Aluminium
| Home | | Inorganic Pharmaceutical Chemistry |Chapter: Essentials of Inorganic Chemistry : The Boron Group - Group 13
The element aluminium has the atomic number 13 and chemical symbol Al. Aluminium forms a diag-onal relationship with beryllium.
Aluminium
Introduction
The element aluminium has the atomic number 13
and chemical symbol Al. Aluminium forms a diag-onal relationship with
beryllium. The name ‘aluminium’ derives from the salt alum (potassium alum,
Initially, it was very difficult to prepare pure aluminium and
therefore it was regarded as a very precious substance. In the mid-1800s,
aluminium cutlery was used for elegant dinners, whereas it is nowadays used as
lightweight camping cutlery. In 1886, the manufacture of aluminium by
electrolysis of bauxite started, and the price for pure aluminium dropped
significantly. Aluminium is a soft, durable and lightweight metal, which makes
it attractive to many applications. Nowadays, aluminium is mainly used for the
construction of cars and aircrafts and can be found in packaging and
construction materials.
Biological importance
The human body contains around 35 mg of Al3+, of
which ∼50% is
found in the lungs and ∼50% in
the skeleton. There is no known biological role for Al3+ and,
indeed, the human body has developed very effective barriers to exclude it.
Only a minimal fraction of Al3+ is taken up from the diet in the
gut, and the kidneys fairly quickly excrete most of it. The bones can act as a
sink for Al3+ if the blood concentration is high and release it
slowly over a long period. The brain is vulnerable to Al3+ and
usually the blood–brain barrier prevents Al3+ entering the brain. Al3+
can sometimes act as a competitive inhibitor of essential elements such as Mg2+,
Ca2+ and Fe2+/3+ because of their similar ionic radii and
charges. It is important to note that at physiological pH, Al3+
forms a barely soluble precipitate Al(OH)3, which can be dissolved
by changing the pH (see Equations 4.8 and 4.9) .
A normal adult diet contains typically between 2.5 mg/day and up
to 13 mg/day Al3+, but patients on aluminium-containing medication
can be exposed to more than 1000 mg/day. Typically, ∼0.001% is absorbed in the digestive tract, but
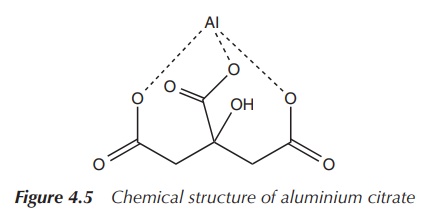
it can be around 0.1–1.0% when it is in the form of aluminium citrate (Figure
4.5).
Al3+ can accumulate in the human body if natural limits are crossed, for example, intravenous admin-istration or patients on dialysis, or when the kidneys are impaired and therefore not able to excrete Al3+ sufficiently. Under normal circumstances, Al3+ would not accumulate in the human body. Nevertheless, in 1972, Alfrey et al. described the new syndrome of progressive dialysis encephalopathy, the so-called dialysis dementia, which was seen in patients being treated with haemodialysis for 15 months or more. The symptoms include speech disorders, problems with the bone mineralisation and general signs of dementia. Investigations showed that brain scans were normal and that there was no connection to the Alzheimer’s disease, as neither neurofibrillary tangles nor senile plaques were found. Increased serum and bone concentrations of Al3+ were found in patients who were on haemodialysis, and the connection was made to the toxicity of the Al3+ present in the dialysate solution.
Nowadays, the use of modern Al3+-free dialysate solutions
or new techniques (e.g. reverse osmosis) prevents ‘dialysis dementia’ [6a].
Al3+ and its use in water purification
Al3+ is used in the purification of
water. Lime (CaO) and aluminium sulfate Al2(SO4)3
are added to waste water in order to accelerate the settling or sedimentation
of suspended matter . The addition of lime increases the pH of the water
slightly (Equation 4.10). The water becomes more basic, which promotes the
precipitation of Al3+ as Al(OH)3 (Equation 4.11).
CaO(s) + H2O(l)
→ Ca2+(aq) + 2OH−(aq) (4.10)
Al3+(aq)+ 3OH−(aq)
→ Al(OH)3(s) (4.11)
Al(OH)3 precipitates as a gelatinous precipitate
which slowly settles. During this process, it incorporates suspended soil,
colloidal material and most bacteria. The water is filtered before leaving the
treatment plant in order to remove the flocculate and the vast majority of the
Al3+. WHO guidelines allow a maximum con-centration for Al3+
of 0.2 mg/l .
