Reaction Energetics
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Mechanisms of Organic Reactions
To attack this problem, we have to begin with those species whose structures we know — the reactants and products.
REACTION ENERGETICS
To
attack this problem, we have to begin with those species whose structures we
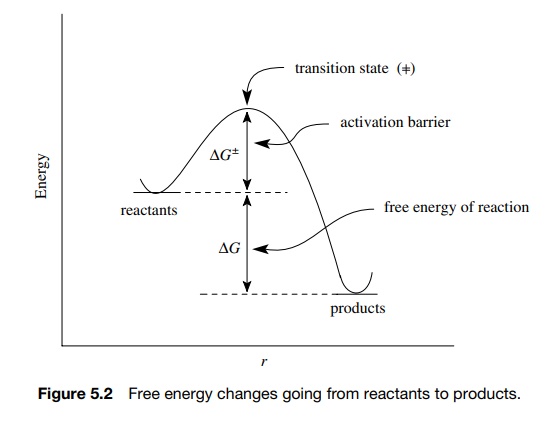
know — the reactants and products (Figure 5.2). Each is a stable collection of
atoms characterized by a free energy. The difference in free energy between the
reactants and products is ΔG — the free energy of reaction which indicates
whether the reaction takes place with evolution of energy (exergic) or with the
uptake of energy (endergic). Connecting reactants and products is an energy profile
which contains the activation barrier ΔG ‡ which must be surmounted if the
reaction is to proceed.
Now, for a given chemical transformation, there may be more than one step. In such cases each step of the transformation can be thought of as a discrete reaction and will have reactants and products and an activation barrier. Furthermore, each minimum on the energy curve between reactants and products is a more or less stable collection of atoms with a finite lifetime. These species are usually higher in energy than either the reactants or products and are called reaction intermediates. We have encountered many common reaction intermediates in reactions we have studied. These include carbocations, carbanions, enolates, free radicals, carbenes, and so on.
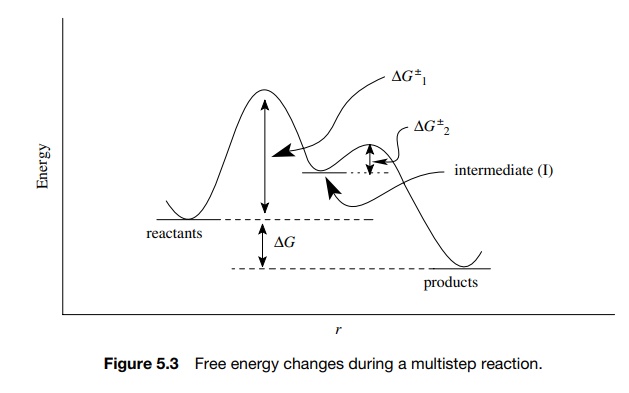
A multistep process involving intermediates still has an overall free energy of reaction ( ΔG ) (reactants vs. products), but each elementary step is a separate chemical
reaction and therefore each step has a free energy, an activation barrier (ΔG ±), a transition
state, and an activated complex corresponding to the chemical changes occurring
in that step of the process (Figure 5.3). The step with the transition state of
highest free energy is called the rate-determining step because only by passing
over the highest barrier can reactants proceed to products.
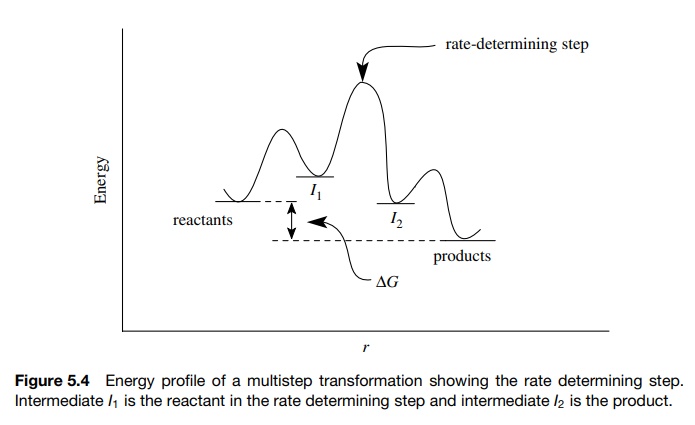
The
rate-determining step thus defines how fast reactants can be converted to
products (Figure 5.4). Steps in the process before the rate-determining step
con-tribute to the rate law for the reaction by contributing to the overall
barrier height. Steps after the rate-determining step have no bearing on the
rate of reaction.
Related Topics
