Silicon-based drugs versus carbon-based analogues
| Home | | Inorganic Pharmaceutical Chemistry |Chapter: Essentials of Inorganic Chemistry : The Carbon Group
Silicon chemistry has been of interest as a source for the design of novel pharmaceutically active compounds.
Silicon-based
drugs versus carbon-based analogues
Silicon chemistry has been of interest as a source for the
design of novel pharmaceutically active compounds. Why is it possible to
introduce a silicon group or replace a carbon centre by silicon and what are
the resulting changes? Carbon and silicon are both group 14 elements exhibiting
similarities and differences:
Valency:
Silicon and carbon both possess four valence electrons as they show an
analogous electron configuration (C: [He]2s22p2; Si:
[Ne]3s23p2).
Coordination
number: Unlike that of carbon, the chemistry of silicon is influenced
by the availability of its 3d
orbitals to be involved in additional bonding interactions. Silicon is
therefore capable of increasing its coordination number from 4 to 6 and thus
forming isolatable penta- and hexacoordinated silicon-based compounds.
Nevertheless, for silicon a coordination number of 4 (sp3
hybridisation) is favoured especially over coordination numbers 2 (sp
hybridisation) and 3 (sp2 hybridisation). Consequently, the
formation of double and triple bonds is disfavoured in contrast to carbon-based
reaction centres.
Bond
length: Silicon has a larger covalent radius than carbon, resulting in
the formation of longer bonds than
carbon–carbon bonds (typical C—C bond length = 1.54 Å), silicon–silicon bonds
(typical Si—Si bond length = 2.33 Å) and silicon–carbon bonds (typical Si—C
bond length = 1.89 Å). As a result, silicon-containing compounds show higher
conformational flexibility and therefore steric arrange-ments different from
analogous carbon-based compounds. Differences in interaction with proteins and
consequently alterations of pharmacodynamics and pharmacological profiles have
been observed.
Electronegativity:
Silicon is more positive than the neighbouring carbon (electronegativity according
to Pauling: Si = 1.90, C = 2.55),
resulting in different bond polarisation of analogous carbon–element and
silicon–element bonds. As a result, chemical reactivity and bond strength can
differ significantly. This can provide improved or altered potency if carbon
moieties are switched to silicon-based ones within pharmacophores, especially
if hydrogen bonding is involved in the mode of action (Figure 5.4).
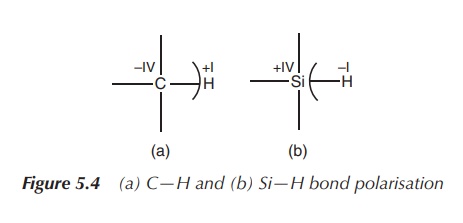
Lipophilicity: In
general, silicon-based compounds demonstrate an enhanced lipophilicity in comparison
to their carbon-analogous due to
their different covalent radii. This provides an interesting opportunity for
exploitable pharmacokinetic potential in drug design, for example, for drugs
that are prone to hepatic metabolism. Silicon-based compounds involved in
hepatic metabolism have been observed to exhibit an increased half-life when
compared to their carbon analogues. Increased lipophilicity is also believed to
be useful in the design of drugs that are supposed to cross the blood–brain
barrier. Therefore silicon analogues with their increased lipophilicity can be
very interesting drug candidates.
Currently, there are only a small number of silicon-containing compounds under investigation for pharma-ceutical use. Silicones are the only silicon-based compounds widely used in medicine. These oligosiloxanes and polysiloxanes have no carbon analogues and they are widely used for plastics, implants, catheters and many other applications.
The limitations of novel organosilicon compounds are very often
attributed to insufficient funding and poor evidence of demonstrated efficacy.
There is also an on-going debate about the toxicity of silicon-based
therapeutics. So far, no increased systematic toxicity of silicon-containing
compounds in comparison to their carbon-analogous has been detected.
Nevertheless, several organosilicon compounds have made it to
clinical trials. In the following sections, a couple of interesting examples
ranging from steroids being used by bodybuilders to anticancer and antispastic
drugs under development are presented.
Introduction of silicon groups
A convenient method to introduce a silicon group is through the
so-called silylation. A hydrogen atom that is bonded to a heteroatom (sulfur,
nitrogen or oxygen) is exchanged by a silyl group (see Figure 5.5).

Carbon silylation, that is, the introduction of a silicon group
next to a carbon centre, is also used for the design of novel drugs. This
approach potentially allows changing the properties of the novel drug candidate
significantly. It can lead to enhanced blood stability, increased cell
penetration and altered pharmacokinetics. Several compounds have entered
clinical trials, including the muscle relaxant silperisone (Figure 5.6) .
