Classification of Hydrogen Bonding
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Hydrogen Bonding
Generally, hydrogen bonding is classified into two types: 1. intermolecular hydrogen bonding 2. intramolecular hydrogen bonding
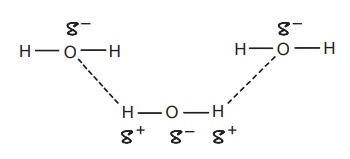
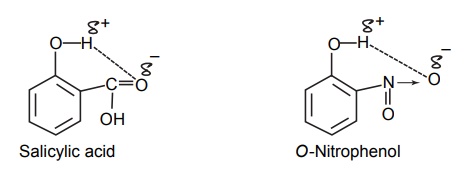
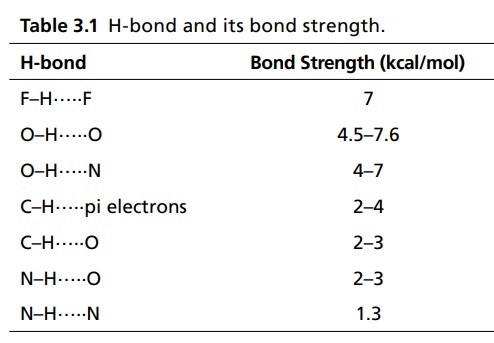
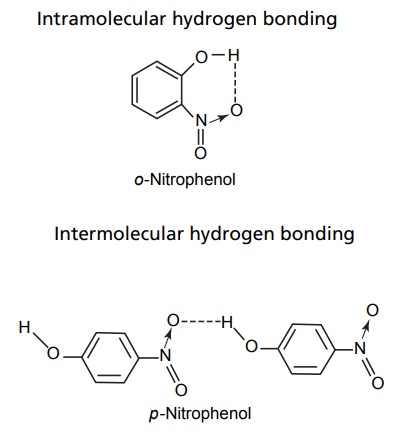
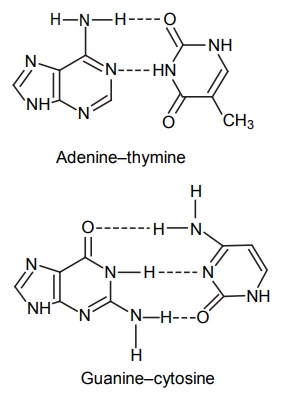
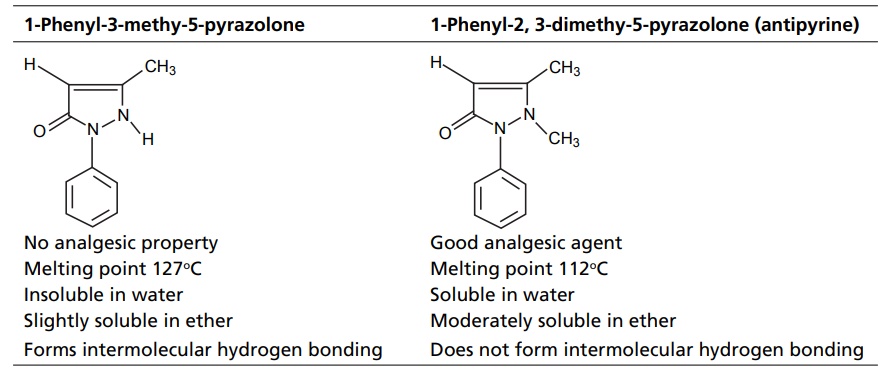
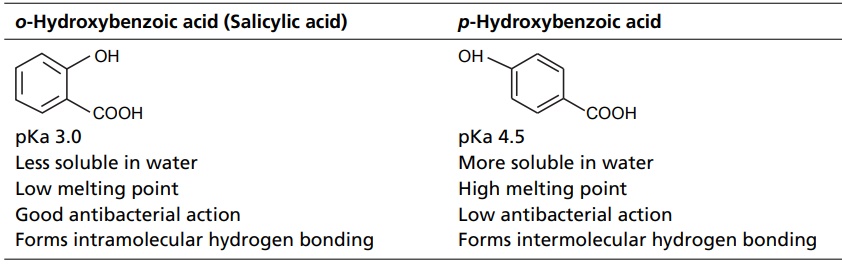
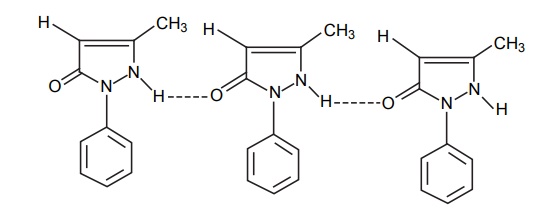
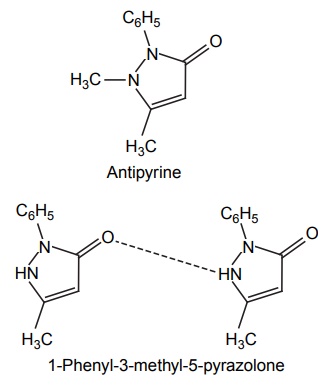
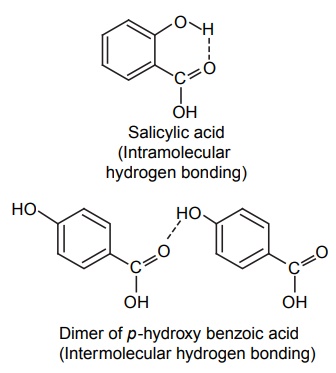
CLASSIFICATION Generally, hydrogen bonding is classified into two types: intermolecular hydrogen bonding intramolecular hydrogen bonding Intermolecular hydrogen bonding: Hydrogen bonding occurs between two or more molecules Intramolecular hydrogen bonding: Hydrogen bonding occurs within the molecules There is also some evidence that the hydrogen attached to a triple bond carbon (e.g. HCN and CHCl3) forms a H-bond. The strength of H-bonds ranges from 1 to 10 kcal/mol, and usually is about 5 kcal/mol. The distance between the electronegative elements in a H-bond is usually in the range of 2.5–2.7 A°. At distance greater than 3 A°, there is very little interaction (Table 3.1). The stability of H-bonds falls roughly in the following order, OHO > OHN > NHN. H-bonds may occur between molecules (intermolecular H-bond), within one molecule (intramolecular H-bond), or as a combination of these two. Intermolecular bonds are frequently much weaker than the intramolecular bonds. Multiple hydrogen bonding groups in any drug molecule would greatly increase its potential for aqueous solubility. Minimal aqueous solubility is essential for all the drug molecules to transport to the site of action on a receptor. Generally, the more H-bonds that are possible, the greater the water solubility of the molecule. The strength of the H-bond depends on the solvent as well as on the physical state. For example, the H-bond strength of O–H ….. O for CH3COOH dimer in vapour state is 7.64 kcal/mol, but CH3COOH dimer in benzene is 4.85 kcal/mol. In water, the H-bond strength is 4.5 kcal/mol; in ice, the bond strength is 6 