Types of Microbiological (Microbial) Assays
| Home | | Pharmaceutical Microbiology | | Pharmaceutical Microbiology |Chapter: Pharmaceutical Microbiology : Microbiological (Microbial) Assays: Antibiotics-Vitamins- Amino Acids
There are mainly two different types of microbiological assays usually encountered bearing in mind the response of an ever-growing population of microbes vis-a-vis ascertaining the profile of antimicrobial agent measurements, such as : (a) Agar Plate diffusion assays, and (b) Rapid-reliable-reproducible microbial assay methods.
TYPES OF
MICROBIOLOGICAL (MICROBIAL) ASSAYS
There are
mainly two different types of microbiological assays usually
encountered bearing in mind the response of an ever-growing population of microbes vis-a-vis ascertaining the profile
of antimicrobial agent measurements,
such as :
(a) Agar
Plate diffusion assays, and
(b) Rapid-reliable-reproducible
microbial assay methods.
Each of
the two aforesaid types of microbiological assays will
now be discussed individually in the sections that follows :
1. Agar Plate Diffusion Assays (Method-A)
In the agar-plate diffusion assays the ‘drug substance’ gets slowly diffused
into agar seeded duly with a susceptible
microbial population. Subsequently, it gives rise to a ‘specific zone of growth inhibition’.
However, the agar-plate diffusion
assay may be one-, two- or
three-dimensional (i.e., 1D, 2D or 3D).
All these
three different types shall now be
discussed briefly in the sections that follows :
1. One-Dimensional Assay
In this
particular assay the capillary tubes consisting of agar adequately seeded with ‘indicator organism’ are carefully overlaid with the ‘drug substance’. The drug substance e.g., an antibiotic normally gets diffused
downwards into the agar thereby giving rise to the formation of a ‘zone of inhi-bition’. However, this
specific technique is more or less obsolete now-a-days.
Merits : There are three points of merits, such as :
·
perfectly applicable for the assay of antibiotics anaerobically,
·
may efficiently take care of very small samples, and
·
exhibits an appreciable precision,
Demerit : It essentially has a critical
demerit with regard to the difficulty in
setting up and subsequent standardization.
2. 2D- or 3D-Assay
As to
date, the 2D- or 3D-assay methods represent the commonest and widely accepted
form of the microbiological assay.
Nevertheless, in this particular instance the samples need to be assayed are
adequately applied in a certain specific
type of reservoir viz., cup, filter-paper disc, or well, to a
thin-layer of agar previously seeded with an indicator microorganism aseptically in a Laminar Air Flow Bench. In
this way, the ‘drug substance’ gets
gradually diffused into the medium, and after suitable incubation at 37°C for 48–72 hrs. in an ‘incubation chamber’, a clear cut distinctly visible zone of growth inhibition comes into being*. However, the diameter of the zone of inhibition very
much remains within limits, provided
that all other factors being constant, and the same is associated with the concentration of the antibiotic present
in the reservoir.**
3. Dynamics of Zone Formation
It has
been duly observed that during the process
of incubation the antibiotic
gets diffused from the reservoir.
Besides, a proportion of the bacterial population is moved away emphatically
from the influence of the antibiotic
due to cell-division.
Important Observations : Following
are some of the important observations, namely
:
(1) Edge of a zone is
usually obtained in a situation when the
minimum concentration of the antibiotic that will effectively cause the
inhibition in the actual growth of the organism on the agar-plate (i.e.,
critical concentration accomplished)
attains, for the very first time, a specific population density which
happens to be excessively too big in dimension and quantum for it to inhibit effectively.
(2) The
precise and exact strategic position of the zone-edge is subsequently determined by means of the following three vital factors, such as :
·
initial population
density,
·
rate of diffusion
of ‘antibiotic’, and
·
rate of growth
of ‘organism’.
(3) Critical Concentration (C′) : The critical concentration (C′) strategically
located at the edge of a ‘zone of inhibition’ and formed duly
may be calculated by the following expression :
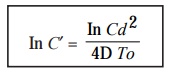
where,
C = Concentration of drug in Reservoir,
d = Distance between Reservoir and
zone-edge,
D =
Diffusion coefficient***, and
To = Critical time at which the
position of zone-edge was determined
critically.
Graphical Representation : It is
feasible and possible to have a
‘graphical representation’ to obtain
a zone of inhibition in different
ways, for instance :
(1) An assay wherein the value of To and D happen to be constant, an usual plot of In C Vs d2
for a definite range of concentrations shall, within certain limits, produce a ‘straight line’ that may be
conveniently extrapolated to estimate C′ i.e., critical concentration.
