Free-Radical Polymerization
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Carbon-Carbon Bond Formation By Free-Radical Reactions
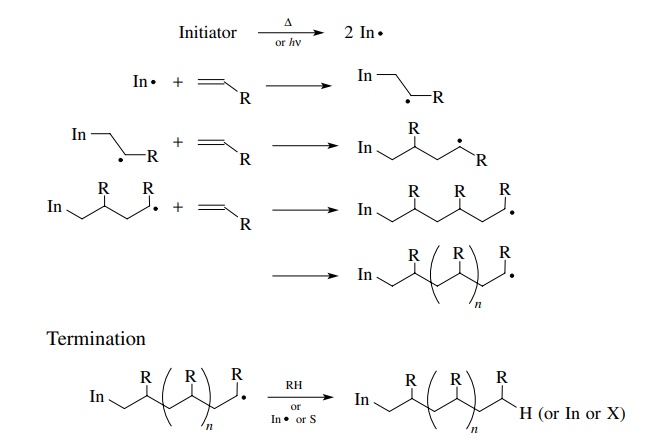
The first major use of free radicals was in olefin polymerization reactions. Polymerization reactions are amenable to free-radical initiation for several reasons.
FREE-RADICAL POLYMERIZATION
The
first major use of free radicals was in olefin polymerization reactions.
Polymerization reactions are amenable to free-radical initiation for several
reasons. First the olefin is the only reagent present so as to minimize
competing reactions. Second the initiator radical is produced by heat or light
or catalysis in the pres-ence of a huge excess of the olefin. Under these
conditions free-radical addition to the double bond is virtually the only process
that occurs. Moreover, the new radical species resulting from olefin addition
is also produced in the presence of a huge excess of olefin so that it adds to
another olefin molecule to give a larger free radical. The process continues.
By controlling the purity of the starting olefin and the reaction conditions so
that terminations are rare, chain lengths in the tens of thousands can be
achieved. This leads to the formation of thousands of carbon–carbon bonds per
polymer molecule and extremely long polymer chains.
As
a consequence of the fact that free-radical reactions are chain processes, they
are very well suited for the preparation of polymers rather than single
products. That is, products are obtained whose size is determined by the
number of propagation cycles that occur before a termination event stops the
growing chain.
If
the number of propagation cycles is between 200 and 300, then the product
mixture will contain molecules which contain between 200 and 300 monomers. It is
more reasonable to describe the product mixture in terms of the “average
molecular weight” rather than a single product with a discrete molecular
weight. The physical properties reported for a polymer are those of a mixture
of polymeric molecules rather than of a single polymeric compound.
Free-radical
polymerization was a mainstay of the plastics industry for many years. While
new and better methods have been developed for the polymerization of many
substrates, free-radical polymerization is still used for the preparation of
many plastics and composites. The success of these methods is based on an
under-standing of the process. Huge amounts of effort have been expended in
finding initiation reactions that produce free radicals controllably and
reproducibly. The reaction environment has been studied intensively so that
propagation reactions are maximized and termination events minimized. Finally
the rational control of termination reactions, which are necessary to control
the chain length and thus the average size of the polymer molecules produced,
has been successfully developed. It is important to emphasize that the
properties of the product mixture were the gauge by which the understanding and
control were measured.
Thus
it is not necessary to produce one product molecule with a defined molecular
weight. It is only necessary to obtain a product mixture whose average
molecular weight and physical properties fall within a defined range. Now it is
true that obtaining a more narrow range of molecular weights in a polymer leads
to much more consistent physical properties. This explains why so much effort
was made to control free-radical polymerization. Those efforts played a large
role in the creation of the “plastic society” in which we live.
Related Topics
