Gene therapy
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Biotechnology-based drugs
Gene therapy is a method for the treatment or prevention of disease that uses genes to provide the patient’s somatic cells with the genetic information necessary to produce specific therapeutic proteins needed to correct or to modulate a disease.
Gene therapy
Gene
therapy is a method for the treatment or prevention of disease that uses genes
to provide the patient’s somatic cells with the genetic information necessary
to produce specific therapeutic proteins needed to correct or to modulate a
disease. The promise of gene therapy is to overcome limitations associated with
the administration of therapeutic proteins, including low bio-availability,
inadequate pharmacokinetic profiles, and high manufacturing cost. Gene therapy
approaches are utilized for treating genetic and acquired diseases.
Two
approaches are currently used for gene transfer: viral and nonviral. Viral gene
transfer utilizes a genetically modified natural virus with a part of the viral
genome replaced by a therapeutic gene (called transgene) and making the virus replication deficient. Viruses have
evolved to efficiently penetrate cells and transfer their genetic material into
host cells (a process called transduction).
Thus, a viral vector efficiently transfers the desired genetic material into
target cells, leading to transgene expression. Several different viral vectors
have been developed for gene therapy, including ret-rovirus, adenovirus,
adeno-associated virus (AAV), and herpes simplex virus (HSV). The advantages
and disadvantages of different viral vectors are listed in Table 26.1.
Table 26.1 Characteristics of viral vectors

AAVs
are the most common viral vectors used in the clinic. This is mainly due to
their low safety risk compared to other viral vectors. Adenoviruses and AAVs do
not integrate their genes into the host cell genome. Retroviruses, on the other
hand, integrate their genetic material with the host cell genome. Thus, the
duration of gene expression is much longer with retroviruses (weeks to months)
as compared to adenoviruses and AAVs (days to weeks).
Retroviral vector
Retroviral
vectors are RNA viruses (i.e., their genome is RNA) possessing the main feature
of reverse transcribing their viral RNA genome into a double-stranded viral
DNA. Retroviral vectors can stably insert into the host DNA. The retroviral
genome consists of three encoding regions (por-tions of DNA strand that code
for specific functional proteins) responsible for viral replication: (1) gag region, encoding group-specific
antigens and proteins; (2) pol
region, encoding reverse transcriptase; and (3) env region, encoding viral envelope protein. These regions are
flanked on either side by a long terminal repeat (LTR) region. LTRs are
responsible for regulation and expression of the viral genome.
These vectors can carry foreign genes of <8 kb (kilo base pair length). They carry an inherent risk of mutagenesis by inserting their genome (called insertion mutagenesis) within a functional gene, which can compromise the functionality of a critical normal human protein.
Defective retroviral vectors
are devoid of the genes encoding viral pro-teins but retain the ability to
infect cells and insert their genes into the chromosomes of the target cells.
Members of this class include the Moloney murine leukemia viruses (MuLVs) and
the lentiviruses.
1. MuLV
MuLV
consists of three functional genes: gag,
pol, and env, flanked by the viral LTRs. Removing these structural genes and
inserting therapeutic genes in their place make muLV-based vectors.
MuLV-derived vectors inte-grate exclusively in dividing cells.
2. Lentiviruses
The
human immunodeficiency virus (HIV) is a lentivirus and is known to cause AIDS.
Their special ability to infect and integrate into nondividing cells has application
for the construction of lentiviral vectors for gene deliv-ery into nondividing,
terminally differentiated cells such as neuronal tissue, hematopoietic cells,
and myofibers.
Adenoviral vectors
Adenoviruses
are nonenveloped DNA viruses carrying linear double-stranded DNA of about 35 kb
length. The base pair length genome carrying capacity of a virus is limited by
the size of genome that can be accommodated within the capsid. Adenoviral
vectors infect both dividing and nondividing cells. Adenoviral vectors do not
integrate into the host cell chromosomes. Genes introduced into cells using
adenoviral vectors are maintained extrachromosomally in the nucleus and
provides transient transgene expression.
