Time Sequence Analysis
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: Spontaneous Reporting - France
The criteria include challenge, dechallenge and rechallenge.
TIME SEQUENCE ANALYSIS
The
criteria include challenge, dechallenge and rechallenge.
·
Challenge can be classified
into ‘very suggestive’ (when there is
an obvious temporal association between drug administration and the onset of
the reaction, such as anaphylaxis during intravenous drug injection),
impossible (when the drug is given after event onset), and compatible (other
cases). The ‘impossible’ category is especially pertinent, since it justifies
knowing the reason for which the drug was given to eliminate protopathic bias,
the prescribing of a drug for early symptoms of the event later reported as a
reaction (e.g. agran-ulocytosis attributed to an antibiotic that was prescribed
for the sore throat and fever that are the first signs of agranulocytosis, or
stomach cancer and H2 antagonists prescribed for undiagnosed dyspepsia).
·
Dechallenge can be suggestive
when the reaction abates when the
drug is stopped. It can be non-conclusive when there is no assessable
dechallenge (e.g. drug not stopped, or patient dies), or there is no
information on dechallenge, or the reaction is irreversible (renal failure,
death), or specific treat-ment was applied to the reaction, and so on. It is
against the role of the drug if the reaction persists (if reversible) when the
drug is stopped, within pharmacokinetic constraints.
·
Rechallenge is positive when the
reaction recurs when the patient
takes the drug again (for whatever reason, bearing in mind recurrent
protopathic bias), negative when the reaction does not recur when the drug is
taken again at the same dose, for the same duration, with the same concomitant
diseases and medication (a rare event), and not assessable in all other cases.
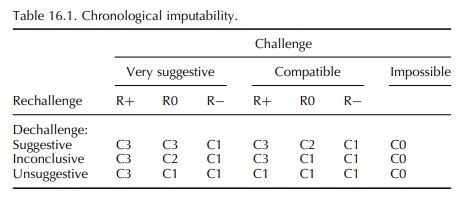
Information
on challenge, dechallenge and rechal-lenge is input into the appropriate
three-way table, which results in a grade from CO (drug excluded) to C3 (very suggestive
time association or positive rechallenge) (Table 16.1).
Related Topics
