine and titrate the solution with 0.1 methanolic sodium hydroxide. Endpoint is the appearance of blue colour. Synthesis Properties and uses: Phenobarbital sodium is a hygroscopic substance. It is a white, crystalline powder, freely soluble in water and also soluble in alcohol. It is used as sedative, hypnotic and antiepileptic (drug of choice in the treatment of grandmal and petitmal epilepsy). It is useful in nervous and related tension states. An overdose of it can result in coma, severe respiratory depression, hypotension leading to cardiovascular collapse, and renal failure. Synthesis Assay: Dissolve the sample in water and add 0.05 M sulphuric acid. Heat the mixture to boiling point and then cool it. Add methanol and shake until dissolution is complete. Perform a potentiometric titration using 0.1 M sodium hydroxide. After the first point of inflexion, interrupt the addition of sodium hydroxide, add 10 ml of pyridine, mix, and continue the titration. Read the volume added between the two points of inflexion. Storage: It should be stored in well closed airtight container. Dose: For sedation: Adult: 30–120 mg/day in 2–3 divided doses. Children: 6 mg/kg/day. For hypnotic: Adult: 100–320 mg at bedtime. Do not administer for more than 2 weeks for the treatment of insomnia. Through IV route for preoperative sedation: Child: 1–3 mg/kg 1–1.5 h