Pharmacoepidemiology of Hormone Therapy: An Evolving Picture
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: Pharmacoepidemiology of Hormone Therapy: An Evolving Picture
When the FDA first approved menopausal hormone replacement therapy (Emmenin) in 1933, the formulation was not easy to mass-produce.
Pharmacoepidemiology
of Hormone Therapy: An Evolving Picture
When
the FDA first approved menopausal hormone replacement therapy (Emmenin) in
1933, the formulation was not easy to mass-produce (Rothenberg, 2005). In
1942, Premarin, a more easily manufac-tured estrogen, was introduced to the US
mass market (CDER, 1997). At that time, Premarin was known to contain two main
oestrogens, estrone and equilin, and some additional oestrogens in smaller
amounts. It was approved primarily for the short-term treatment of
postmenopausal symptoms – for relief of vasomo-tor symptoms associated with
menopause. According to the FDA website, the drug’s approval in 1942 predated
the current requirements for such comprehensive analysis of products under
review for marketing approval. At that time, Premarin’s approval was based on
acceptable chemistry, manufacturing, and controls information and a showing,
from reports of clinical investiga-tions, that the drug was safe for its
intended use in the treatment of menopausal symptoms and related conditions
(CDER, 2005).
Over
the years, however, many US women began to use the product off label for
prevention of certain diseases such as coronary heart disease (CHD) or
osteoporosis and for much longer periods of times than first envisioned. By the
mid-1960s, about 12% of all postmenopausal women were taking oestro-gen
(Harvard, 2003). Eventually, in 1986, the FDA approved the drug for prevention
of osteoporosis. By the time WHI was initiated in 1993, progestins had been
added to protect the endometrium and most postmenopausal women in the US had been
prescribed hormone therapy (HT). In addition, most, if not all of these women,
had used HT in a way not orig-inally approved by the FDA. A study by Hersh,
Stefanick and Stafford (2004) estimated that from 1999 to June 2002,
approximately 15 million post-menopausal women per year were taking HT. This
figure was up substantially from earlier years primar-ily due to an increase in
oral oestrogen/progestin combinations. It is of interest to note that more than
one-third of patients who were prescribed HT were older than 60 years and thus
several years post menopause.
There
is a long history of observational studies of HT, starting shortly after the
drug’s approval to treat menopausal symptoms in 1942. Epidemiological stud-ies,
animal studies and studies of surrogate biomarkers of CHD, as well as clinical
studies such as angiogra-phy and bone mineral density studies, all pointed in
the direction that HT was beneficial for the prevention of CHD and
osteoporosis. Some of the larger, and more influential, observational HT
studies, such as the Million Women Study (MWS) and Nurses’ Health Study (NHS),
are listed and described in Table 46.1 (Banks et al., 2004; Beral, 2003; Beral, Bull and Reeves, 2005; Grodstein,
Manson and Stampfer, 2001). In general, these observational studies, as well as
several clinical trials, showed the efficacy of oestrogen, and later a
combination of oestrogen and progestin, for treating menopausal symptoms (NIH,
2005; Rothenberg, 2005). Most observational studies also indicated that HT
conferred a protective effect on CHD and was associated with a decreased risk
of fractures, higher bone mineral density (BMD) and a lower colorectal cancer
risk than women of the same age and menopausal status who were not receiving
therapy (Banks et al., 2004; Beral,
2003; Grodstein et al., 1999;
Komulainen et al., 1999; Lufkin et al., 1992; Speroff et al.,
1996).

