Regulation of Insulin Secretion
| Home | | Pharmacology |Chapter: Essential pharmacology : Insulin, Oral Hypoglycaemic Drugs and Glucagon
Under basal condition ~1U insulin is secreted per hour by human pancreas. Much larger quantity is secreted after every meal. Secretion of insulin from β cells is regulated by chemical, hormonal and neural mechanisms.
REGULATION OF INSULIN SECRETION
Under basal condition
~1U insulin is secreted per hour by human pancreas. Much larger quantity is
secreted after every meal. Secretion of insulin from β cells is regulated by
chemical, hormonal and neural mechanisms.
Chemical
The β cells have a glucose sensing mechanism dependent on
entry of glucose into cells (through the aegis of a glucose transporter GLUT2)
and its phosphorylation by glucokinase.
Glucose entry and activation of the glucoceptor indirectly inhibits the ATP sensitive
K+ channel resulting in partial depolarization of the β cells. This increases
intracellular Ca2+ availability (due to increased influx, decreased efflux and
release from intracellular stores) → exocytotic release of insulin storing granules.
Other nutrients that can evoke insulin release are—amino acids, fatty acids and
ketone bodies, but glucose is the principal regulator and it stimulates
synthesis of insulin as well. Glucose induces a brief pulse of insulin output
within 2 min (first phase) followed
by a delayed but more sustained second
phase of insulin release.
Glucose and other
nutrients are more effective in invoking insulin release when given orally than
i.v. They generate chemical signals ‘incretins’
from the gut which act on β cells in the pancreas to cause anticipatory
release of insulin. The incretins involved are glucagonlike peptide1 (GLP1),
glucose-dependent insulinotropic polypeptide (GIP), vasoactive intestinal
peptide (VIP), pancreozymin-cholecystokinin, etc.; but different incretin may
mediate signal from different nutrient. Glucagon and some of these peptides
enhance insulin release by increasing cAMP formation in the β cells.
Hormonal
A number of hormones,
e.g. growth hormone,
corticosteroids, thyroxine modify insulin release in response to glucose. PGE
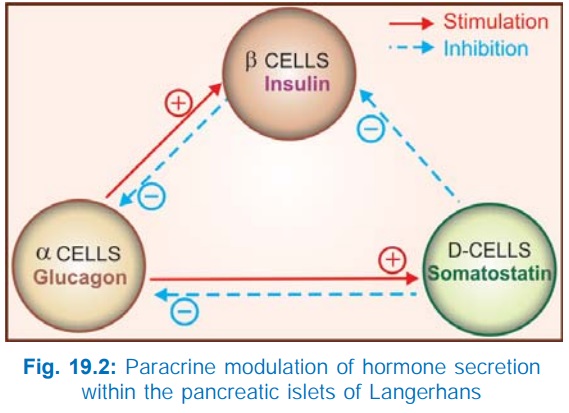
has been shown to inhibit insulin release. More important are the intra-islet
paracrine interactions between the hormones produced by different types of
islet cells. The β cells constitute the core of the islets and are the most
abundant cell type. The α cells, comprising 25% of the islet cell mass,
surround the core and secrete glucagon. The D cells (5–10%) elaborating
somatostatin are interspersed between the α cells. There are some
PP (or F) cells (pancreatic polypeptide containing) also.
·
Somatostatin inhibits release of both insulin
and glucagon.
·
Glucagon evokes release of insulin as well as
somatostatin.
·
Insulin inhibits glucagon secretion.
The
three hormones released from closely situated cells influence each other’s
secretion and appear to provide fine tuning of their output in response to
metabolic needs (Fig. 19.2).
Neural
The
islets are richly supplied by sympathetic and vagal nerves.
· Adrenergic α2 receptor activation
decreases insulin release (predominant) by inhibiting β cell adenylyl
cyclase.
· Adrenergic β2 stimulation increases
insulin release (less prominent) by stimulating β cell adenylyl
cyclase.
· Cholinergic—muscarinic activation by ACh or
vagal stimulation causes insulin secretion through IP3/DAGincreased
intracellular Ca2+ in the β cells.
These
neural influences appear to govern both basal as well as evoked insulin
secretion, because the respective blocking agents have effects opposite to that
mentioned above. The primary central site of regulation of insulin secretion is
in the hypothalamus: stimulation of ventrolateral nuclei evokes insulin
release, whereas stimulation of ventromedial nuclei has the opposite effect.
