Types of protein and peptide therapeutics
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Protein and peptide drug delivery
Antibody is a protein produced by β-lymphocytes in response to substances recognized as foreign (antigens).
Types of protein
and peptide therapeutics
Antibodies
Antibody
is a protein produced by β-lymphocytes
in response to substances recognized as foreign (antigens). Antibodies recognize and bind to antigens, resulting in
their inactivation or opsonization (binding of antibody to the membrane surface
of invading pathogen, thus marking it for phagocytosis) or complement-mediated
destruction. Antibodies are also known as immu-noglobulins (abbreviated Ig)
because they are immune-response proteins that are globular proteins (compact with higher orders of structure and
hydrophilic surface making them soluble; as against fibrous proteins, which have predominantly secondary structure and
are insoluble). Of the five major types of antibodies (Table
25.3), IgG is preferred for therapeutic application due to its wide
distribution and function. Structurally, Ig is commonly represented in a
typical Y-arm structure (Figure 25.5)
consisting of two large/heavy and two small/light polypeptide chains joined by
disul-fide bridges. Antibody fragments consist of a constant region
(designated, Fc) and a variable, antigen-binding region (designated, Fab).
Antibodies that recognize multiple sites of an antigen are termed polyclonal, whereas anti-bodies that
target only a specific site are monoclonal.
Identical immune cells make monoclonal antibodies, whereas polyclonal antibodies
are produced by a mass of immune cells that may produce antibodies against
different regions of the antigen. In industrial application, monoclonal
antibodies are prepared by recombinant DNA technology in cell cultures. For
human clinical applications, generally monoclonal antibodies are pre-ferred.
Polyclonal antibodies are utilized for diagnostic and lab use such as
immunohistochemistry.
Table 25.3 Types of antibodies
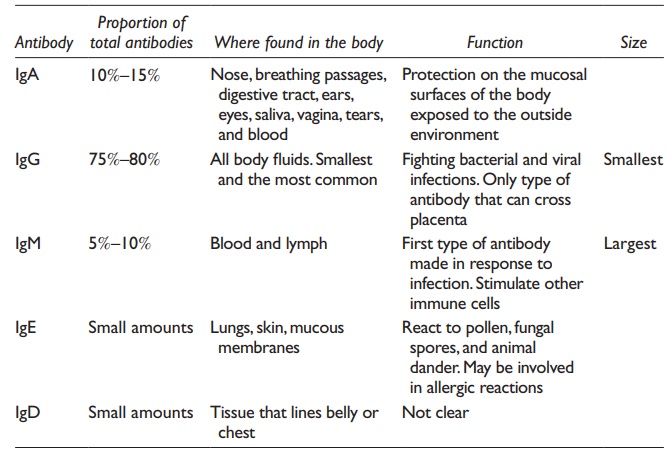
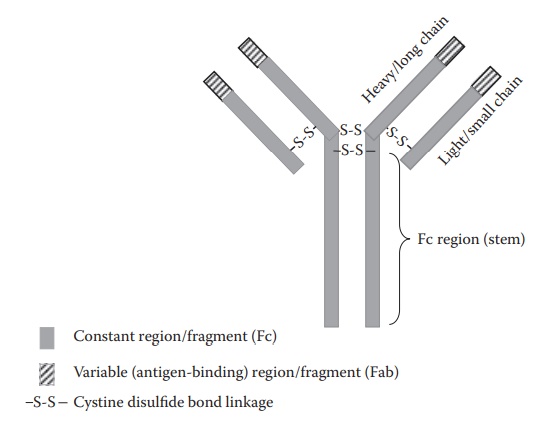
Figure 25.5 Typical structure of an
antibody.
A
number of immunoglobulin (Ig) G products have been developed for therapeutic
use in various immune disorders (Table 25.1).
Due to their specificity, there is a growing interest in the use of monoclonal
anti-bodies and their modifications as therapeutics. For example, antibodies
whose Fab fragment segments have been reduced in size to the small-est known
antigen-binding fragments are known as domain
antibodies. Also, antibodies that can bind two different antigens are
called bispecific antibodies.
The
usefulness of antibodies was limited by the immune response gener-ated by the
host to the administered antibodies, especially when the anti-bodies were
generated by antigen injection in foreign animal species, such as mouse. The antibodies generated in mouse were named
with the suffix ~momab. The use
of humanized/human monoclonal antibodies with the use of recombinant DNA
technology has helped to overcome these limitations.