For
producing transgene-containing therapeutic adenoviruses, their genome is
modified by deletion of the viral replication specific gene known as early gene
1 (E1A). This also creates space for
the insertion of the desired gene. Adenoviral vectors are based on natural
adenoviruses of serotypes 2 and 5. In these first generation
adenoviral-vectors, additional partial dele-tions of E1B and E3 genes can be
made to create more space for transgene insertion.
An
advantage of adenoviruses over retroviral vectors is achievement of very high
viral titers. This suggests efficient gene transfer. Their key disad-vantages
are their episomal (extrachromosomal) status in the host cell that permits only
transient expression of the therapeutic gene. Furthermore, expression of the E2
viral protein provokes inflammatory reactions and toxicities that limit
repeated application of adenoviral vector for therapeu-tic benefit.
Adeno-associated virus vectors
AAV
is a single-stranded DNA virus and belongs to the family of parvovi-ruses. For
example, AAV-2 is a nonpathogenic human virus, and the wild type AAV-2 genome
establishes a latent infection in human cells, where the viral genome
integrates into the chromosomal DNA in a site-specific manner. AAV requires an
adenovirus or a herpes virus for viral replica-tion. Compared to adenoviruses,
AAV has low immunogenicity. It has a limited capacity for insertion of foreign
genes ranging only from 4.1 to 4.9 kb. For construction of rAAV-based vectors,
the rep and cap genes (which are responsible for the production of proteins
that would replicate the virus or
produce structural proteins for the capsid)
are replaced by the therapeutic genes.
Herpes simplex virus vectors
Herpes
simplex virus 1 (HSV-1) is a DNA virus possessing a double-stranded linear
genome of 150 kb. The HSV affords large packaging capacity for insertion of
foreign genes. HSV-1 can infect both dividing and nondividing cells. HSV has
natural tropism toward neuronal cells, and this property can be exploited for
gene therapies for neuronal tumors. HSV-1 particles are relatively stable and
can be concentrated to high virus titers, which are valuable for low-volume
administration of a large number of viral particles. The virus does not
integrate into the host genome, and, therefore, exhibits transient gene
expression in infected cells.
Nonviral gene expression system: Plasmid vectors
Unlike
viral vectors, which have many inherent risks, such as inflamma-tion and the
potential to generate host immune response (both cellular and humoral),
plasmid-based nonviral vectors are fairly safe. As illustrated in Figure 26.4, three essential components of gene medicines are a therapeutic gene that encodes a
specific therapeutic protein; a gene expression system that recruits host cell
machinery to allow the transcription of the encoding gene within a target cell;
and a gene delivery system that translocate the expression system to specific
location within the body and across the cell membrane barrier.
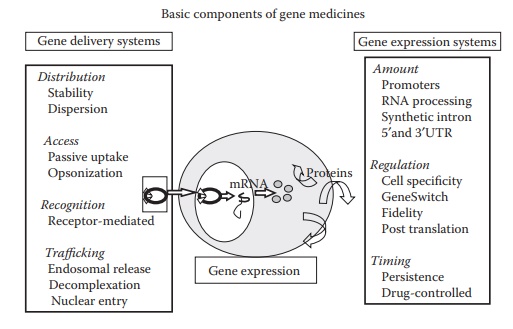
Figure 26.4 Basic components of a nonviral gene medicine. Therapeutic gene, gene delivery system, and gene expression plasmid are the three basic components of a nonviral gene medicine.
The
gene and the gene expression system are the components of plas-mid DNA, which
is a circular double-stranded DNA molecule. Basic components of a gene
expression plasmid are illustrated in Figure 26.5.
