Purpose and Achievements of the Yellow Card Scheme
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: Spontaneous Reporting - UK
It is generally accepted (e.g. Amery, 1999) that it is not possible to detect all the adverse effects of a medicine during the pre-marketing clinical trials, because of a number of factors.
PURPOSE AND ACHIEVEMENTS OF THE
YELLOW CARD SCHEME
It
is generally accepted (e.g. Amery, 1999) that it is not possible to detect all
the adverse effects of a medicine during the pre-marketing clinical trials,
because of a number of factors. First, trials are generally small (on average
1500 patients for a new drug substance); although they will detect common side
effects, partic-ularly those that are predictable from the pharmacol-ogy of the
drug, they are too small to detect side effects that occur rarely (incidence of
1 in 10 000 or less). Additionally, medicines are used in clinical trials in a
very controlled manner, that is they are given for a limited duration, to
carefully selected patients who are closely monitored. This is in complete
contrast to the manner in which the medicine may be used once marketed, when it
may be used in patient populations for which it was not intended, may be given
for long periods of time, and in combination with other medicines.
It
is therefore vital to monitor the safety of medicines as used in routine
clinical practice through-out their marketed life, in order to detect those
side effects that are not identified through clinical trials.
The
best established way to do this is to collect reports of suspected adverse drug
reactions (ADRs) via a reporting Scheme such as the Yellow Card Scheme.
All
spontaneous reporting Schemes, including the Yellow Card Scheme, have a number
of limita-tions, perhaps the most significant of which is under-reporting (e.g.
Griffin and Weber, 1992; see the section on ‘Weaknesses of Yellow Cards’
below). Despite this, such Schemes have a proven track record as an ‘early
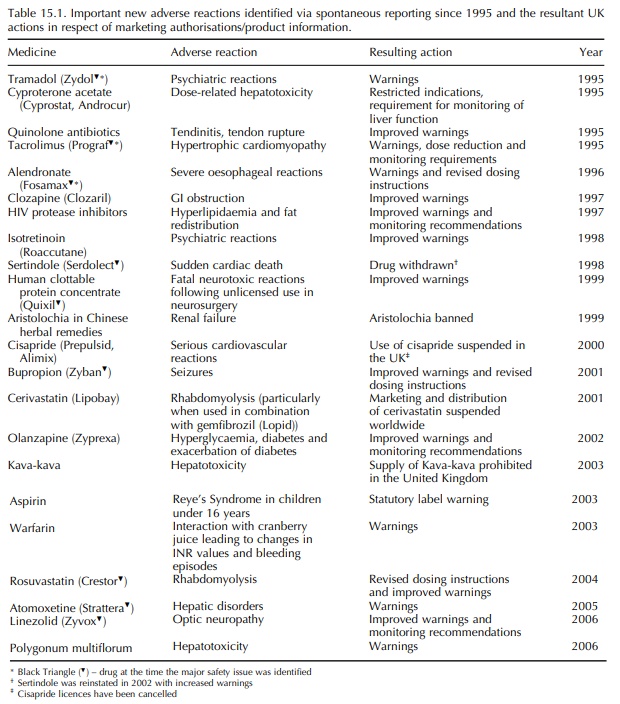
warning’ system for the identifi-cation of new drug safety hazards. Examples of
drug safety hazards identified through spontaneous report-ing have been
described previously (e.g. Rawlins, 1988b; Griffin and Weber, 1992). Examples
of ADRs identified via spontaneous reporting including Yellow Cards are shown
in Table 15.1.
Related Topics