before procedure. Dosage forms: Phenobarbital sodium tablets I.P., B.P., Phenobarbital sodium injection I.P., Phenobarbitone tablets I.P., Phenobarbital injection B.P., Paediatric phenobarbital oral solution B.P. Synthesis Properties and uses: It is a white, crystalline powder, odourless, with a bitter taste, and a saturated is solution acid to litmus. Soluble in water, alcohol, chloroform, and in solutions of alkali hydroxides or carbonates. Mephobarbitone is a strong sedative with anticonvulsant action, but a relatively mild hypnotic. Hence, it is used for the relief of anxiety, tension, and apprehension, and is an antiepileptic in the management of generalized tonic-clonic and absence seizures. Dose: As a sedative 30–100 mg 3–4 times/day; as an anticonvulsant 400–600 mg daily. Synthesis Uses: It can be used both as sedative and hypnotic at different dose intervals. Dose: As a sedative 30 mg 3–4 times a day; as a hypnotic 100–200 mg at night. Synthesis Properties and uses: It is used as a sedative and hypnotic, especially used for the short-term treatment of insomnia. Because of tolerance, barbiturates lose efficacy after two weeks of use. Dose: 30 mg as a sedative and 100–200 mg at night as a hypnotic. Synthesis Properties and uses: It is a white, crystalline powder, very slightly soluble in water, sparingly soluble in alcohol. It forms