Plasmid-based gene expression systems contain a cDNA sequence coding for a
therapeutic gene and several other genetic elements, including introns, polyadenylation
sequences, and transcript stabilizers to control transcrip-tion, translation,
and protein stability. Optional components can be added
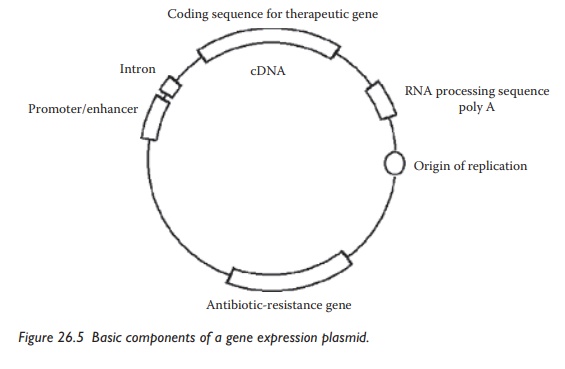
Figure 26.5 Basic components of a gene expression plasmid.
The gene delivery system distrib-utes the
plasmid to the desired target cells and promotes its internalization into the
cells. Once inside the cytoplasm, the plasmid can then translocate to the
nucleus, where gene expression begins through the natural cellular processes of
transcription and translation.
Gene delivery systems
Plasmid
DNA is a long polyanionic polymer. Depending on the number of base pairs, its
hydrodynamic size can range from 100 to 200 nm. As the cell membrane is
negatively charged, electrostatic repulsion is a bar-rier to the cell membrane
translocation of the plasmid DNA. This barrier is commonly overcome through the
use of polycationic lipids, polymers, or lipopolymers utilized as gene delivery
systems. Most commonly used synthetic gene carriers are cationic polymers and
lipids, which condense plasmids into small particles and protect them from
degradation by nucle-ases. These positively charged lipids, polymers, or
lipopolymers interact with the negatively charged plasmid DNA in aqueous
solution to form condensed colloidal particles with low hydrodynamic diameter
and an overall positive charge, which have higher cellular uptake. The
positively charged gene delivery systems can have interactions with other
physiologi-cal proteins that are negatively charged, leading to toxicities. The
apparent potency of a plasmid is further reduced by its chemical, enzymatic,
and colloidal instability, sequestration by cells of the immune system, uptake
and adsorption by nontarget cells and structures, access to target tissues,
cellular uptake, and trafficking to the nucleus of the cells. Therefore, there
is a growing need for novel delivery systems, which should be safe for repeated
administration.
1. Lipid-based gene delivery
Plasmids
may be incorporated into cationic or neutral liposomes. With the right
selection of lipids for making liposomes, the liposomes can be made pH
sensitive so that they are fusogenic (i.e., fuse with the cell membrane) at
acidic pH. This feature has been used to facilitate the endosomal disruption
and subsequent release of plasmids in the cytoplasm. The cellular uptake
process involves incorporation of a plasmid in an intracellular organelle
called endosome. The endosome slowly acidifies its contents in an attempt to
degrade its contents. The acidified endosome is called a lysosome. The
fusogenic lipids fuse with the lysosomal cell membrane as the pH becomes
acidic, thus disrupting the lysosome and releasing its cargo. Thus, the
plas-mid DNA escapes into the cytoplasm without getting degraded within the
lysosome.
Transfection
reagent LipofectinTM, for example, is a cationic liposome composed of 1:1 w/w
mixture of the cationic lipid N[1-(2,3-dioleyloxy)
propyl]-N,N,N-trimethylammonium
chloride (DOTMA) and the colipid dioleoyl phosphatidylethanolamine (DOPE).
Cationic lipids interact elec-trostatically with the negatively charged
phosphate backbone of DNA, neu-tralizing the charges and promoting the
condensation of DNA into a more compact structure. Usually, cationic lipids are
mixed with a zwitterionic or neutral colipid such as DOPE or cholesterol,
respectively to form liposomes or micelles. The cationic lipid and colipid are
mixed together in chloroform, which is then evaporated to dryness. Water is
added to the dried lipid film, and the hydrated film is then either extruded or
sonicated to form cationic liposomes. Cationic liposomes have also been
prepared by an ethanol injec-tion technique, whereby lipids dissolved in
ethanol at a high concentration are injected into an aqueous solution to form
liposomes.
