SAR of Phenothiazines
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Tranquillizers
It is presumed that phenothiazines (neuroleptic) mediate their pharmacological effect mainly through interaction at D2 type dopamine receptors. 1. Modification of alkyl side chain 2. Amino group modification 3. Phenothiazine ring
SAR of Phenothiazines It is presumed that phenothiazines (neuroleptic) mediate their pharmacological effect mainly through interaction at D2 type dopamine receptors. Examination of X-ray structures of dopamine and chlorpromazine substituted with chlorine shows that these two structures can be partly superimposed. Chlorpromazine base could be superimposed on aromatic ring of dopamine base with sulphur atom aligned with p-hydroxy of dopamine. These substances are chemically constituted by lipophilic linearly fused tricyclic system having hydrophilic basic amino alkyl side chain. Activity of phenothazines is determined by the following: Nature of alkyl side chain at C-10. Amino group of side chain. Substituents on aromatic ring. Potency is maximum when there is three carbon between two ‘N’ atom (ring and side chain N ). Introduction of methyl group at C-1 decreases antipsychotic activity and produces imipramine-like activity. If C-1 is incorporated into cyclopropane ring imipramine-like activity is obtained. When oxygen is introduced into C-1 results in potent antidepressant effect. Example: Chloracizine. Addition of –CH3 at C-2 or C-3 has very little effect on activity. Bridging of position 3 of side chain to position 1 of phenothiazine nucleus decreases neuroleptic activity. 3° nitrogen shows maximum potency and 2° or 1° nitrogen shows reduced or abolished activity. N-alkylation with more than one carbon decreases activity. Activity is decreased when dimethylamino group is replaced by pyrolidinyl, morpholinyl, or thiomorpholinyl groups. However, piperidine or piperazine is more potent than dimethylamino group. Bridged piperidine derivates retain high degree of activity although bulky. Introduction of OH, CH3 , CH3CH2OH at C-4 of piperazine results in increased activity. Piperazine and phenothiazines may be esterified with long-chain fatty acids to produce slowly absorbed long-acting lipophilic prodrugs. Due to the slow release from oily deposition, significant activity is retained When N-4 piperazine substituents are as large as phenyl, ethyl, or p-amino phenyl ethyl (e.g. Azaspirane, Chlorspirane) are active. Substitution at C-2 position is optimal for neuroleptic activity. In general, potency at different positions increases in the following order 1 < 4 < 3 < 2. Potency of the various groups increase in the following order OH < H< CN < CH3 < Cl < CF3. Disubstitution (or) trisubstitution of the C-2 substituted drugs results in harmful potency. CF3 is more potent than Cl, but EPS appears, hence, chlorpromazine is much used, rather than triflupromazine. The electro-negative chlorine atom at C-2 is responsible for imparting asymmetry to this molecule and the attraction of the amine side chain towards the ring containing the chlorine atom indicate an important structural feature of such molecules. Oxidation of the sulphur at 5th position of antipsychotic phenothiazine decreases activity.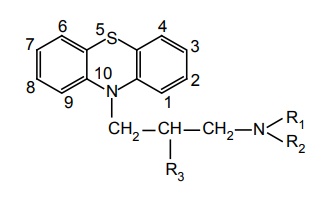
1. Modification of alkyl side chain
2. Amino group modification
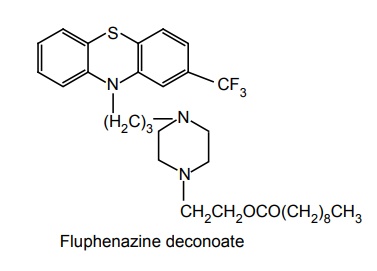
3. Phenothiazine ring
