Multiplication of Human Viruses
| Home | | Pharmaceutical Microbiology | | Pharmaceutical Microbiology |Chapter: Pharmaceutical Microbiology : Viruses
The objective of the replication cycle is to ensure the multiplication of the virus with the formation of identical viral progeny. Viruses differ in their replication cycle and the time to produce and release new virions.
MULTIPLICATION OF HUMAN VIRUSES
The objective of the
replication cycle is to ensure the multiplication of the virus with the
formation of identical viral progeny. Viruses differ in their replication cycle
and the time to produce and release new virions. The multiplication cycle of
human viruses is generally slow, from 4 to more than 40 hours. Bacterial
viruses are generally faster and can take as little as 20 minutes to replicate
within the bacterial host. The replication cycle can be divided into six
distinct phases (Figure 5.4) that are common to all viruses, although detail
within each phase varies greatly between viruses. Understanding the viral
multiplication process is crucial for the development of new antiviral drugs.
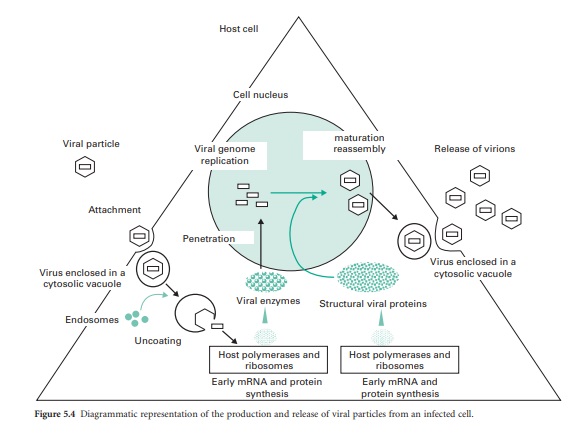
1) Attachment To The Host Cell
Viral attachment to the
cell surface can be divided into three phases: (1) an initial contact mainly
dependent on brownian motion, (2) a reversible phase during which electrostatic
repulsion is reduced and (3) irreversible changes in virusreceptor–hostreceptor
configuration that initiates viral penetration through the cell membrane.
All viruses possess receptors on their surface, usually in
the form of glycoproteins embedded in the viral envelope or protruding as spike
from the viral capsid. These structures recognize and bind receptors on the
host cell and provide the virus with its high specificity although different
viruses might share the same receptor. The virus–cell recognition event is
similar to any protein–protein interaction in that it occurs through a
stereospecific network of hydrogen bonds and lipophilic associations. For
example, the haemagglutinin receptor of influenza virus binds the terminal
glycoside residues of gangliosides (cell surface glycolipids) of the target
cell leading directly to the virus particle adhering to the cell. Similarly,
the interaction between the HIV receptor (i.e. gp120) and the Tlymphocyte
receptor (i.e. CD4) has been intensively studied.
2) Penetration Of The Viral Particle
Following the
irreversible attachment of the virus to the host cell, penetration of the virus
through the cell membrane is initiated following two energydependent mechanisms,
endocytosis or fusion. A third mechanism has been identified in some
bacteriophages that can inject their nucleic acid inside the bacterium (see
section 8.1). During endocytosis, the association between virus receptor and
host receptor triggers a number of mechanisms that draw the cell membrane to
engulf the virus particle forming a cytosolic vacuole. This process is
widespread among nonenveloped viruses, but is also used with some enveloped
viruses such as influenza (orthomyxoviruses). Certain enveloped viruses (e.g.
herpes simplex virus, HIV) can penetrate following fusion of their envelope
with the host cell membrane, liberating the viral capsid within the cell
cytoplasm.
3) Uncoating Of The Viral Particle
Following penetration of
the virus in a vacuole or directly into the cell cytoplasm, the viral nucleic
acid then needs to be released from the capsid/coat(s) to initiate viral
replication. This is the uncoating
process. For viruses that penetrate by endocytoses, the acidification of the
cytosolic vacuoles following endosome fusion induces a conformational change in
the capsid and the release of viral nucleocapsid (some helper proteins are
associated with the viral nucleic acid) into the cytoplasm. For certain
viruses, such as reovirus, only a partial uncoating is necessary for the
expression of the viral genome. The release of the nucleocapsid from vacuoles
can occur in the cytoplasm, close to the nucleus or within the cell nucleus.
