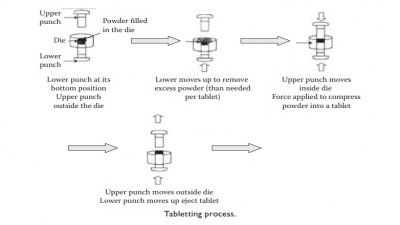
Evaluation of tablets
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Tablets
The compendia, such as the United States Pharmacopeia (USP), and the regulatory bodies, such as the United States Food and Drug Administration (FDA), in addition to historic product-development experience, inform the desired quality attributes of the tablets. Tablets are usually tested for the following characteristics:
Evaluation of
tablets
The
compendia, such as the United States Pharmacopeia (USP), and the regulatory
bodies, such as the United States Food and Drug Administration (FDA), in
addition to historic product-development experience, inform the desired quality
attributes of the tablets. Tablets are usually tested for the following
characteristics:
Appearance
All
tablets should have identical size, shape, thickness, color, and surface
markings. The general appearance of the tablet allows monitoring a lot-to-lot
and tablet-to-tablet uniformity. Tight control of tablet thickness is required
to ensure automated machine operations during its packaging and handling.
Tablet-to-tablet thickness within a batch and average thickness of tablets
across all batches are defined and controlled.
Uniformity of content
All
tablets must be demonstrated to contain the labeled active ingredient and there
should be tablet-to-tablet uniformity in drug content. This is usually tested
by an analytical method for drug potency (such as high-performance liquid
chromatography) in a several individual tablets.
Hardness
Tablet
hardness refers to the amount of force required to diametrically crush a
tablet. It is representative of the tensile strength of a tablet and is
determined by the cohesion characteristics of the powder blend. Tablet hardness
impacts tablet disintegration, dissolution, and friability. If tablets are too
hard, they may not disintegrate within a reasonable period of time. This can
lead to reduced bioavailability and failure to meet the dissolution
specification. If they are too soft, then they may not withstand the handling
and shipping operations, leading to tablet breakage or chipping (breaking away
from edges) and failure during friability testing. Friability is the ten-dency
of the tablets to chip or break by tumbling motion.
Friability
Tablet
friability represents the tendency of a tablet to shed powder or break into
smaller pieces under mechanical stress, such as falling from a fixed dis-tance.
It is a function of the fragility of the compressed powder blend, tab-let
shape, cohesion, and hardness. Low tablet friability is desired to ensure its
physical integrity during packaging, shipment, and handling.
Weight uniformity
Tablets
are compressed at a predefined weight. Under the assumption of normality of
statistical distribution of tablet weight, all tablets are required to be
within a certain range of the predefined tablet weight. Several tablets are
weighed individually, and both the average weight and variation of indi-vidual
tablet weight from the average are calculated and controlled during the
manufacturing to ensure that the tablets contain the desired amounts of drug
substances, with no more than acceptable variation among tablets within a
batch.
Disintegration
Disintegration
of tablets is evaluated to ensure that the tablet dissolves or breaks apart
into smaller particles or granules on contact with water under agitation. This
allows the DS to dissolve from its primary particles, being fully available for
dissolution and absorption from the GI tract. Tablet dis-integration is
evaluated in a standardized apparatus that subjects six tablets to a defined
mechanical stress in individual reciprocating cylinders in a suitable aqueous
medium at 37°C, to reflect the conditions on oral inges-tion. The time it takes
for the last of six tablets to disintegrate into smaller particles and
disappear from the reciprocating cylinders is called disinte-gration time. The disintegration media required varies
depending on the type of tablets to
be tested. The disintegration time is generally not more than 15 min for IR
tablets.
The
disintegration test is used as a control for tablets intended to be
admin-istered by mouth, but not for the tablets intended to be chewable and SR.
Dissolution
As
drug absorption and physiological availability depend on having the DS in the
dissolved state at the site of absorption, dissolution, also termed drug
release, is an important property of tablets. The rate and extent of
dissolution of a drug are tested in vitro
by a suitable dissolution test. Dissolution is used as both a quality control
tool to ensure batch-to-batch and tablet-to-tablet uniformity in drug-release
characteristics of the tab-lets and sometimes also as a tool for in vitro–in vivo correlation (IVIVC) of
drug release (in vitro) and drug
absorption (in vivo). Dissolution
test provides a means of control in ensuring that a given tablet formulation is
similar with respect to the rate and extent of drug release as the batch of
tablets were shown initially to be clinically effective.
Related Topics