sodium salt with sodium hydroxide, which is soluble in water. It is used as hypnotic. Assay: Dissolve the sample in ethanol; to this add thymolphthalein solution and silver nitrate solution in pyridine. Titrate with 0.1 Methanolic sodium hydroxide. Endpoint is the appearance of blue colour. Dose: The usual dose for adult, oral hypnotic is 250–500 mg. Synthesis Properties and uses: It is hygroscopic in nature and a white crystalline powder, very soluble in water. It is used as a sedative or hypnotic for the short-term management of insomnia and is a preanaesthetic medication, used in the treatment of strychnine poisoning. It is also indicated in the anaesthetic doses and administered intravenously, for the control of certain convulsive syndromes. This barbiturate is thought to reduce cerebral blood flow and, thereby, decrease oedema and intracranial pressure. Assay: Dissolve the sample in silver nitrate solution in pyridine and titrate with 0.1 methanolic sodium hydroxide until a pure blue colour is obtained, using 0.5 ml of thymolphthalein solution as indicator. Storage: It should be stored in well-closed airtight containers. Dose: The usual oral sedative dose for adult is 30 mg 2–4 times daily. Through I.M. route, for preoperative, sedative, 150–200 mg. Through IV route, anticonvulsant, 100 mg initially up to 400 mg. Dosage forms: Pentobarbital