(2) In
fact, C′ duly
designates the obvious minimum value of
C
that would yield a specific zone of inhibition. Evidently, it is
absolutely independent of D and To.
(3) However,
the resulting values of D and To
may be manipulated judiciously to lower
or en-hance the dimensions of zone based on the fact that the concentrations of C
is always greater than C′. i.e.,
the concentration of ‘drug’ in reservoir > critical concentration of the ‘drug’.
(4) Pre-incubation would
certainly enhance the prevailing number and quantum of microbes present actually on the agar-plate ;
and, therefore, the critical population
density shall be duly accom-plished rather more rapidly (i.e., To gets reduced
accordingly) thereby reducing the observed zones
of inhibition.
(5) Minimizing
the particular microbial growth rate
suitably shall ultimately give rise to rela-tively ‘larger zones of inhibition’.
(6) Carefully
enhancing either the sample size or lowering the thickness of
agar-layer will critically increase
the zone size and vice-versa.
(7) Pre-requistes of an Assay—While
designing an assay, the following
experimental param-eters may be strictly adhered to in order to obtain an
optimized appropriately significant fairly
large range of zone dimensions spread
over duly the desired range of four
antibiotic concentrations, such as
:
·
proper choice of ‘indicator organism’,
·
suitable culture
medium,
·
appropriate sample
size, and
·
exact incubation
temperature.
4. Management and Control of Reproducibility
As the
observed dimensions of the zone of
inhibition depend exclusively upon a plethora of variables*, as discussed above, one should meticulously take great
and adequate precautionary meas-ures not only to standardise time, but also to accomplish reasonably desired good precision.
Methodologies : The various steps involved in the management and control of reproducibil-ity
are as stated under :
(1) A
large-size flat-bottomed plate [either 30 × 30 cm or 25 × 25 cm] must be
employed, and should be meticulously levelled before the agar is actually
poured.
(2) Explicite
effects of variations in the ‘composition of agar’ are adequately
reduced by pre-paring, and making use of aliquots
of large batches.
(3) Inoculum dimension variants
with respect to the ‘indicator
organisms’ may be minimized proportionately
by duly growing a reasonably large volume of the organism by the following two
ways and means, such as :
·
dispensing it accordingly into the aliquots just
enough for a single agar plate, and
·
storing them under liquid N2 so as to preserve its viability effectively.
(4) In
the specific instance when one makes use of the ‘spore inocula’, the same may be ad-equately stored for even longer
durations under the following two
experimental parameters, for instance :
·
absolute inhibition
of germination, and
·
effective preservation
of viability.
(5) It is
a common practice to ensure the ‘simultaneous
dosing’ of both calibrators and sam-ples onto a single-agar plate. In this manner, it is possible and feasible to
achieve the following three cardinal objectives :
·
thickness
of the agar-plate variants, critical edge-effects, and
·
incubation
temperature variants caused on account of irregular warming
inside the ‘incuba-tor’ must be
reduced to bare minimum by employing some sort of ‘predetermined random layout’.
(6) ‘Random Patterns’ for Application in
Microbiological Plate Assay : In usual practice, we frequently come across two prevalent types ‘random patterns’ for application in the microbiological plate assay, namely :
(a) Latin-Square Arrangement – in this
particular case the number of replicates
almost equals the number of
specimens (samples) ; and the ultimate result ensures the maximum preci-sion, as shown in Fig.
10.1(a).
(b) Less Acceptable (Demanding) Methods –
employing rather fewer replicates are invariably acceptable for two vital and important purposes, such
as :
·
clinical
assays, and
·
pharmacokinetic
studies,
as
illustrated in Figs. 10.1(b) and (c).
