Determining Activation Energies
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Mechanisms of Organic Reactions
By using the above methods, the rate constants for most organic reactions can be obtained.
DETERMINING ACTIVATION ENERGIES
By
using the above methods, the rate constants for most organic reactions can be
obtained. Rate constants, by virtue of the fact that they relate directly to
the passage of reactants over the barrier of the rate-determining step, can be
used to probe the energy and structure of the activated complex. The energy of
the activated complex corresponds to the height of the activation barrier for
the rate-determining step. The barrier height can be calculated by the
Arrhenius equation
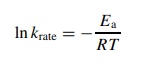
In
krate = - Ea/RT
where
krate is the rate constant
of a reaction, R is the ideal gas
constant, and T is the absolute
temperature. The Arrhenius activation energy Ea is determined from plots of ln k versus 1/T at various
temperatures and largely corresponds to the enthalpy of activation since Ea = ΔH± + RT. The enthalpy and
entropy of activation and hence the free energy of activation are determined by
the Eyring equation
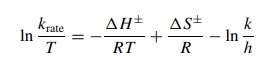
where
k is Boltzmann’s constant and h Planck’s constant. Plots of ln(krate/T ) versus 1/T give straight lines with slope – ΔH±/R and
intercept ΔS ± /R − ln(k/ h). Each
can be numerically evaluated to give ΔH±, ΔS±, and
finally ΔG± by ΔG±
= ΔH± − TΔS±.
Related Topics