tablets I.P., B.P. Synthesis Properties and uses: It is a white, hygroscopic powder having a bitter taste, with pH between 9.7 and 10.5, soluble in water and alcohol. It is used in status epilepticus and in toxic reactions to strychnine and as local anaesthetic. Dose: The usual adult dose is 50–200 mg.1. Barbiturates
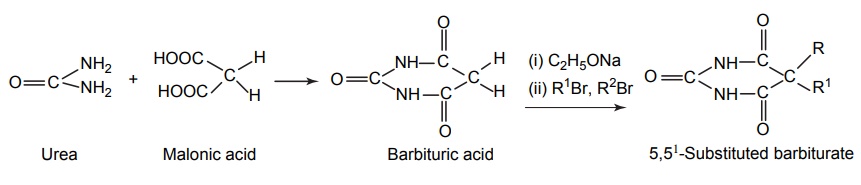
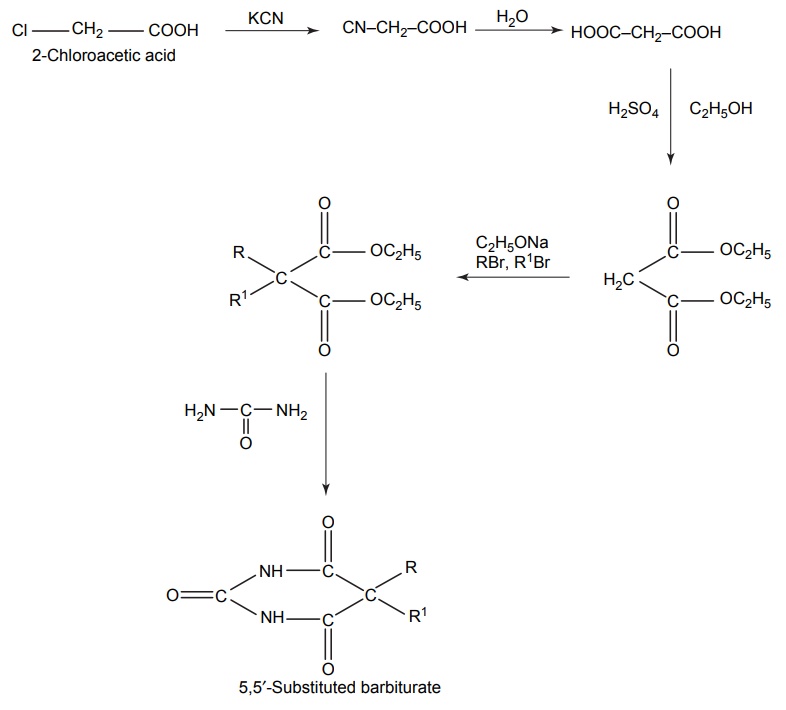
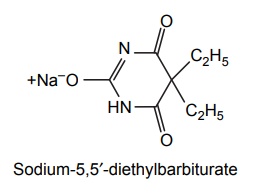
1. Barbitone sodium (Barbital sodium)
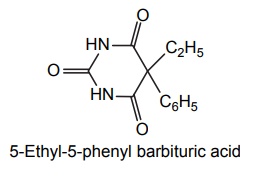
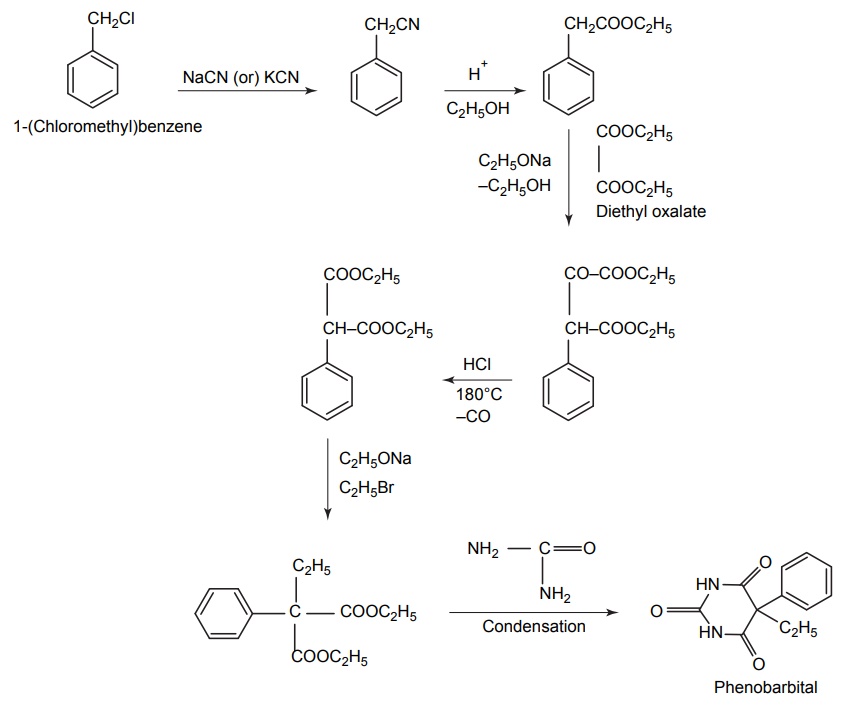
2. Phenobarbitone (Phenobarbital, Luminal)
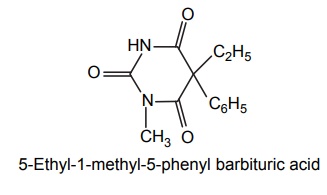
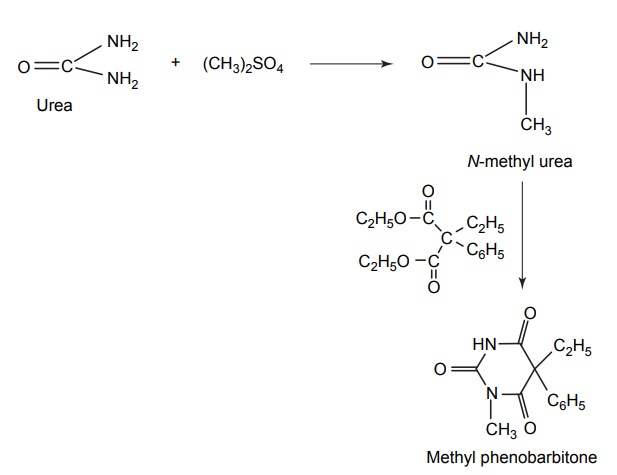
3. Methyl phenobarbitone (Mephobarbital)
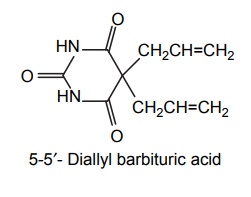
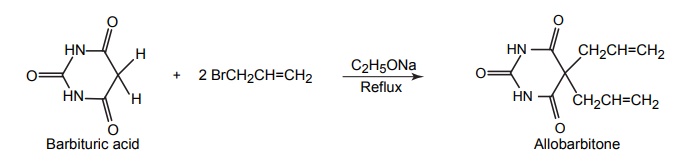
4. Allobarbitone
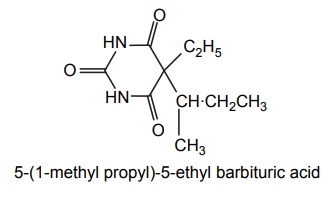
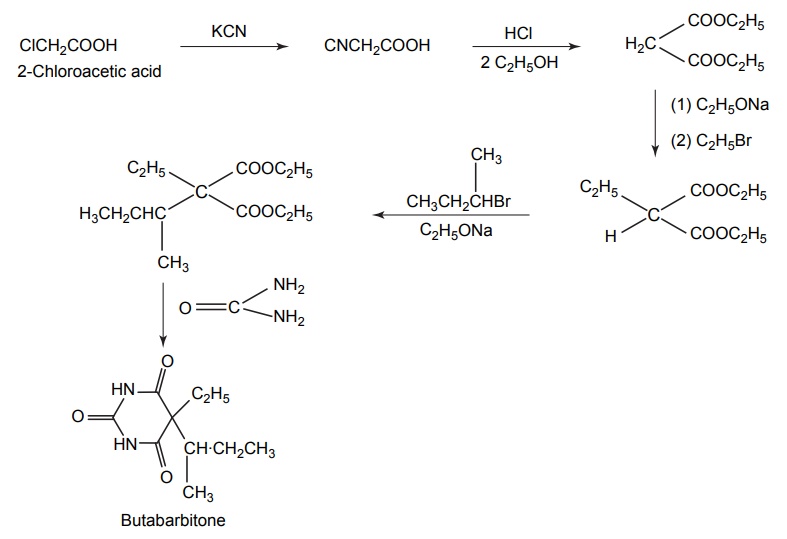
6. Butabarbitone (Neonal)
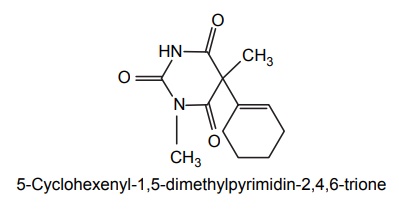
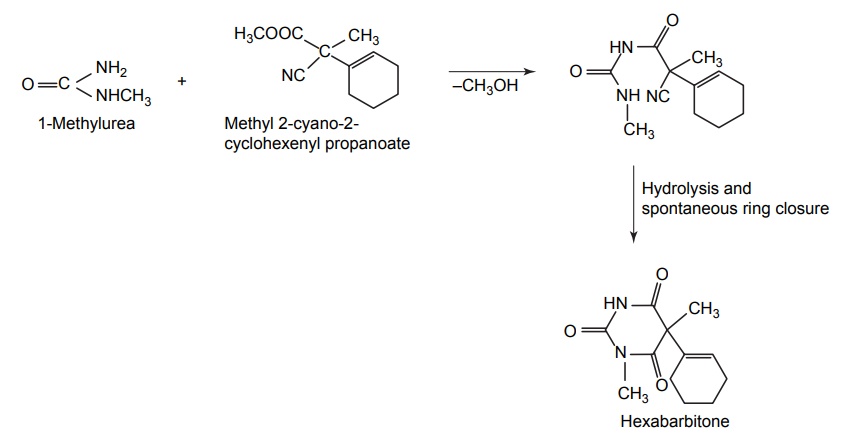
5. Hexabarbitone (Hexobarbital, Evipal, Sombulex)
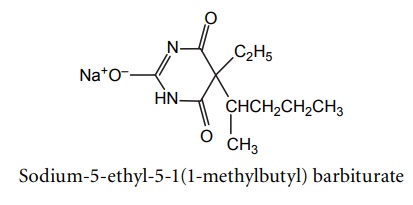
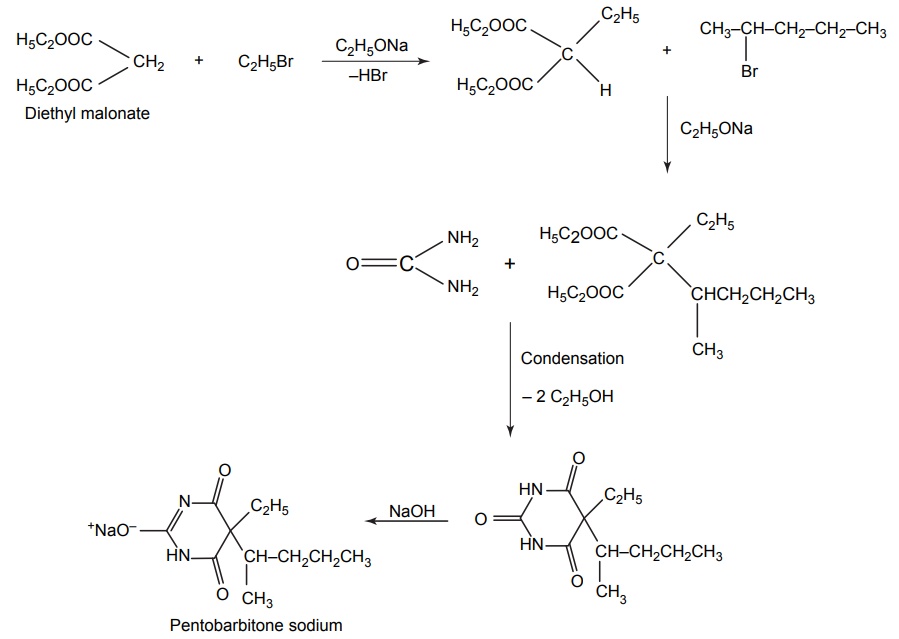
6. Pentobarbitone sodium (Pentobarbital sodium, Palapent, Sodital)
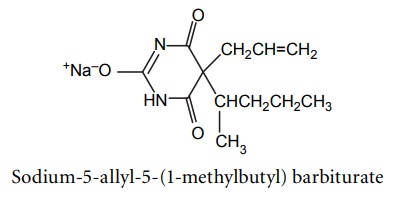
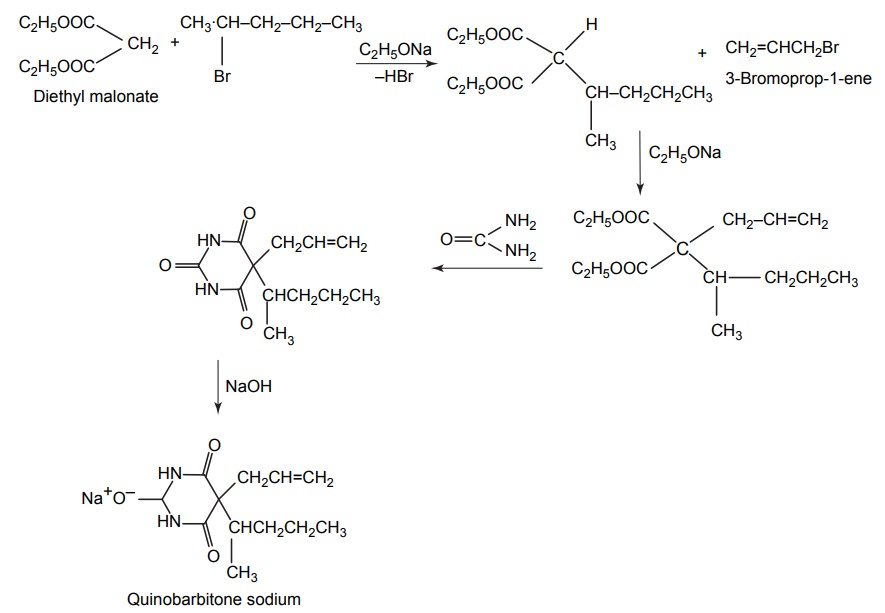
7. Quinobarbitone sodium (secobarbital sodium)
Related Topics
