Treatment of Leprosy
| Home | | Pharmacology |Chapter: Essential pharmacology : Antileprotic Drugs
Leprosy is a chronic granulomatous infection caused by Mycobacterium leprae ; primarily affecting skin, mucous membranes and nerves. It is more prevalent among the lowest socioeconomic strata.
TREATMENT OF LEPROSY
Leprosy is a chronic
granulomatous infection caused by Mycobacterium
leprae ; primarily affecting skin, mucous membranes and nerves. It is more
prevalent among the lowest socioeconomic strata. Many patients exploit it for
begging and do not come forward for treatment. In India, the National Leprosy
Control Programme was launched in 1955, and was changed to National Leprosy
Eradication Programme (NLEP) in 1982. With the use of multidrug therapy (MDT),
India has achieved elimination of leprosy as a public health problem (prevalence
rate < 1 case per 10,000 population) in Dec. 2005, though some states
(Bihar, West Bengal, Orissa, Chhatisgarh, Jharkhand, UP) still have >1 case
per 10,000.*
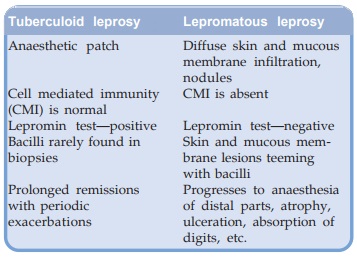
Two polar types—lepromatous (LL) and tuberculoid (TT) with 4
intermediate forms— borderline (BB), borderline lepromatous (BL), borderline
tuberculoid (BT) and indeterminate (I) of the disease are recognized. The
important features of the two polar types are:
For operational purposes, leprosy has been divided into:
Paucibacillary Leprosy (PBL) (Noninfectious):
This includes TT, BT, I and polyneuritic.
Multibacillary Leprosy (MBL) (Infectious): This includes LL, BL and BB.
Subsequently the definition of MBL has been widened to include
any active patient with > 5 lesions irrespective of results of skin smear
tests.
Conventionally, all forms of leprosy had been treated with
dapsone alone (monotherapy: MT) 100–200 mg daily, 5 days a week; duration of
treatment depending on the type: TT–4 to 5 years, LL–8 to 12 years or lifelong.
With this, symptomatic relief occurs in few months, but bacteriological cure is
delayed or may not occur. Emergence of dapsone resistance since 1964 threatened
the efficacy of monotherapy; upto 20% patients relapsed. Even primary dapsone
resistance was increasingly encountered. Monotherapy is no longer used.
Multidrug Therapy (MDT) Of Leprosy
To deal with dapsone resistant strains of M. leprae and the problem of microbial persisters (dormant forms),
multidrug therapy with rifampin, dapsone and clofazimine was introduced by the
WHO in 1981. This was implemented under the NLEP. The MDT is the regimen of
choice for all cases of leprosy. Its advantages are:
· Effective in cases
with primary dapsone resistance.
· Prevents emergence of
dapsone resistance.
· Affords quick symptom
relief and renders MBL cases noncontagious.
· Reduces total duration
of therapy.
Initially under standard MDT, the PBL cases were treated with
dapsone + rifampin for 6 months, while the MBL cases were treated with dapsone
+ rifampin + clofazimine for a minimum of 2 years or till disease inactivity/skin
smear negativity was achieved. The MBL cases were kept under surveillance
without treatment for the next 5 years.
A WHO expert group (1994) reviewed the data collected over the
past 12 years as well as results of clinical trials, and made the following
observations:
· MDT had been highly successful, both in MBL
and PBL. The estimated cases of leprosy fell from 10–12 million to 2.7 million.
·
Relapse rate after MDT had been 0.74% in MBL
and 1.09% in PBL over a period of 9 years.
· The efficacy, safety and acceptability of MDT had
been excellent.
· Some reports, mostly from India, had found
that for uniformly satisfactory response, treatment of PBL had to be extended
beyond the mandatory 6 months (mostly to 12 months). However, no difference in
the relapse rate was found among 12000 Indian patients treated with MDT either
for 6 months or for 1 year. As such, the WHO expert group recommended
continuation of 6 month MDT for PBL.
· No resistance to rifampin developed with MDT:
nearly all M. leprae isolated from relapse
cases remained fully sensitive to rifampin. No resistance to clofazimine had
been reported. New cases of drug resistance were not reported after application
of MDT. Retreatment of relapse cases with the same MDT had been successful, and
was recommended.
·
Drug toxicity had not been a major problem in
MDT.
·
Prevalence of lepra reaction had not increased
by MDT.
· No specific association of leprosy with HIV
infection had been found. Leprosy in HIVpositive cases is to be treated in the
same manner as in others.
On the basis of the above, the WHO (1994) recommended a ‘fixed
duration therapy’ (FDT) of 2 years for MBL and 6 month for PBL, whether disease
inactivity or skin smear negativity was attained or not.
MBL Encouraged by the very low relapse rates with 2 yrs FDT and keeping in view operational
constraints, studies were undertaken under the aegis of WHO to compare shortduration
(12 month) FDT with standard 24 month FDT. In the field situation the two were
found to yield similar relapse rates over 3–5 yr follow up. Accordingly, a WHO
expert committee on leprosy (1995) recommended shortening of MDT to 12 months.