In
addition to demonstrating benefits of HT, early studies demonstrated several
risks. The main risk was an increased risk of venous thromboembolic events
(VTEs), or blood clots, in women on HT, a finding that has been borne out over
time (BCDSP, 1974; Grady et al.,
2000; Hulley, 2002; Nachtigall et al.,
1979; PEPI, 1995; Petitti et al.,
1979; WHI, 2002, 2004). Results of observational research studies were less
conclusive about HT’s risks or benefits related to CHD, breast cancer and
uterine cancer, though the results trended towards showing a benefit of HT on
CHD risk and an increased risk of breast and uterine cancer (Beral, 2003;
Grodstein, Manson and Stampfer, 2001; Lerner, 1986; Speroff et al., 1996; Wilson et al., 1985). The addition of
progestins to the HT formula-tions in the mid-1980s was thought to be
protective of the uterus, and some studies, though not all, demon-strated a
decrease in the risk of uterine cancer in those women with a uterus who were
taking combination oestrogen progestin therapy (Beral, Bull and Reeves, 2005)
(Figure 46.1)
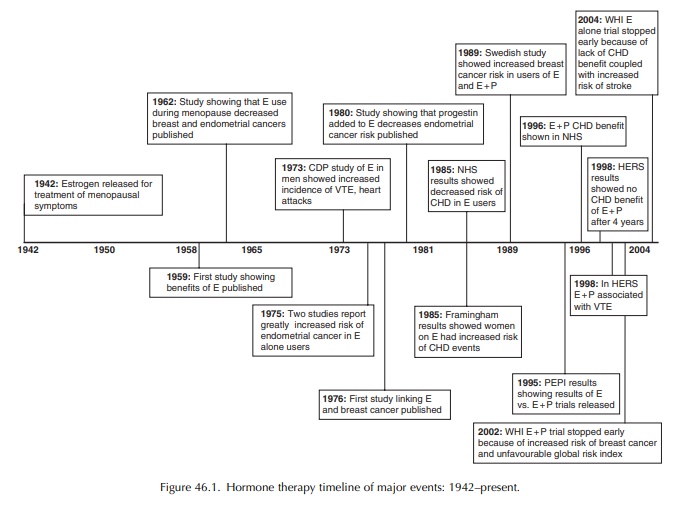
Though
these observational studies were helpful for evaluating some of the risks and
benefits of HT, they had known biases that could impact the observed benefits
and risks of HT (Grodstein, Clarkson and Manson, 2003). In general, women who
are prescribed oestrogen are leaner, less likely to smoke, less likely to eat a
high fat or high salt diet, more physically active, more highly educated, and
more likely to visit physi-cians regularly and take part in recommended
screen-ings than women who are not prescribed hormones (WHI, 1998). These
factors in and of themselves tend to decrease the risk of both CHD and some
cancers. Also, though not well documented, women taking HT and the physicians
who prescribe it may be less likely to attribute ischaemic symptoms to CHD if
they believe a priori that HT reduces
CHD risk (Col and Pauker, 2003). This makes it difficult to draw conclu-sions
about the effects of HT separate from the effects of these socio-behavioural
factors.
In
addition to the previously mentioned biases that make interpreting the results
of observational trials of hormone replacement difficult, additional
interpretability issues arise due to major differences between the studies. The
age ranges of individuals in the various observational studies mentioned
previ-ously and their racial and socioeconomic makeup differed greatly from
study to study. Major differ-ences in these factors make comparing results
(both within and across different observational studies) difficult, especially
since these factors have been shown to be related to CHD development. In
addition to these population differences, observational studies of HT differed
in how they defined CHD and other endpoints. For example, some studies, such as
the Framingham Heart Health Study, that examined the relationship between HT
and CHD included angina as part of the constellation of conditions that
comprise CHD while others, such as the Nurses Health Study, did not. This makes
comparison of the results of these studies difficult at best.