·
Chimeric and
humanized antibodies are the antibodies produced from nonhuman species whose protein sequences have been modified
to increase their similarity to the antibody variants that are naturally found
in humans.
·
Chimeric antibodies consist of murine
variable regions fused with human
constant regions, resulting in ~65% human amino acid sequence. This reduces
immunogenicity and increases plasma half-life. These antibodies are named with the suffix ~ximab. For example, rituximab is a chimeric antibody.
·
Humanized antibodies
are
made by grafting the murine variable amino
acid domains (which determine antigen specificity) onto human antibodies,
resulting in ~95% human amino acid sequence. These, however, have lower
antigen-binding affinity than murine antibodies. These antibodies are named with the suffix ~zumab. For example, bevacizumab (Avastin®) is a humanized antibody
that targets vascular endothelial growth factor (VEGF) and is rec-ommended as
first-line therapy in advanced colorectal cancer in combination with other
drugs.
·
Human monoclonal
antibodies can
be produced using phage display or
transgenic mice. Transferring the human Ig genes into the mouse genome can
produce these antibodies. These
antibod-ies are named with the suffix ~mumab.
For example, ipilimumab is a human
mAb that inhibits the checkpoint receptor cytotoxic T lymphocyte-associated
antigen 4 (CTLA4) and is recommended for advanced-stage melanoma.
Most
therapeutic antibodies exert their therapeutic effects by binding to selected
cellular targets, which are then destroyed by physiological mech-anisms
activated by the effector functions of the antibody. In addition, antibodies
can also be used as drug delivery and targeting vehicles. Active research and
development is being pursued on customized antibodies conju-gated to toxins,
radioisotopes, small drugs, enzymes, and genes for selectively destroying
harmful cells in the body. For example, several ADCs have been developed for
the treatment of cancer that utilizes a toxin, which is a small molecule
attached to an antibody. For example, Adcetris® and Kadcyla® are ADCs for tumor
treatment.
Hormones and physiological proteins
Protein
therapeutics to replace or supplement endogenous protein mol-ecules are used
for several diseases such as diabetes (insulin), growth hormone deficiency
(growth hormone), and hemophilia (factors VIII and IX). Table 25.1 lists some protein therapeutics and their clinical
applications.
Chemically modified proteins and peptides
Chemical
modifications of proteins are carried out to either
·
Increase target specificity, for example, abatacept (Table 25.1) and conjugation to sugars.
·
Increase therapeutic ability, for example, radiolabeled
antibodies and ADCs (Table 25.1).
·
Increase plasma half-life, for example, by PEGylation of
antibodies.
Conjugation with sugars
Conjugation
of sugars, such as sucrose, mannose (mannosylation), or lactose
(lactosylation), to proteins can be used to provide targeted deliv-ery of
proteins. For example, receptors for carbohydrates, such as the
asialoglycoprotein receptor on hepatocytes, and the mannose receptor on
macrophages, such as Kupffer cells, recognize corresponding sugars.
Mannosylated bovine serum albumin (Man-BSA) and galactosylated BSA (Gal-BSA)
preferentially bind to alveolar macrophages and hepatocytes, respectively.
Galactosylated and mannosylated recombinant human super-oxide dismutase
(Gal-SOD, Man-SOD) exhibited inhibitory effects supe-rior to native SOD against
hepatic ischemia-perfusion injury.
PEGylation
Proteins
may be conjugated to Polyethylene glycol (PEG), a nonimmunogenic, nontoxic, and
FDA-approved polymer, to increase their plasma half-life. The process of
conjugation with PEG is called PEGylation,
and the protein after the conjugation is called the PEGylated protein. PEG consists of a flexible polyether chain that
provides a hydrophilic surface, thus shielding hydropho-bic groups and
minimizing nonspecific interactions. Attachment of PEG on protein surface also
increases the hydrodynamic diameter of proteins. Either straight chain or
branched PEG can be used for PEGylation. The flexibility of the side chain
allows the PEGylated protein to interact with the target.
PEGylation
can increase biocompatibility, reduce immune response, increase in vivo stability, delay clearance by
the reticuloendothelial system, and prevent protein adsorption on the surface
of the delivery device, such as syringe.
