Structure Activity Relationship
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Adrenergic Blockers
Propranolol has become one of the most thoroughly studied and widely used drug in the therapeutic armamentarium; it is the standard against which all other β antagonists are compared.
STRUCTURE-ACTIVITY RELATIONSHIP Propranolol has become one of the most thoroughly studied and widely used drug in the therapeutic armamentarium; it is the standard against which all other β antagonists are compared. The aromatic ring and its substituent is the primary determinant of β1 antagonistic activity. The aryl group also affects the absorption, excretion, and metabolism of the β blockers. β blockers are structurally similar to β agonist. The catechol ring can be replaced by a variety of ring system without loss of antagonistic activity. Replacement of catechol hydroxyl group with chlorine of phenyl ring system retains β blocking activity. Example: pronethalol, dichloroisoproterenol. N, N–disubstitution decreases the β blocking activity, and the activity is maintained when the phenyl ethyl, hydroxy phenyl ethyl, or methoxy phenyl ethyl groups are added to amine as a part of the molecule. The two carbon chains are essential for activity. The introduction of –OCH2 group into the molecule between the aromatic ring and the ethyl amine side chain provides β blocking agents, for example, propranolol. As in the sympathomimetics, bulky aliphatic groups, such as the tert-butyl and isopropyl groups are normally found on the amino function of the aryloxypropanolamine β receptor antagonists. It must be a secondary amine for optimal activity. As with the sympathomimetic agents, the configuration of the hydroxyl bearing carbon of the aryloxypropanolamine side chain play a critical role in the interaction of β antagonist drugs with β receptor. The carbon must possess the (S) configuration for optimal affinity to the β receptor. The enantiomer with the (R) configuration is typically 100 times less potent.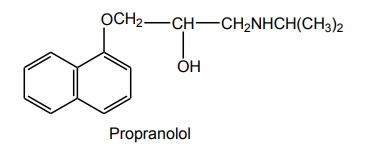
Related Topics
