Post-Marketing Cardiovascular Safety Signal
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: NSAIDs - COX-2 Inhibitors – Risks and Benefits
At the time of market approval of celecoxib and rofecoxib, upper gastrointestinal safety information of these two drugs was based on endoscopy studies.
POST-MARKETING CARDIOVASCULAR
SAFETY SIGNAL
LARGE-SCALE CLINICAL TRIALS
At
the time of market approval of celecoxib and rofecoxib, upper gastrointestinal
safety information of these two drugs was based on endoscopy studies. As the
reduced frequency of mucosal injury in the upper gastrointestinal tract may not
correlate well with incidence of serious gastrointestinal events that include
ulcer, perforation, obstruction, or bleeding, manufacturers of celecoxib and
rofecoxib sponsored large-scale clinical trials that were powered to provide
definitive evidence on upper gastrointestinal safety for these drugs.
The
Celecoxib Arthritis Safety Study (CLASS) was the first large-scale randomized
trial of a COX-2 inhibitor (Silverstein et
al., 2000). Patients with osteoarthritis or rheumatoid arthritis were
randomly assigned to receive celecoxib, ibuprofen, or diclofenac for more than
six months (Table 47.1). Approximately 20% of the study subjects took low dose
aspirin (325 mg a day or less) for cardiovascular disease prevention during the
study. There was no signifi-cant reduction of risk of upper gastrointestinal
ulcer complications within the first 12 months of therapy (Hrachovec and Mora,
2001). Among patients who did not use aspirin, risk of upper gastrointestinal
ulcer complication was reduced by approximately 50%. In the first published
report of CLASS results, incidence of stroke, myocardial infarction, and angina
was virtu-ally the same in the celecoxib group and in the ibupro-fen/diclofenac
group during the first six months of therapy. No cardiovascular safety signal
was observed in this study.
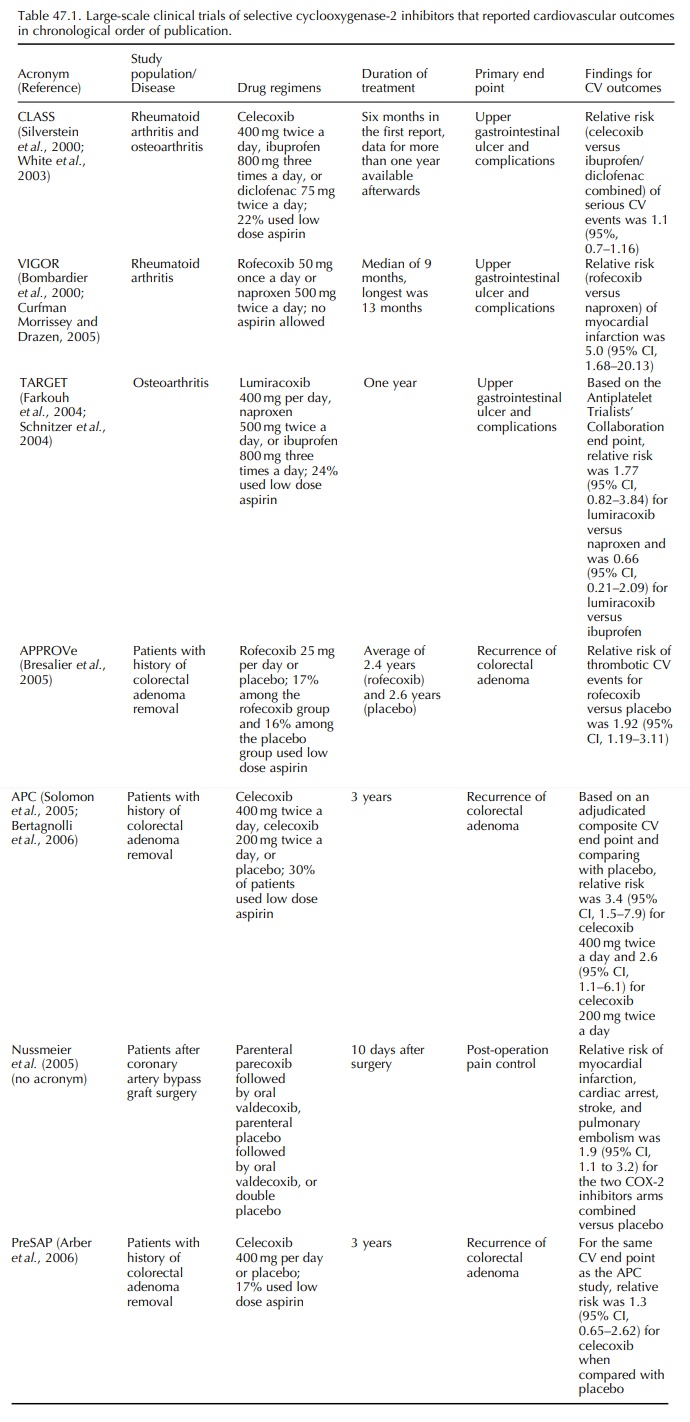
The
first cardiovascular safety signal of a COX-2 inhibitor came from the Vioxx
Gastrointestinal Outcomes Research Study (VIGOR) (Bombardier et al., 2000), which was another large
trial that evaluated the risk of
adverse upper gastrointestinal outcomes among patients on COX-2 inhibitor or
non-selective NSAID. Patients with rheumatoid arthritis were randomly assigned
to receive rofecoxib 50 mg once per day or naproxen 500 mg twice per day, with
a median follow-up of 9 months. Unlike the enrollment criteria for CLASS,
aspirin use was not allowed in VIGOR. Rofecoxib users had 60% lower risk of
perfo-ration, obstruction, and severe upper gastrointestinal bleeding than
naproxen users. However, reported inci-dence of myocardial infarction was
higher among the rofecoxib group (relative risk, 5.0; 95% confidence interval
[95% CI], 1.68–20.13) (Curfman, Morris-sey and Drazen, 2005). The VIGOR
investigators hypothesized that the increased relative risk among the rofecoxib
users could be a result of inherent cardio-vascular risk of rofecoxib,
cardio-protective effect of naproxen, or both.
Related Topics