Aluminium-based adjuvants
An adjuvant is an agent or a mixture of agents that possesses
the ability to bind to a specific antigen. Adju-vants are added to vaccines in
order to increase the antibody responses to the vaccination and/or to stabilise
the preparation. Adjuvants can absorb many antigenic molecules over a wide
surface area, thus enhancing the interaction of immune cells with the
presenting antigens and leading to an increase of the immune response
stimulation. Some adjuvants (including aluminium-based ones) can function as a
slow-release delivery sys-tem. They trap the antigen in a depot created by the
adjuvant at the injection site. From there, the antigen is slowly released,
which causes a steady stimulation of the immune system.
Aluminium-based adjuvants have a long-standing tradition and
have been used for more than 50 years. They are the most widely used adjuvants
in human and veterinary vaccines and regarded as safe if applied correctly. Al3+
salts are the only kind of adjuvant licensed by the FDA. They are also the only
kind of adju-vants used in anthrax vaccines for humans in the United States.
Anthrax vaccine contains Al(OH)3, as do the FDA-licensed diphtheria,
haemophilus influenzae type B, hepatitis A, hepatitis B, Lyme disease,
pertussis and tetanus vaccines. In many countries, vaccines for children
contain aluminium-based adjuvants .
The adjuvant effect of potassium alum (KAl(SO4)2⋅12H2O) was first discovered in
1926. Researchers examined diphtheria toxoids precipitated with alum and were
able to show that an injection of this alum precipitate led to a significant
increase in immune response. Leading on from this research, alum has found
widespread use as an adjuvant. Vaccines containing alum as adjuvant are
referred to as alum-precipitated vaccines. Unfortunately, it has been
shown that alum precipitations can be highly heterogeneous. The homo-geneity of
the preparation depends on the anions and the conditions present at the point
of precipitation .
Subsequent research showed that aluminium hydroxide (Al(OH)3)
hydrogels can be pre-formed in a stan-dardised manner and be used to absorb
protein antigens to form a homologous preparation. Following on from this
research, researchers have shown that it is possible to co-precipitate
aluminium phosphate (AlPO4) and the diphtheria toxoid in order to
form active vaccines. These vaccines are called aluminium-absorbed vaccines and,
in contrast to alum-precipitated vaccines, the antigens are distributed
homogeneously. Nowa-days, aluminium-absorbed vaccines have taken over from
alum-precipitated ones. Nevertheless, there is a lot of ambiguity found in the
literature, where both terms are interchangeably used .
In summary, immunisation vaccines containing adjuvants are more
effective than those without them. Typ-ical adjuvants are alum [KAl(SO4)2⋅12H2O], Al(OH)3, AlPO4,
Al2O3, but oxides of other metals, such as ZrO2,
SiO2 and Fe2O3, are also under investigation.
The formation of the aluminium hydrogels is generally achieved
by reacting Al3+ ions (from compound such as AlCl3) under
alkaline aqueous conditions. Conditions are strongly regulated, as even
smallest changes to parameters such as temperature, concentration and others
can influence the quality of the hydrogel. Alu-minium phosphate gels are
typically produced by reacting Al3+ salts in the presence of
phosphate ions under alkaline conditions .
The mode of action is highly complex and still not fully
understood. Initial theories included the physical absorption of the antigen,
which is still considered as an important feature, and the gradual release of
antigen from the injection side with the adjuvant working as an agglomeration.
The latter theory was disproved quickly. Research has shown that antigens need
to be adsorbed to the adjuvant before the immunisation reaction. It is believed
that the adjuvant will then present the antigen to the immunocomponent of the
targeted cell .
Antacids
The function of antacids is to neutralise excess stomach acid.
They also exhibit cytoprotective effects towards attacks against the gastric
mucosa. They are additionally known to heal gastric and duodenal ulcerations;
nevertheless, the mechanism is still uncertain.
Antacids have been in use for the past 2000 years, and the
initial formulations were based on CaCO3 (coral and limestone).
Nowadays, the antacid/anti-gas market is a significant income stream for the
pharmaceutical industry and the demand for antacids is expected to grow. The
number of people suffering from heartburn increases with an ageing population,
more stressful lifestyles and changing eating habits such as eating out more
often.