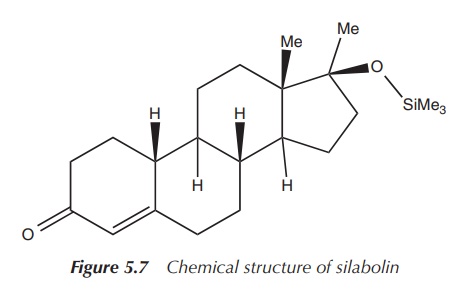
1. Silabolin
The anabolic compound silabolin is an example of a drug in which
this silylation approach has been used. Silabolin is an injectable steroid
containing a trimethylsilyl group, and was and still is used as an anabolic
preparation by bodybuilders. It is known to have a relatively low androgenic
activity like the natural anabolic hormone testosterone. Silabolin was
officially registered in the (former) USSR as a domestic anabolic drug. It is
believed to influence the protein synthesis in humans. Silabolin itself is a
white powder, which is sparingly soluble in ethanol but not soluble in water .
Its propensity to cause heart and liver defects is under discussion and its
effectiveness is being critically discussed amongst bodybuilders (Figure 5.7) .
2. Silperisone
Tolperisone is a centrally acting muscle relaxant used, for example, in the treatment of acute muscle spasms in back pain. Previous in vitro and in vivo studies in mice have demonstrated that silperisone may have the potential to reduce both central nervous system depressing and motor side effects. Phase I clinical trials were conducted with doses up to 150 mg/day.
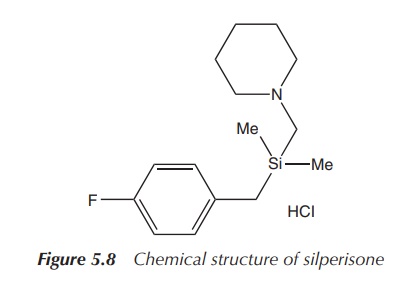
No adverse side effects
were detected, and the observed plasma levels were deemed to be effective in
preclinical trials. Nevertheless, chronic toxicities were observed in animal
studies and the research was discontinued (Figure 5.8) .
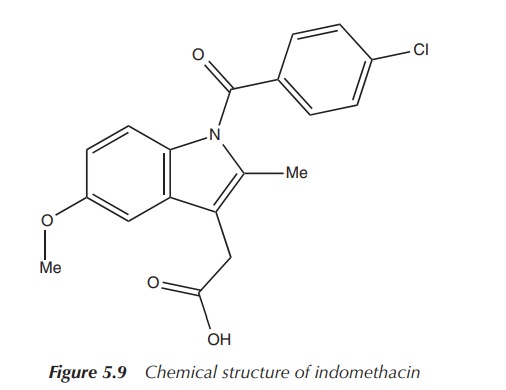
3. Indomethacin
Indomethacin (see Figure 5.9) is a nonsteroidal
anti-inflammatory agent used in pain and moderate to severe inflammation in
rheumatic diseases and other musculoskeletal disorders. It is a COX
(cyclooxygenase) inhibitor and therefore interrupts the production of
prostaglandins .
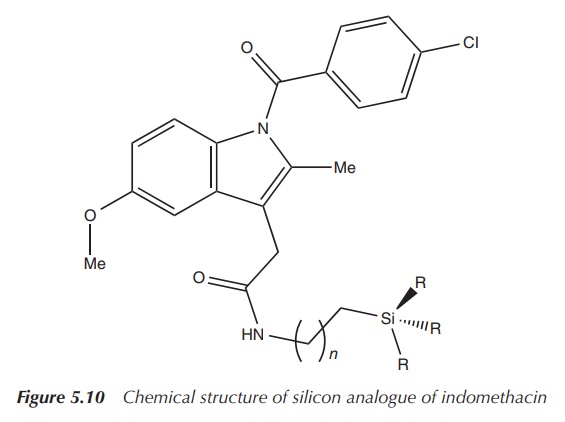
A series of new silicon compounds, based on the structure of
indomethacin, have been synthesised and are under investigation as novel
anticancer agents. The carboxyl group of indomethacin was reacted with a series
of amino-functionalised silanes. The resulting products have been shown to be
significantly more lipophilic and more selective to COX-2. Furthermore, in vitro testing has shown an increased
uptake of the new compounds at the tumour site. The silane-functionalised
indomethacin derivatives exhibited a 15-fold increased antiproliferative effect
when tested against pancreatic cancer (Figure 5.10) .
Silicon isosters
The carbon/silicon switch strategy, meaning the replacement of carbon centres by analogous silicon groups in known biologically active reagents, is currently mainly used for the development of novel silicon-based drug candidates .
The idea is that the new silicon-based drug candidates have the
same chemical structure, with one carbon atom exchanged by a silicon one. The
resulting physiochemical changes include, amongst others, altered bond length
and changes in the lipophilicity. These alterations can have a significant
effect on the biological activity of these novel silicon-based compounds. A
variety of these compounds have been synthesised and tested ; two examples are
presented in the following:
1. Sila-haloperidol
Haloperidol is an analogue of the dopamine D2 receptor antagonist and is an older antipsychotic drug. The drug is used in the treatment of schizophrenia, a neuropsychiatric disorder. Schizophrenia is characterised by symptoms such as hallucinations, delusions and disorganised speech. It is believed that schizophrenia is caused by problems involving the dopamine regulation in the brain. In general, antipsychotic drugs work by blocking the dopamine D2 receptors .