4) Replication Of Viral Nucleic Acids And Translation Of The Genome
This stage of viral
replication ensures that (1) the host cell synthesis machinery is taken over by
the virus, and
the viral genome is
replicated. The structure, size and nature of the viral genome are extremely
diverse and thus this stage of the viral multiplication cycle reflects this
diversity. Three main mechanisms are, however, common to all viruses: the
transcription of viral genes into viral mRNA, the translation of the viral
genome into proteins, and the replication of the viral genome. Early transcription
and translation usually occurring immediately after the release of the
nucleocapsid in the cytoplasm is also common, and ensures the production of
early proteins such as viral polymerases, and the hijacking of the cell
synthesis machinery. In addition, some viruses can encode for genes the
products of which regulate the host synthetic processes according to the needs
of the virus (e.g. tat gene in HIV).
The replication of the
viral genome depends on the type of nucleic acid carried by the virus. The
positive strand RNA in viruses such as the poliovirus can be used directly as
mRNA following the acquisition of a terminal sequence from the host cell.
Negative strand RNA (e.g. in influenza virus) is transcribed into a positive
RNA complementary in base sequence to the parent RNA using an RNAdependent RNA
polymerase carried by the virus. In ds DNA viruses (e.g. adenoviruses), the
nucleic acid passes into the nucleus where it is usually transcribed by a host
DNAdependent RNA polymerase. In some viruses (e.g. poxvirus), this enzyme is
contained within the virus and released during uncoating, allowing the viral
genome to be replicated in the cell cytoplasm. In retroviruses (e.g. HIV), a
single stranded proviral DNA is produced from the viral ss RNA by a viral
enzyme called reverse transcriptase. This unique enzyme acts both as an RNA and
DNA directed DNA polymerase, and has associated RNAase activity. The proviral
DNA can be transported to the cell nucleus where it can be integrated within the
cell host genome by a viral integrase.
One important difference
between the host cell and the virus is in the nature of their mRNA. Host cell
mRNA encodes directly for functional proteins, whereas viral mRNA is polycistronic, which means several distinct
proteins are encoded within a single piece of mRNA. This implies that the virus
needs to use a virus-specific protease to cut at the correct place the polyprotein produced by translation to
restore the functionality of viral proteins.
Late protein synthesis
during the replication cycle concerns the production of structural components
(e.g. capsomeres) of the new virions.
5) Maturation Or Assembly Of Virions
Towards the end of the
multiplication process, large amounts of viral materials accumulate within the
host. Viral capsid starts to form from individual structural proteins. In
certain viruses (e.g. poliovirus) the capsid self-assembles. The replicated
viral genome and some viral proteins become packaged within the capsid. Most
nonenveloped viruses accumulate within the cytoplasm or nucleus and are only
released when the cell lyses. Packaging of viral components can occur within
the cytoplasm or in the cell nucleus. For example, with influenza virus, the
capsomeres are transported to the cell nucleus where they combined with the
viral RNA and assemble into helical capsids. The envelope of enveloped viruses
originates from the host. With the influenza virus, viral proteins such as
neuraminidase and haemagglutinin migrate to the cell membrane, displacing cell
protein. The assembled nucleocapsids pass out from the nucleus to the cytoplasm
and as they impinge on the altered cytoplasmic membrane they cause it to bulge
and bud off completed enveloped particles from the cell. In the herpesvirus,
the envelope originates from the nucleus membrane. The nucleocapsid assembles
into the nucleus and it acquires its envelope as it passes through the inner
nuclear membrane. The complete virus is then incorporated into a vesicle which
migrates to the cell surface.
The maturation of
viruses and their assembly is not well understood at present. The presence of
chaperone proteins may play an important role in the interaction between the
viral nucleic acid and the structural proteins.
6) Release Of Virions Into The Surrounding Environment
At the end of the
multiplication process, the mature virions are released from the host cell.
This can occur in a number of different ways. For most nonenveloped viruses, the
virus progeny accumulates within the host cell cytoplasm and is released
following cell lysis. Some viruses (e.g. bacteriophages) produce a lytic enzyme
(peptide) or proteases to lyse the host enabling the release of infectious
particles, although the host often selfdisintegrates as it cannot maintain
normal housekeeping functions during a viral infection. Enveloped viruses are
usually released by a budding process over a period of hours. Ultimately the
host cell will die following damage to its metabolism and housekeeping
functions during viral replication.
Related Topics