Aluminium hydroxide (Al(OH)3) has several medical
applications. It is used as an antacid for treating heartburn as well as acid
indigestion (reflux oesophagitis). It is also known to have healing properties
of peptic ulcers. In patients suffering from kidney failure, who show elevated
serum phosphate levels (hyper-phosphataemia), Al(OH)3 is used as a
phosphate binder.
Al(OH)3 is an amphoteric compound,
which means it can react as a base or as an acid. In its application as an
anti-acid, Al(OH)3 reacts with any excess stomach acid (mainly HCl)
with the formation of AlCl3 and water (Equation 4.12).
Al(OH)3 + 3HCl → AlCl3
+ 3H2O (4.12)
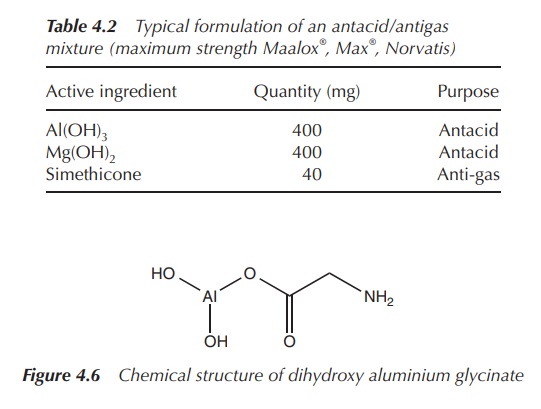
Al(OH)3 is known to cause constipation, so formulations of anti-acids often include a combination with Mg2+ antacids. Usually, oral antifoaming agents, such as simethicone, are added in order to reduce bloating and discomfort/pain. Simethicone is a mixture of poly(dimethyl siloxane) and silica gel, which decreases the surface tension of gas bubbles (Table 4.2).
Ancient anti-acid formulations contained
sodium bicarbonate (baking soda, NaHCO3), which resulted in a rapid
reaction with the gastric acid. The result was an increase in the gastric pH
and the production of CO2 gas as a by-product (Equation 4.13). Large
doses of NaHCO3 can cause alkaline urine and this can result in
kidney problems. Acid neutralisation using Al(OH)3 does not produce
CO2 and therefore these side effects can be avoided.
NaHCO3 + HCl → NaCl
+ H2O + CO2 (4.13)
Aluminium glycinate [Al(NH2CH2COO)(OH)2]
(Figure 4.6) is also used in anti-acid formulations. For example, Gastralgine®
contains, amongst other ingredients, dihydroxy aluminium glycinate [Al(NH2CH2COO)(OH)2],
Al(OH)3, magnesium trisilicate and simethicone. It is known to have
additionally protective effects from ulcers.
Aluminium-based therapeutics – alginate raft formulations
Heartburn is the primary symptom of the so-called
gastro-oesophageal reflux disease (GERD), which is caused by the oesophageal
influx of gastric HCl from the stomach. There are also close links to oesophageal
cancer, which has a very low survival rate. Relief can be achieved with the use
of alginate raft formulations, which typically contain alginic acid, NaHCO3,
magnesium trisilicate and Al(OH)3. Alginates are natural
polysaccharide polymers which are isolated from brown seaweeds.
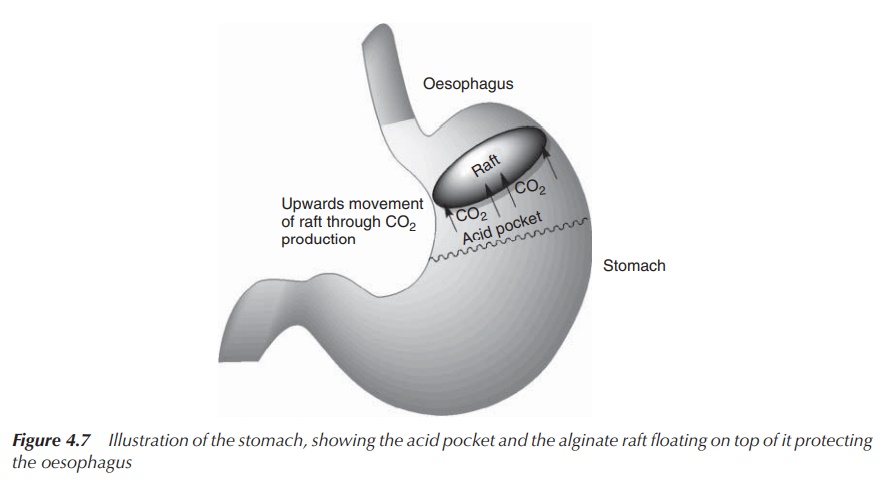
In the acidic stomach, alginate salts and
alginic acids precipitate to form a low-density viscous gel. When the mixture
comes into contact with gastric HCl, the gel matrix formation occurs. HCO3−,
which is trapped in the gel, leads to the formation of CO2 gas
(Equation 4.13). The gas bubbles trapped in the gel convert it to foam and
provide buoyancy, allowing the gel to float on the surface of stomach contents
(like a raft on water). Al(OH)3 provides an additional capacity to
neutralise any excess stomach acid (Equation 4.12). The raft physically acts as
a barrier to gastric reflux and moves into the oesophagus during reflux. It
acts as mobile neutralising sealant in the oesophageal space when the gastric
pressure is high. Once the pressure reduces, the raft drops back into the
stomach and can be digested (Figure 4.7).
