Guar Gum
| Home | | Pharmacognosy |Chapter: Pharmacognosy and Phytochemistry : Drugs Containing Carbohydrates and Derived Products
Guar gum is a seed gum produced from the powdered endosperm of the seeds of Cyamopsis tetragonolobus Linn belonging to family Leguminosae.
GUAR GUM
Synonyms
Guar gum, Jaguar gum, Guar flour and Decorpa.
Biological Source
Guar gum is a seed gum produced from the powdered endosperm
of the seeds of Cyamopsis tetragonolobus
Linn belonging to family Leguminosae.
Geographical Source
Guar or cluster bean is a drought-tolerant annual legume
that was introduced into the United States from India in 1903. Commercial
production of guar in the United States began in the early 1950s and has been
concentrated in northern Texas and south-western Oklahoma. The major world
suppliers are India, Pakistan and the United States, Australia and Africa.
Rajasthan in western India is the major guar-producing state, accounting for
70% of the production. Guar is also grown in Gujarat, Haryana, Punjab and in
some parts of Uttar Pradesh and Madhya Pradesh. India grows over 850,000 tons,
or 80% of the total guar produced all over the world. 75% of the guar gum or
derivatives produced in India are exported, mainly to the United States and to
European countries.
Cultivation, Collection and Preparation
The plant of gaur gum is draught resistance and quite hardy
in its constitutions. It is generally shown in May– June and harvested in
September–October. At the stage of full maturity, the plant yields 600–800 lb
of seeds per acre under un-irrigated conditions but the production nearly
doubles under irrigated conditions.
First of all the fully developed white seeds of guar gum are
collected and freed from any foreign substances. The sorted seeds are fed to a
mechanical ‘splitter’ to obtain the bifurcated guar seeds which are then
separated into husk and the respective cotyledons having the ‘embryo’. The gum
is found into the endosperm. Generally, the guar seeds comprise the endosperm
35–40%, germ (or embryo) 45–50% and husk 14–17%.
The cotyledons, having a distinct bitter taste are separated
from the endosperm by the process called ‘winnowing’. The crude guar gum, that
is, the endosperms is subsequently pulverized by means a ‘micro-pulverizer’
followed by grinding. The relatively softer cotyledons sticking to the
endosperms are separated by mechanical ‘sifting’ process. Thus, the crude guar
gum is converted to a purified form (i.e. devoid of cotyledons), which is then
repeatedly pulver-ized and shifted for several hours till a final white powder
or granular product is obtained.
Morphology

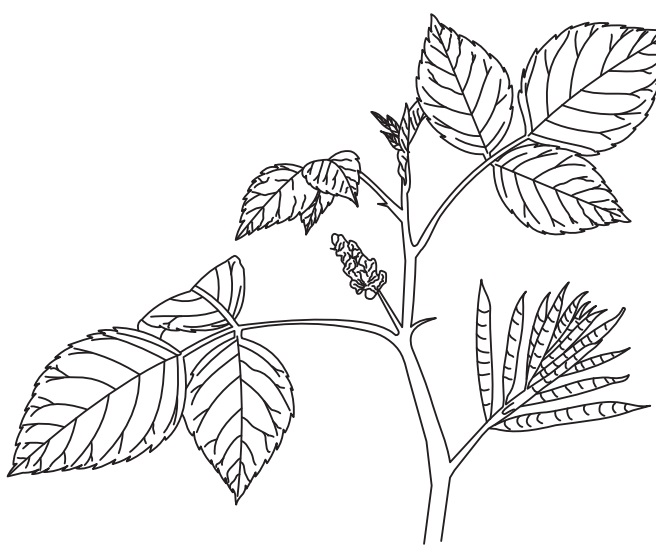
History
Guar gum is a dietary fibre obtained from the endosperm of
the Indian cluster bean. The endosperm can account for more than 40% of the
seed weight and is separated and ground to form commercial guar gum.