Haloperidol is such an antipsychotic drug, which was developed
in the 1950s and entered the clinic soon after that. Its use is limited by the
high incidence of extrapyramidal symptoms (movement disorders caused by drugs
affecting the extrapyramidal system, a neural network which is part of the
motor system) . Nev-ertheless, haloperidol may be used for the rapid control of
hyperactive psychotic states and is popular for treating restlessness in the
elderly.
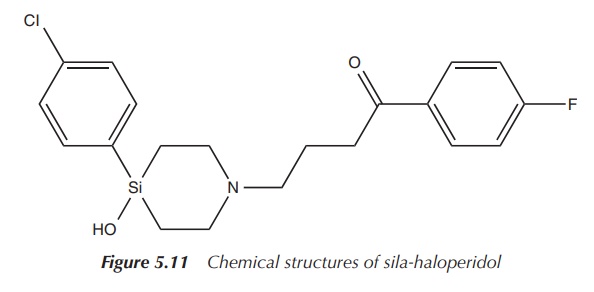
The silicon analogue, sila-haloperidol, has been synthesised by
a sila-substitution of the quaternary R3COH carbon atom of the
4-hydroxy-4-(4-chlorophenyl)piperidin-1-yl group of haloperidol (see Figure
5.11). Chemical analyses have shown that haloperidol and sila-haloperidol both
exist as two analogous conformers but with a different conformer ratio for the
carbon and silicon analogues. Biological studies have also shown large
differences between the metabolic pathways of the silicon and carbon analogues.
Radiolabelling studies have shown similar potencies of the silicon and the
carbon compounds at the human dopamine hD1, hD4 and hD5
receptors. Sila-haloperidol was significantly more potent with the hD2
receptor, thus giving hope to improved side effects related to the metabolism .
2. Sila-venlafaxine
Venlafaxine is a serotonin and noradrenalin reuptake inhibitor
(SNRI) and is used as an antidepressant. Com-pared to tricyclic
antidepressants, it lacks the antimuscarinic and sedative side effects.
Nevertheless, treatment with venlafaxine can lead to a higher risk of withdrawal
symptoms .
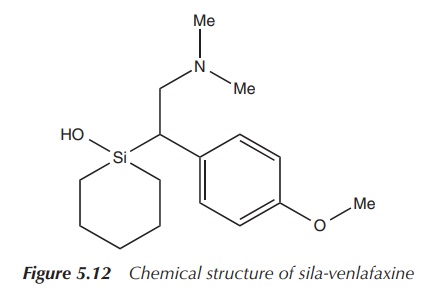
The silicon analogue, rac-1-[2-(dimethylamino)-1-(4-methoxyphenyl)ethyl]-1-silacyclohexan-1-ol,
has been synthesised and tested for its biological properties. The
hydrochloride salts were examined for their efficacy in reuptake inhibition assays
for serotonin, noradrenalin and dopamine. It was concluded that the
carbon–silicon switch changed the pharmacological profile significantly in
regard to the reuptake inhibition depending on the stereoisomer. (R)-Sila-venlafaxine was found to be
consistent with selective reuptake inhibition at the noradrenaline inhibitor
(Figure 5.12) .
Organosilicon drugs
There are several classes of silicon compounds with a clinical use or a proposed biological activity that have no apparent carbon analogues. These compounds use the properties specific to silicon, mainly its ability to form molecules with a penta- and hex-acoordinated silicon centre.
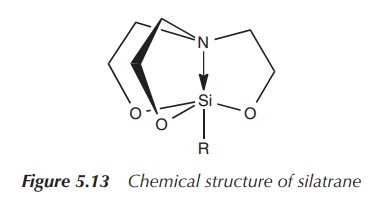
Silatranes are silicon compounds in which the central silicon
atom is pentacoordinated. Silatranes can be highly toxic depending on the
organic rest at the silicon centre. Aryl
and 2-thienyl-substituted silatranes have been proposed as rodenticides
. These compounds are known for their self-detoxification, result-ing in a low
hazard for dermal toxicity or long-lasting secondary risk of poisoning (Figure
5.13) .
Silatranes substituted with alkyl, alkenyl and other groups are
significant less toxic and are under evaluation for a variety of biological or
clinical applications ranging from the stimulation of collagen biosynthesis to
the proposed use as anticancer agents.
Silicones (oligo and polysiloxanes) are the
most widely used class of silicon-based com-pounds clinically. Silicones can be
found in plastics, lubricants, catheters, implants and a variety of other
medically used items. Silicone fluids, such as simethicone, are known for their
antifoaming properties. Sime-thicone is an orally administered suspension
containing polysiloxanes and silicon dioxide. It is an antifoaming agent and is
used to reduce bloating by decreasing the surface tension in bubbles. Excessive
formation of gas bubbles in the stomach and intestines can be painful and can
also be of hindrance for any ultrasound examination. Simethicone can be found
in antacids and in suspensions given to babies against colic.
Related Topics