Phosphate binders
Hyperphosphataemia, that is, increased levels of serum
phosphate, is a disorder commonly seen in patients with end-stage renal
(kidney) disease where the kidneys are not able to excrete excess phosphate as
a result of a low renal clearance rate. This disorder is often seen in patients
who are on dialysis treatment. Persistent hyperphosphataemia results in renal
osteodystrophy, that is, the weakening of bones due to disturbances in the
calcium and phosphate metabolism.
Generally Al3+-containing drugs are given in order to
promote the binding of phosphate in the gut. Antacids containing Al(OH)3
can be used as phosphate binders. When Al(OH)3 enters the acidic
stomach (pH ∼ 1),
Al3+ ions are formed. Some Al3+ ions will be absorbed in
the stomach, but the majority is passed to the distal intestines, where the pH
is significantly increased (pH 6–8.5). In this high pH range, Al3+
freshly precipitates as a colloidal, amorphous Al(OH)3. Its large
surface area adsorbs phosphate ions (usually in form of HPO42−)
and passes them through the remaining intestine without decomposition, as the
pH is too high. The Al3+-phosphate complex (AlPO4) is
then excreted via the faeces.
Aluminosilicates can also be used as a phosphate binder and is,
for example, the active ingredient in Malinal®. In contrast to
Al(OH)3, which acts as an efficient PO4 3−
binder directly, aluminosilicates need prior exposure to a acid in order to
produce free Al3+. Once the free Al3+ is formed, it
follows the same mode of action.
Initially, aluminium-based phosphate binders were also used in
dialysis exchange fluids, especially in patients being treated with
haemodialysis. Nevertheless, as a result of the exposure to high concentrations
of Al3+ salts, relatively high concentrations were found in
patients. A significant number of patients developed dementia symptoms after 15
or more months of treatment, which was linked to the high Al3+
concentrations in the body including the brain [6a, 10].
Antiperspirant
Aluminium trichloride (AlCl3) was the first compound
that was used as an antiperspirant. The mechanism of action is still under
investigation, but it appears to act by forming a plug of Al(OH)3
within the sweat duct. AlCl3 is a very strong antiperspirant and
only advised by doctors if normal antiperspirants do not work. Leading brands
of antiperspirants contain usually a ∼20% aluminium hexahydrate solution in an alcoholic base. It is
thought to work by blocking the openings of the sweat ducts. It tends to work
best in the armpits. However, it may also work for sweating of the palms and
soles. It can also be applied to the face, taking care to avoid the eyes.
Potential aluminium toxicity
The excessive use of aluminium preparations negatively
influences human health. Excessive intake of Al3+ has been found to
accumulate in sensitive loci and can lead to pathological aberrations and
result in dialysis dementia or similar symptoms. It is important to note that
Al3+ is a major component in over-the-counter drugs such as
antacids. Special attention has to be given by the dispensing pharmacist, and
the patient has to be made aware of the consequences of overdoses of Al3+-containing
products. Al3+ is known to have embryonic and foetal toxic effects
in humans and animals, causing osteomalacia, which is the softening of the
bones due to defective bone mineralisation .
Albumin and transferrin bind around 95% of serum aluminium,
which is then cleared mainly via the kidneys (a small amount can be found in
the faeces). In healthy humans, only 0.3% of orally administered aluminium is
absorbed, whereas it has the potential to accumulate when the GI tract is
bypassed, for example, in intravenous infusions .
Related Topics