As
shown in Figure 26.6, the general structure of
a cationic lipid has three parts: (1)
a hydrophobic lipid anchor group,
which helps in forming liposomes (or micellar structures) and can interact with
cell membranes; (2) a positively charged headgroup,
which interacts with plasmid, leading to its condensation; and (3) a linker group that connects the lipid
anchor with the charged headgroup. The net charge of the complex has a
signifi-cant effect on transection efficiency (i.e., efficiency of cellular
transfer of plasmid DNA) and DNA stability. Usually, positively charged
complexes show high transfection efficiency in
vitro. The relative proportions of each component and the structure of the
head group influence the physicochemi-cal properties of liposome/plasmid
complexes.
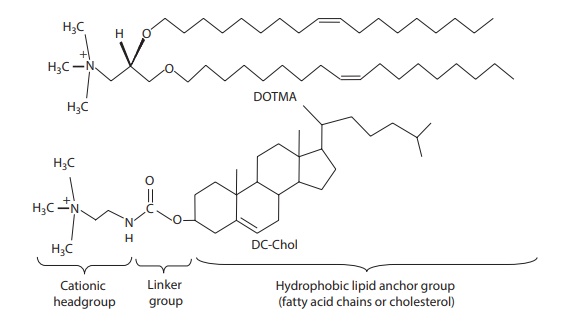
Figure 26.6 Basic components of a cationic lipid. (a) hydrophobic lipid group, (b)
linker group, and (c) cationic headgroup.
2. Peptide-based gene delivery
For
site-specific delivery of plasmids, positively charged macromolecules, such as
poly(l-lysine) (PLL), histones, protamine, or poly(l-ornithine) may be linked
together to a cell-specific ligand and then complexed to plasmids via
electrostatic interaction. The resulting complexes retain their ability to
interact specifically with target cell receptors, leading to receptor-mediated
internaliza-tion of the complex into the cells. Receptor ligands currently
being investigated include glycoproteins, transferrin, polymeric
immunoglobulin, insulin, epider-mal growth factor (EGF), lectins, folate,
malaria circumsporozoite protein, α2-macroglobulin, sugars, integrins
(asp-gly-asp [RGD] peptides), thrombo-modulin, surfactant protein A and B,
mucin, and the c-kit receptor.
Site-specific
gene delivery and expression are influenced by the extent of DNA condensation,
the method of complexation, the molecular weights of both polycations and
plasmids, and the number of ligand residues bound per polycation molecule. To
avoid high cytotoxicity, molecular heteroge-neity, and possible immunogenicity
of PLL and polyethylenimine (PEI), molecularly homogenous lysine and
arginine-rich peptide-based gene delivery systems are being investigated.
Peptides with moieties that pro-vide cooperative hydrophobic behavior of the
alkyl chains of cationic lipids would improve the stability of the peptide-based
DNA delivery systems. Short synthetic peptides containing the first 23 amino
acids of the HA2 subunit of influenza hemagglutinin protein (HA) are attractive
because of their pH-dependent lytic properties, with little activity at pH 7
but greater than or equal to a 100-fold increase in transfection efficiency at
pH 5.
3. Polymer-based gene delivery
Polymeric
biomaterials can be tailored to interact more on cellular and protein levels to
achieve high degrees of specificity, activity, and functional-ity. These
polymeric materials include (1) polymers that can be covalently attached to
proteins and antibodies to form drug conjugates, (2) stimuli sensitive
polymers, (3) polymer/cell matrix, (4) functional biodegradable polymers, and
(5) polymeric gene carriers. These polymers are being uti-lized for the
delivery of proteins, ODNs, and genes. Noncondensing poly-mers, such as
polyvinylpyrrolidone (PVP) and pluronics, can also be used for delivery of
nucleic acids to muscles and tumors. These polymer-based DNA formulations are
hyperosmotic and result in an improved dispersion of plasmids through the
extracellular matrix of solid tissues, such as mus-cles or solid tumors,
possibly by protecting plasmids from nuclease degra-dation, dispersing plasmids
in the muscle, and facilitating their uptake by muscle cells. Synthetic
polymers offer a wide array of choices in influencing different aspects of DNA
condensation, targeting, cellular uptake, intracel-lular release, and
bioactivity.
Related Topics