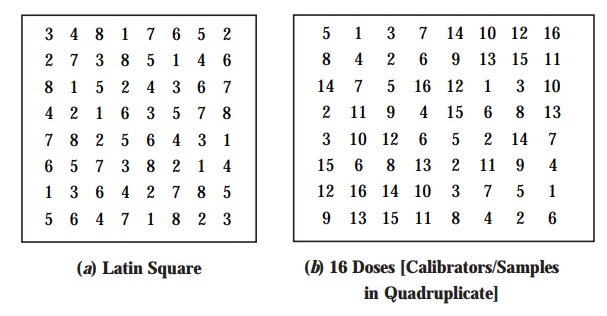
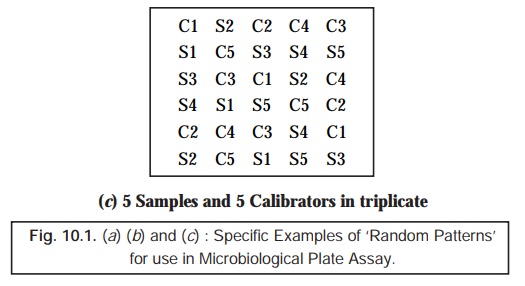
5. Measurement of Zone of Inhibition
To
measure the zone of inhibition with
an utmost precision and accuracy, the use of a Magnify-ing Zone Reader must be employed carefully. Besides, to
avoid and eliminate completely the subjective bias, the microbiologist taking the reading of
the incubated agar-plate must be totally unaware of the ground realities whether he is recording the final reading of
either a ‘treat zone’ or a ‘calibrator’. Therefore, the judicious
and skilful application of the ‘random’
arrangements as depicted in Fig. 10.2 may go a long way to help to ensure
critically the aforesaid zone of
inhibition. However, the ‘random
pattern’ duly installed could be
duly decephered after having taken the reading of the agar-plate.
6. Calibration
Calibration may be accomplished by means of two universally recognized and accepted methods, namely :
(a) Standard
Curves, and
(b) 2-By-2-Assay.
Each of
these two methods will now be discussed
briefly in the sections that follows :
(a) Standard Curves
While
plotting the standard curves one may
make use of at least two and even up to seven ‘calibrators’ covering entirely the required range of operational
concentrations. Besides, these selected concentrations
must be spaced equally on a ‘Logarithmic
Scale’ viz.,starting from 0.5, 1,
2, 4, 8, 16 and up to 32 mg. L– 1.
However,
the exact number of the ensuing replicates
of each calibrator must be the bare mini-mum absolutely necessary to
produce the desired precision
ultimately. It has been duly observed that a ‘manual plot’ of either :
·
zone size
Vs log10 concentration, or
·
[zone
size]2 Vs log10 concentration,
will give
rise to the formation of ‘near straight
line’, as depicted in Fig. 10.2.
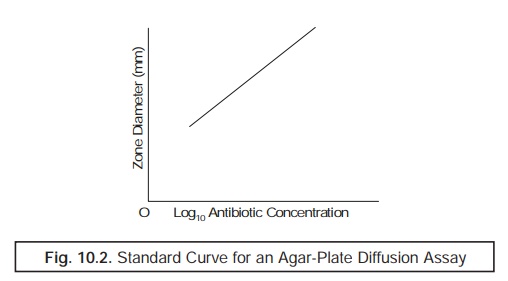
Note : A microcomputer may by readily installed and
programmed to derandomise the realistic and actual zone pattern by adopting
three steps in a sequetial manner viz.,
(a) consider the mean of the ‘zone
sizes’ ; (b) compute the standard
curve ; and (c) calcuate the ultimate
results for the tests ; and thereby enabling the ‘zone sizes’ to be read almost
directly from the incubated agar-plate right into the computer.
(b) 2-By-2-Assay
The 2-by-2-assay is particularly suitable
for estimating the exact and precise potency of a plethora of ‘Pharmaceutical Formulations’. In this
method a relatively high degree of precision is very much required, followed by
another two critical aspects may be
duly taken into consideration, such as :
·
Latin
square design with tests, and
·
Calibrators
at 2/3
levels of concentration.
Example : An 8 × 8 Latin square may be employed gainfully in two different ways :
First— to assay 3
samples + 1 calibrator, and
Second— to assay 2
samples + 2 calibrators,
invariably
at two distinct levels of
concentrations* each, and having a ‘coefficient
of variation’ at about 3%.
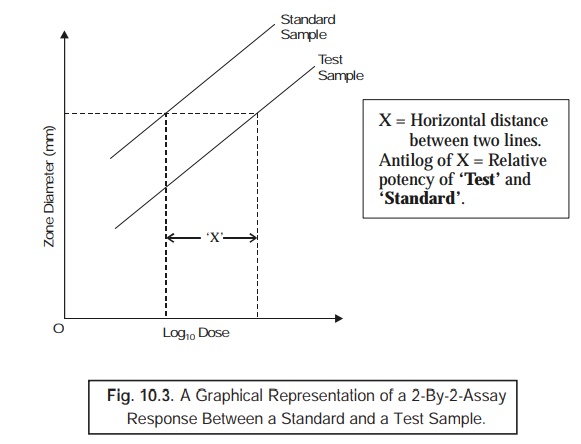
Evidently,
based on this technique, one may obtain easily and conveniently the ‘parallel dose– response lines’ strategically required for the calibrators vis-a-vis the tests performed at two distinct dilutions, as depicted in Fig.