This has been adopted by many countries and Govt. of India has implemented it
since 1999.*

Blister packs of tablets for 28 day treatment are made available
free of cost to all MBL cases, and 12 such blister packs have to be consumed by
each MBL patient. Separate blister packs are given to PBL cases and 6 packs are
to be taken by each patient.
A few studies, (mostly institutional) have shown that despite 2
yr MDT, some patients continue to harbour viable M. leprae (persisters). Relapse rates are higher in the later years
of follow up and in the subgroup of patients with large bacillary load, i.e.
bacillary index (BI) > 4 +. Thus, the length of MDT could depend on
the aim of therapy, resources, and feasibility of follow up.
The primary purpose of
mass programmes (WHO Action Programme for the Elimination of leprosy, or NLEP-India)
is to render patients noncontagious so as to cut down transmission. For this, 1
yr FDT may be considered adequate. Even if some patients are not totally cured
and relapse, they can be treated by reinstituting MDT (dormant bacilli remain
sensitive to the same drugs). This is more cost-effective than treating all
patients with a longer MDT to prevent a few relapses.
On the other hand, in
private or institutional care, the aim is cure of every individual patient. For
this extended treatment is required till disease inactivity or skin smear
negativity is achieved. Upto 4 years may be needed for this, particularly in
highly bacillated patients (BI > 4+).
PBL For PBL, 6 month 2 drug therapy has now been used for > 20 yrs with very encouraging
results. Field studies from various parts of the world suggest that this is
adequate, provided the patient is kept on follow up for the subsequent 1–2
years. However, institutional studies have found larger proportion of patients
to have active disease after 6 month FDT. Some reports indicate that proportion
of patients staying active can be reduced by 12 month MDT. Independent
leprologists prefer to extend therapy of PBL for 12 month or longer till
disease inactivity is achieved.
It may be concluded
that, where feasible, treatment till cure of individual patient should be
ensured both in MBL and in PBL.
Highlights Of Multidrug Therapy (MDT) Of Leprosy
· In 1985, leprosy was endemic in 122 countries
(prevalence rate >1 case/10,000 population). By implementing MDT this number
was reduced to 9 countries by the beginning of 2004, and the trend is
continuing.
· As per WHO, over 14
million cases of leprosy have completed MDT with very few relapses.
· Worldover the case
load of leprosy has fallen by >90%.
· At the beginning of
2004 only 0.46 million leprosy patients were registered globally, out of which
0.266 million were in India.
· Prevalence of leprosy
in India was 57.4 cases/10,000 population before implementing MDT, which fell
to 1.3 per 10,000 in March 2005, and to <1 per 10,000 in Dec. 2005.
Alternative Regimens
Many alternative regimens incorporating newer antileprotic drugs
have been investigated. However, these are used only in case of rifampin-resistance
or when it is impossible/inadvisable to employ the standard MDT regimen. Some
of these are:
· Cloflazimine 50 mg + any two of ofloxacin 400
mg/ minocycline 100 mg/clarithromycin 500 mg daily for 6 month, followed by
clofazimine 50 mg + any one of ofloxacin 400 mg/minocycline 100 mg daily for
additional 18 months.
· Four drug regimen of rifampin 600 mg +
sparfloxacin 200 mg + clarithromycin 500 mg + minocycline 100 mg daily for 12
weeks has yielded equivalent/better clinical improvement in MBL cases than
standard 12 month MDT.
· In case of refusal to accept clofazimine: ofloxacin
400 mg or minocycline 100 mg daily can be substituted for it in the standard
MDT. Use of ethionamide as a substitute is not recommended due to
hepatotoxicity of the latter.
· A multicentric trial© has shown that PBL cases
having few bacteria in the body and only one skin lesion can be treated with a
single dose of rifampin 600 mg + ofloxacin 400 mg + minocycline 100 mg (ROM
regimen). This regimen has been recommended by the WHO for patients with solitary
lesion PBL.
Many other shorter regimens are under evaluation.
Reactions In Leprosy
Lepra Reaction: These occur in LL,
usually with institution of
chemotherapy and/or intercurrent infection. It is a Jarish Herxheimer (Arthus)
type of reaction due to release of antigens from the killed bacilli. It may be
mild, severe or life-threatening (erythema nodosum leprosum).
Sufone Syndrome: It is the reaction which develops 4–6 weeks after dapsone treatment:
consists of fever, malaise, lymph node enlargement, desquamation of skin, jaundice
and anaemia. It is generally seen in malnourished patients.
Lepra reaction is of abrupt onset; existing lesions enlarge,
become red, swollen and painful; several new lesions may appear. Malaise, fever
and other constitutional symptoms generally accompany and may be marked.
Temporary discontinuation of dapsone is recommended only in
severe cases. Clofazimine (200 mg daily) is highly effective in controlling the
reaction (except the most severe one), probably because of its anti-inflammatory
property.
Other drugs used are—analgesics, antipyretics, antibiotics, etc.
according to need. Chloroquine and thalidomide also suppress lepra reaction.
Corticosteroids (prednisolone 40–60 mg/day till reaction is controlled, then
tapered over 8–12 weeks), should be used only in severe cases.
Reversal Reaction: This is seen in TT—is a manifestation of delayed hypersensitivity to M. leprae
antigens. Cutaneous ulceration, multiple
nerve involvement with pain and tenderness occur suddenly even after
completion of therapy. It is treated with clofazimine or corticosteroids.
Related Topics