Guar gum has been used for centuries as a thickening agent
for foods and pharmaceuticals. It continues to find extensive use for these
applications and also is used by the paper, textile and oil-drilling
industries.
Chemical Constituents
The water-soluble part of guar gum contains mainly of a high
molecular weight hydrocolloidal polysaccharide, that is, galactomannan, which
is commonly known as guaran. Guaran consists of linear chains of (1→4)—β—D— mannopyranosyl units with α—D—galactopyranosyl units attached by (1→6) linkages. However, the ratio of D— galactose to D—mannose
is 1: 2. The gum also contains about 5–7% of proteins.
Chemical Tests
1. On being treated with iodine
solution (0.1 N), it fails to give olive-green colouration.
2. It does not produce pink colour when
treated with Ruthenium Red solution (distinction from sterculia gum and agar).
3. A 2% solution of lead acetate gives
an instant white precipitate with guar gum (distinction from sterculia gum and
acacia).
4. A solution of guar gum (0.25 g in 10
ml of water) when mixed with 0.5 ml of benzidine (1% in ethanol) and 0.5 ml of
hydrogen peroxide produces no blue colouration (distinction from gum acacia).
5. Aqueous solution of guar gum is
converted to a gel by addition of a small amount of borax.
Uses
Guar gum is used as a protective colloid, a binding and
disintegrating agent, emulsifying agent, bulk laxative, appe-tite depressant
and in peptic ulcer therapy. Industrially, it is used in paper manufacturing,
printing, polishing, textiles and also in food and cosmetic industries. Guar
gum is extensively used as flocculent in ore-dressing and treat-ment of water.
Guar gum has been shown to decrease serum total cho-lesterol
levels by about 10–15% and low-density lipoprotein cholesterol (LDL-cholesterol)
by up to 25% without any significant effect on triglycerides or high-density
lipoprotein cholesterol (HDL-cholesterol) levels.
The ability of guar to affect gastrointestinal transit may
contribute to its hypoglycemic activity. Guar reduces postprandial glucose and
insulin levels in both healthy and diabetic subjects and may be a useful
adjunct in the treatment of noninsulin-dependent diabetes.
Guar gum remains important ingredient in over-the-counter
weight loss preparations. Even in the absence of weight loss, guar
supplementation for 2 weeks reduced blood pressure by 9% in moderately
overweight men.
Toxicology
In the colon, guar gum is fermented to short-chain fatty
acids. Both guar and its resultant by-products do not appear to be absorbed by
the gut. The most common adverse effects, therefore, are gastrointestinal,
including gastrointestinal pain, nausea, diarrhoea and flatulence.
Approximately half of those taking guar experience flatulence; this usually
occurs early in treatment and resolves with continued use. Starting with doses
of about 3 g three times a day, not to exceed 15 g per day, can minimize
gastrointestinal effects.
Guar gum may affect the absorption of concomitantly
administered drugs. Bezafibrate, acetaminophen (e.g. Tylenol), digoxin (e.g.
Lanoxin), glipizide (e.g. Glucotrol) or glyburide (e.g. DiaBeta, Micronase) are
generally unaf-fected by concomitant administration. The ingestion of more than
30 g of guar per day by diabetic patients did not adversely affect mineral balances
after six months. Guar gum in a weight-loss product has been implicated in
esophageal obstruction in a patient who exceeded the recommended dosage. In a
recent review, 18 cases of esophageal obstruc-tion, seven cases of small bowel
obstruction, and possibly one death were associated with the use of Cal-Ban
3000, a guar gum containing diet pill. The water-retaining capacity of the gum
permits it to swell to 10- to 20-fold and may lead to luminal obstruction,
particularly when an anatomic predisposition exists. Guar always should be
taken with large amounts of liquid. Occupational asthma has been observed among
those working with guar gum. Because of its potential to affect glycemic
control, guar gum should be used cautiously by diabetic patients.
Marketed Product
Ascenta Omega Smooth
Orange Sensation by Ascenta Health Ltd.