Applications
Interferon
(IFN)-2α has a low plasma
half-life and needs daily injections. However, IFN-2α conjugated to branched PEG 40 (i.e., PEG of 40 kDa average molecular weight)
provides sustained plasma concentrations upon once a week injection. Other
examples of PEG-modification to modulate clearance rate of proteins include
PEG-adenosine deaminase (PEG-ADA), PEG-asparaginase, PEG-rIL2, and
PEG-interferon. Native ADA is not effective due to its short half-life (<30
min) and is immunogenic due to bovine source, whereas PEGylated ADA (Adagen®)
is quite effective, has long half-life, and is nonimmunogenic.
Chemistry
PEG
has two hydroxyl groups at each end of the linear chain. PEGylation is often
done by creating a reactive electrophilic intermediate with succinimide (thus
producing N-hydroxysuccinimide, NHS),
which undergoes electrophilic
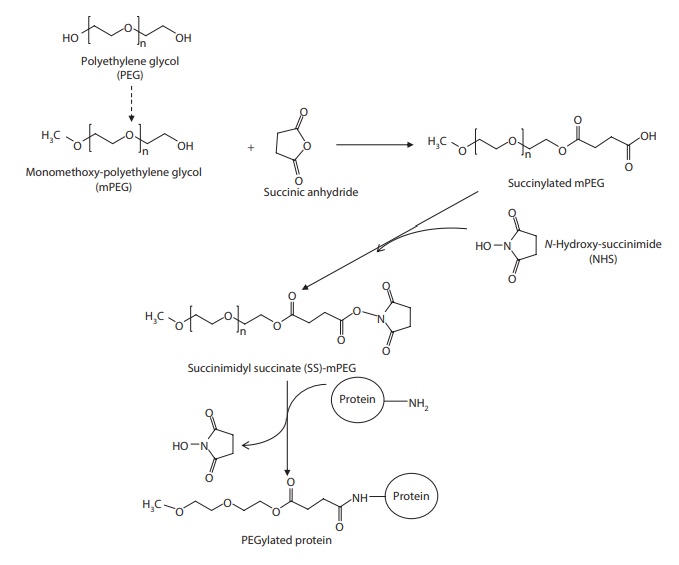
Figure 25.6 PEGylation of proteins using
N-hydroxysuccinimide (NHS) derivative of methoxy PEG.
The NHS ester groups primarily react with the α-amines at the N-terminals and the ε–amines of lysine side chains. Two
hydroxyl groups—one at either end—make the natural PEG bifunctional. To prevent
the potential for cross-linking and polymerization with the natural
bifunctional polymer, monofunctional PEG polymer can be used. To make PEG
monofunctional, one end of the chain is blocked with a methyl ether (methoxy)
group. Such a monofunctional PEG is termed monomethoxyPEG (mPEG). Thus, mPEG
contains only one hydroxyl group per chain, thus limiting activation and
coupling to one site.
Limitations
PEGylation
usually reduces binding affinity of the protein to its target. PEGylation also
increases the viscosity of protein formulations, which may limit the
development of concentrated solutions for injection. Protein reac-tion with PEG
generally has low efficiency and is difficult to optimize. In addition, PEG
often contains peroxide impurities, which can lead to oxida-tive protein
degradation during shelf life storage.
Other protein conjugation approaches
Proteins
can also be conjugated to hydroxyethyl starch (HESylation) or to polysialic
acid (PSAylation) using similar chemistry to increase their plasma half-life.
PEGylation remains the most common protein modification.
Antibody drug conjugates
In
recent years, several mAb-based therapeutics that have a small molecule
conjugated to the antibody—the ADCs—have been commercialized, such as Kadcyla®
and Adcentris®. Most current ADCs are developed for oncology indications and
utilize a high potency cytotoxic drug called payload attached through a
covalent linker to a monoclonal antibody that serves as a targeting moiety. The
discovery and development of ADCs follow unique paradigms that overlap both
small and large molecule drug discovery and development but have unique
distinctions. For example, the attachment of hydrophobic drug on the mAb
changes mAb surface properties and conformational stabil-ity. It can increase
protein aggregation and surface hydrophobicity.
Chemistry
of conjugation of small molecule drug to the antibody is con-stantly evolving.
In general, the conjugation can be random (through, e.g., lysine or cysteine
residues) or site specific (through, e.g., engineered antibod-ies that have
specific amino acid residues). One needs to pay attention to the selection of
mAb, payload, and linker for an effective ADC. Currently, several ADCs are in
clinical trials as monotherapies or in combination with other anticancer drugs.
Related Topics