10.3. Importantly, it is quite feasible and possible to establish the exact and precise potency of samples may be computed effectively or
estimated from meticulously derived nomograms.
2. Rapid-Reliable-Reproducible Microbial Assay Methods
It is
worthwhile to mention here that the usual ‘conventional
agar-plate assays’ not only re-quire stipulated incubation for several hours
but also are rather quite slow. Furthermore, reasonably judicious constant,
rigorous, and honest attempts do prevail for the development of ‘rapid-reliable-reproducible microbial
assay methods’ based on the exploitation of techniques that essentially meas-ure
definite cognizable variations in the pattern of growth-rate invariably after a short
incubation.
Nevertheless,
these so called ‘rapid methods’
generally suffer from the similar critical problems usually encountered in the ‘slow methods’ namely :
·
inadequate specificity, and
·
lack of precision.
In actual
practice there are two well-known
techniques that provide rapid-reliable-reproduc-ible
microbial assay methods, namely :
(a) Urease
Activity, and
(b) Luciferase
Assay.
These two aforesaid techniques shall now be
discussed briefly in the sections that follows :
(a) Urease Activity
Urease refers to an enzyme that specifically catalyzes the hydrolysis of urea to ammonia (NH3) and
carbon dioxide (CO2) ; it
is a nickel protein of microbes and plants which is critically employed in
carrying out the clinical assays of
plasma-urea concentration.
Importanlty,
the microorganism Proteius mirabilis grows significanlty in a urea-containing
culture medium, whereupon it particularly causes the hydrolysis of urea to ammonia, and thereby helps to raise the pH of the medium. However,
the actual production of urease is
reasonably inhibited by the so called ‘aminoglycoside
antibiotics’,* such as : amikacin,
gentamicin, kanamycin, neomycin, netilmicin,
tobramycin, doxorubicin, cephalosporins, cephamycius, thienamycin, lincomycin,
clindamycin, erythromycin, clarithromycin, azithromycin, oleandomycin,
spramycins, and the like.
Methodology : The various steps involved are as
follows :
(1) Assay
is performed with two series of tubes
of urea-containing culture medium
that have been duly incorporated with a range of calibrator solutions.
(2) First series of tubes in duly
added a certain volume of the sample which
is essentially equivalent to the
volume of the calibrator.
(3) Second series of tubes is duly
added exactly half the volume of the
sample.
(4) Both ‘set of tubes’ are subsequently
inoculated with P. mirabilis, and duly incubated for a duration of 60–70
minutes.
(5) pH of
the resulting solution is measured accurately upto 0.01 pH units.
(6) In
fact, it is possible to obtain two
distinct ‘calibration curves’ by
plotting pH Vs log10 i.e.,
the ensuing calibrator concentration for each of the two series.
(7) The ‘vertical distance’ existing between
the two curves is found to be almost
equal to the legarithm of 1/2 the concentration of ‘drug substance’ present in the sample.
Note : (1) In usual practice, it is rather
difficult to obtain ‘reliable’ results by adopting the ‘Urease Activity’
method.
(2) A standardized, senstitive, and reliable pH
Meter is an absolute must for this particular assay.
(b) Luciferase Assay
In the
specific ‘Luciferase Assay’, the
firefly luciferase** is made use of
for the actual meas-urement of small quantum of ATP*** duly present in a microbial culture, whereby the levels of ATP get proportionately
reduced by the ensuing action of the aminoglycoside
antibiotics (see Section 10.3.2.1).
Methodology : The various steps involved in the ‘Luciferase Assay’ are as enumerated
under sequentially :
(1) Both test solutions (i.e., after preliminary heating provided the matrix is serum) along
with calibrators are carefully added
into the various tubes of the culture medium specifically containing a growing microbial culture (i.e., organism).
(2) After
adequate incubation for a 90 minute duration the cultures are duly treated with
‘apyrase’ so as to ensure the
complete destruction of the
extracellular ATP.
(3) The
resulting solution is duly extracted with EDTA/sulphuric acid, and thus the intracellular ATP critically assayed with the
firefly enzyme using a
‘Luminometer’.
(4) Finally,
a ‘calibration curve’ is constructed
meticulously by plotting the two vital compo-nents, namely : (a) intracellular
ATP content, and (b) log10 i.e., the calibrator
concentration.
Note : As to date, the ‘Luciferase Assay’ has not
yet accomplished a wide application ; however, it may find its enormous usage
in the near future with the advent of such ‘luciferase formula-tions’ that
would turn out to be even much more active, reliable, and dependable.
Related Topics
