Similarities Between Terodiline and Prenylamine
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: Withdrawal of Terodiline: A Tale of Two Toxicities
In the context of the ICH E2E guideline on Pharma-covigilance Planning, some vital pieces of information that might have presaged the potential proarrhythmic risk from terodiline were already known at the time of its re-development.
SIMILARITIES BETWEEN TERODILINE
AND PRENYLAMINE
In
the context of the ICH E2E guideline on Pharma-covigilance Planning, some vital
pieces of information that might have presaged the potential proarrhythmic risk
from terodiline were already known at the time of its re-development. The
analogy between terodiline and prenylamine goes well beyond their therapeu-tic
class, and extends into their chemical structures and stereoselective
pharmacological and toxicological profiles (Table 10.1).
First,
it was well known that the use of antianginal drugs (prenylamine and
lidoflazine) might be asso-ciated with QT interval prolongation and torsade de
pointes. Prenylamine was introduced in the United Kingdom in the early 1960s
and lidoflazine in 1979. Secondly, both prenylamine and terodiline are highly
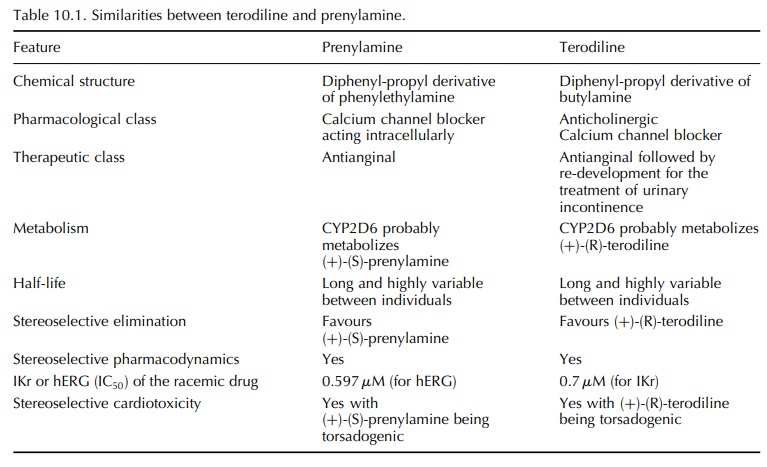
related in their chemical structures. While terodiline is a diphenyl-propyl
derivative of buty-lamine (Figure 10.1), prenylamine is a diphenyl-propyl
derivative of phenylethylamine (Figure 10.2).
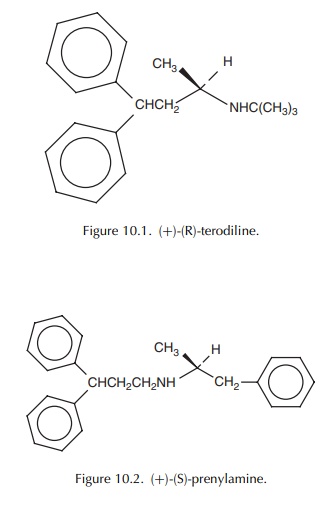
The
presence of a chiral centre in each drug gives rise to a pair of enantiomers.
It is acknowledged that even a minor modification in the structure of a
molecule can dramatically alter the activity of a drug, and indeed this is the
basis of metabolic inactivation of most drugs. However, notwithstanding the
minor structural differences between terodiline and preny-lamine, it is
intuitive that terodiline must have some cardiac effects since it was marketed
originally as a cardioactive antianginal agent. Not surprisingly, both drugs
share a very similar complex pharmacological profile that is discussed later.
Thirdly, both preny-lamine and terodiline are chirally active and there was
already evidence of stereoselectivity in the proarrhyth-mic potential of
prenylamine. Fourthly, there was wide inter-individual variability in the
metabolism of terodiline, with aberrant pharmacokinetic behaviour of one of the
enantiomers. This is also a feature of the pharmacokinetics of prenylamine.
Finally, there was evidence of stereoselectivity in the pharmaco-dynamic
activities of the two enantiomers of terodi-line, and therefore the
unexpectedly high frequency of anticholinergic effect observed during its use
as an antianginal agent should have already suggested an unusual behaviour of
one of the enantiomers (the enantiomer with predominantly anticholinergic
activity).
To
illustrate the regulatory deliberations at the time, frequent references will
be made to prenylamine in the commentary that follows. This will highlight in
detail the striking similarity between these two drugs, and hence the logic
that should have supported the re-development of terodiline. Importantly, this
comparison emphasizes the strengths of both a scien-tific synthesis of all the
available information when evaluating the significance of even a handful of
spontaneous reports of a unique drug reaction, and of formulating the most
appropriate regulatory strategies for risk management.
Related Topics