kcal/mol. The description of the drug receptor interaction based on hydrogen bonds is closely related to the importance of these bonds in maintaining the integrity of biological systems and in determining the physicochemical properties of drug molecules. The physical state of substances, such as water, DNA, protein, and various drug molecules, are maintained by hydrogen bonding. The most frequently observed H-bonds in biological systems are between the hydroxyl (OH) and amino (NH) groups. In the DNA helix, hydrogen bonding links the complementary base pairs of adenine-thymine and guanine-cytosine. Since the physical and chemical properties of a compound may be greatly altered by hydrogen bonding, it is reasonable to expect that it may also have a significant effect and some correlation with biological properties. In a number of cases, such a correlation is present (Tables 3.2 and 3.3). Table 3.2 Differences between 1-phenyl-3-methy-5-pyrazolone and 1-phenyl-2, 3-dimethy-5pyrazolone (antipyrine). Table 3.3 Differences between o-hydroxybenzoic acid and p-hydroxybenzoic acid. 1-Phenyl-3-methy-5-pyrazolone forms intermolecular hydrogen bonding The alkylating agents (nitrogen mustards) are thought to act by replacing the weak and reversible H-bonds between adjacent nucleic acid strands with strong and relatively irreversible covalent bonds. In this way, nucleic acid regeneration and cell division in the rapidly proliferating cancer cells may be inhibited. Antipyrine, 1-phenyl-2,3-dimethyl-5-pyrazolone has analgesic activity, but 1-phenyl-3-methyl5-pyrazolone is inactive. This is due to the formation of hydrogen bonding in the 1-phenyl-3-methyl5-pyrazolone and gives rise to a linear polymer, which cannot pass through biomembranes. Salicylic acid (o-hydroxy benzoic acid)has antibacterial activity, but not the p-isomer and m-isomers, that is, p and m-hydroxy benzoic acids are inactive. This is because salicylic acid form intramolecular hydrogen bonding, therefore, it is less water-soluble and its partition coefficient is also greater. The mand the p-isomer can form intermolecular H-bonds, results in dimer, and does not easily pass through the biomembranes. The partition coefficient of it is also less, and hence, low antibacterial action.