In part because of these difficulties in interpreting data from observational studies (given the potential biases mentioned previously), a number of random-ized controlled clinical trials (RCTs) of HT were initiated in an attempt to definitively answer ques-tions about the risks and benefits of HT (Thera-peutic Letter, 1999). These studies were conducted first in highly selected populations with trials such as the Coronary Drug Project (CDP) and the Heart and Estrogen/Progestin Replacement Study (HERS), and then in the more general population, with stud-ies such as the Postmenopausal Estrogen/Progestin Interventions (PEPI) and the much larger, and more comprehensive, Women’s Health Initiative (WHI). The first RCT relating to this question – the HERS trial – was published in 1998 (Hulley et al., 1998; Petitti, 1998). Several additional small, short-term RCTs were also conducted. One of these trials, The Coronary Drug Project, was actually conducted in men (The Coronary Drug Project Research Group, 1973), though the others were conducted in women (Hall et al., 1998; Komulainen et al., 1999; Lufkin et al., 1992; Speroff et al., 1996). These trials sought more conclusive evidence of the relationship between HT and CHD, uterine cancer, or other endpoints and all seem to corroborate evidence from the earliest published RCTs on HT. Table 46.1 summarizes the study populations and results from some of these randomized clinical trials as well as several of the larger, more influential observational studies.
It
was clear from the findings from these small, short-term trials that a larger,
longer-term random-ized clinical trial with a greater diversity of
post-menopausal women and ‘hard’ disease endpoints needed to be undertaken to
definitively answer these questions. And so, the timing was right for the
Women’s Health Initiative, which was made possible because of the efforts of
the NIH’s first female director, cardiologist Bernadine Healy, the Women’s
Health Caucus, and other groups who successfully lobbied for a line item in
congress’ budget to try to erase a 25-year gender gap in our knowledge of
diseases which affect women in their later years. In addition, the FDA would
not approve a statement in the HT label claiming a benefit for heart disease
until a definitive clinical trial was conducted. This was the state of the
field when the WHI was first proposed.
Flash
ahead to 2002, when the WHI oestrogen plus progestin trial was stopped early
because of an increased risk of breast cancer (HR = 1 26 CI = 100–159) CHD (HR=129 CI=102–163) , Stroke (HR = 1 41 CI = 1 07–1 85) , and PE (HR = 2 13 CI = 1 39–3 25) (WHI Writing Group,
2002). Immediately following the publication of these trial results in July of
that year, oral HT prescriptions began a steady decline, while a slight
increase was seen in the use of vaginal formu-lations. In 2003, the FDA, in
conjunction with some members of Congress, launched a national campaign to
provide information and increase aware-ness about the recent findings on
menopausal use of HT (FDA, 2003). In April 2004, the results of the WHI
oestrogen-alone trial were published (WHI Writing Group, 2004). The
investigators noted an increased risk of fatal and non-fatal strokes (HR = 1 39 CI = 1 10–1 77) and venous thrombosis (HR
= 1 47 CI = 1 04–2 08 ); no significant
difference in risk of CHD (HR = 0 91CI = 0 75–1 12) , colorec-tal cancer HR = 1 08 CI = 0 75–1 55 , total cancer (RR = 0 93 CI = 0 81–1 07) , or all cause (HRR = 104 CI=091–112) .
The
effect on breast cancer was uncertain (HR = 0 77 CI = 0 59–1 01) and there was an
increased benefit on bone fractures (HR = 0 70 CI = 0 63–0 79) .
In
March of 2005, an NIH State-of-the-Science Conference on the Management of
Menopause Related Symptoms took place to discuss and form consensus on menopause-related
symptoms and preventive and treatment modalities. The report discusses HT as
‘menopause hormonal therapy’, reflecting the belief that menopause is a natural
state of being for women of a certain age and not a disease state (NIH, 2005).
After the release of results from the WHI, a black box statement pertaining to
cardiovas-cular risks was added to the label for oestrogen. This statement read
‘The Women’s Health Initiative (WHI) reported increased risks of myocardial
infarction, stroke, invasive breast cancer, pulmonary emboli, and deep vein
thrombosis in postmenopausal women during 5 years of treatment with conjugated
equine estrogens (0.625 mg) combined with medroxyproges-terone acetate (2.5 mg)
relative to placebo.’ The warn-ing goes on to state that the FDA assumes these
findings will hold for all HT formulations containing oestrogen and suggests
that HT drugs should be used in the lowest doses necessary for the shortest
duration possible (FDA, 2003).
Related Topics
