Enzymes
| Home | | Pharmacology |Chapter: Essential pharmacology : Pharmacodynamics Mechanism Of Drug Action; Receptor Pharmacology
Almost all biological reactions are carried out under catalytic influence of enzymes; hence, enzymes are a very important target of drug action. Drugs can either increase or decrease the rate of enzymatically mediated reactions. However, in physiological systems enzyme activities are often optimally set.
ENZYMES
Almost all biological
reactions are carried out under catalytic influence of enzymes; hence, enzymes
are a very important target of drug action. Drugs can either increase or
decrease the rate of enzymatically mediated reactions. However, in
physiological systems enzyme activities are often optimally set. Thus, stimulation
of enzymes by drugs, that are truly foreign substances, is unusual. Enzyme
stimulation is relevant to some natural metabolites only, e.g. pyridoxine acts
as a cofactor and increases decarboxylase activity. Several enzymes are
stimulated through receptors and second messengers, e.g. adrenaline stimulates
hepatic glycogen phosphorylase through β receptors and cyclic
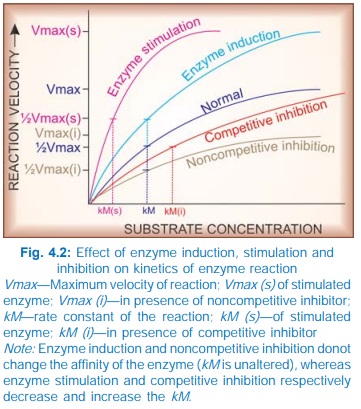
AMP. Stimulation of an enzyme increases its affinity for the substrate so that
rate constant (kM) of the reaction is
lowered (Fig. 4.2).
Apparent increase in
enzyme activity can also occur by enzyme
induction, i.e. synthesis of more enzyme protein. This cannot be called
stimulation because the kM does not
change. Many drugs induce microsomal enzymes.
Inhibition
of enzymes is a common mode of drug action.
A.
Nonspecific inhibition
Many
chemicals and drugs are capable
of denaturing proteins. They alter the tertiary structure of any enzyme with
which they come in contact and thus inhibit it. Heavy metal salts, strong acids
and alkalies, alcohol, formaldehyde, phenol inhibit enzymes nonspecifically.
Such inhibitors are too damaging to be used systemically.
B.
Specific inhibition
Many
drugs inhibit a particular enzyme
without affecting others. Such inhibition is either competitive or noncompetitive.
i) Competitive (equilibrium type) The drug being structurally similar competes with the
normal substrate for the catalytic binding site of the enzyme so that the
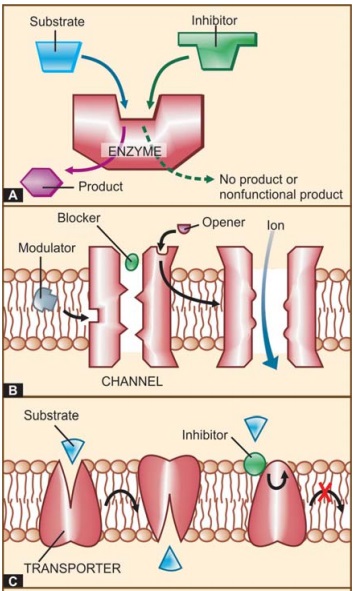
product is not formed or a nonfunctional product is formed (Fig. 4.1A), and a
new equilibrium is achieved in the presence of the drug. Such inhibitors
increase the kM but the Vmax remains unchanged (Fig. 4.2), i.e.
higher concentration of the substrate
is required to achieve ½ maximal reaction velocity, but if substrate
concentration is sufficiently increased, it can displace the inhibitor and the
same maximal reaction velocity can be attained.
§ Physostigmine and
neostigmine compete with acetylcholine for cholinesterase.
§ Sulfonamides compete
with PABA for bacterial folate synthetase.
§ Moclobemide competes
with catecholamines for monoamine oxidaseA (MAOA).
§ Captopril competes
with angiotensin 1 for angiotensin converting enzyme (ACE).
§ Finasteride competes
with testosterone for 5αreductase
§ Letrozole competes
with androstenedione and testosterone for the aromatase enzyme.
§ Allopurinol competes
with hypoxanthine for xanthine oxidase; is itself oxidized to alloxanthine (a
non competitive inhibitor).
§ Carbidopa and
methyldopa compete with levodopa for dopa decarboxylase.
A nonequilibrium type of enzyme inhibition
can also occur with drugs which react with the same catalytic site of the
enzyme but either form strong covalent bonds or have such high affinity for the
enzyme that the normal substrate is not able to displace the inhibitor, e.g.
§ Organophosphates react
covalently with the esteretic site of the enzyme cholinesterase.
§ Methotrexate has
50,000 times higher affinity for dihydrofolate reductase than the normal
substrate DHFA.
In these situations, kM is increased and Vmax is reduced.
ii) Noncompetitive The inhibitor reacts with an adjacent site and not with the catalytic
site, but alters the enzyme in such a way that it loses its catalytic property.
Thus, kM is unchanged but Vmax is reduced. Examples are given in
the box.
N
Acetazolamide — Carbonic anhydrase
Aspirin, indomethacin — Cyclooxygenase
Disulfiram — Aldehyde dehydrogenase
Omeprazole — H+ K+ ATPase
Digoxin — Na+ K+ ATPase
Theophylline — Phosphodiesterase
Propylthiouracil — Peroxidase in thyroid
Lovastatin — HMGCoA reductase
Sildenafil — Phosphodiesterase5
