Redox Potential
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Redox Potential
The redox potential may be defined as a quantitative expression of the tendency of a compound that has to give or receive electrons.
Redox Potential
INTRODUCTION
The redox potential
may be defined as a quantitative expression of the tendency of a compound that
has to give or receive electrons. The redox potential may be compared with an
acid-base reaction. In the case of acid–base reaction, there is the transfer of
a proton from an atom in one molecule to the atom in another molecule, while in
the case of oxidation–reduction reaction there is an electron transfer. Since
living organisms function at an optimum redox potential range, which varies
with the organism, it might be assumed that the redox potential of the
compounds of a certain type would correlate with the observed biological
effect. This correlation is applicable for all compounds of similar structure
and physical properties.
The redox potential
of a system may be calculated from the following equation: Eh = E1–0.06/n (concentration of
reductant/concentration of oxidant), where
Eh :
redox potential of the system being studied
E10
: standard potential at given pH
n: number of
electrons transferred
However,
there are a number of reasons why only a few satisfactory correlations have
been observed.
●
The
redox potential applies to a single reversible ionic equilibrium, which does
not exist in a living system.
●
A living
cell carries on many reactions simultaneously involving oxidation of ionic and
a nonionic character, some of which are reversible and others are irreversible.
●
The
access of a drug to the sites of oxidation-reduction reactions in the intact
animal is hindered by the complex competing events occurring during absorption,
distribution, metabolism, and excretion.
Therefore, it is to
be expected that correlations between redox potential and biological activity,
generally, hold only for compounds of very similar structure and physical
properties. In such series, variations in the route of distribution and in
steric factors, which might modify the redox system interaction, would be
minimized.
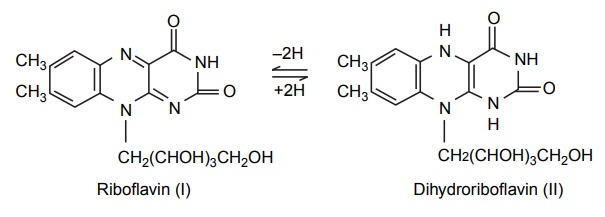
When Riboflavin (I)
accepts electrons, it is converted into its dihydro (II) form. This reaction
has a redox potential E0 =
–0.185 volt. Kuhn (1943) prepared the analogue in which the two methyl groups
of riboflavin were replaced by chlorine. The resulting compound had a potential
of E0 = –0.095 volt, and
its antagonistic properties were suggested as being due to the dichloro-dihydro
form being a weaker reducing agent than the dihydro form of riboflavin. It may
be absorbed at the specific receptor site, but may not have a negative potential
to carry out the biological reductions of riboflavin.
Reist et
al (1960) prepared the nonredox analogues of riboflavin as potential anticancer
agents. Replacement of the N5-nitrogen of dihydroriboflavin
(1,5-dihydro-7,8-dimethyl-10-ribitylisoalloxazine) by a methylene group (III)
would be expected to have a profound effect on the redox potential as compared
to riboflavin. Similarly, replacement of the N5-nitrogen of
dihydroriboflavin by an isopropylidene group (IV) fixes the molecule in the
dihydro form, thus, eliminating the redox system completely.
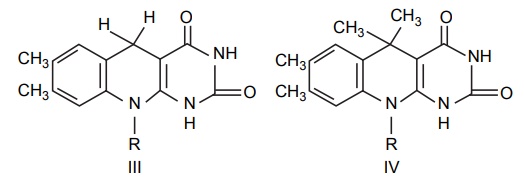
Although
(IV) is derived from dihydroriboflavin (II) rather than from riboflavin, the
redox enzyme system employing riboflavin coenzymes utilizes both the oxidized
and reduced forms; thus, analogues of either I or II should be effective
antagonists.
Craig et
al (1960), studied a series of substituted phenothiazine with regard to
potentiometric titration, electrode potentials, and their correlation with
anthelmintic activity and measured them in the biological assay using mixed
infestation of Syphacia obvelata and Aspicularis tetraptera in mice. From
these studies, it appeared that two factors were necessary for their activity,
namely, the ability to form a high proportion of a stable semiquinone radical
(as measured by the index potential in aqueous CH3COOH) and the
presence of free 3 or 7 position.
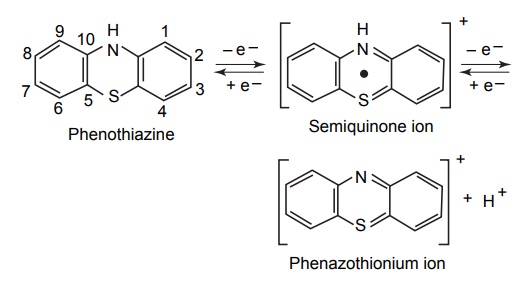
In
addition to the two factors mentioned above, Craig et al (1960) also noted that
only these compounds with electrode potential in the range of 550–850 mV in
aqueous CH3COOH had significant activity. If the toxic or paralyzing
effect of the phenothiazines were due to an inhibition by the semiquinone of
the oxidation–reduction system in the parasite, it would seem reasonable that
active phenothiazines would have reduction potentials corresponding to these of
oxidation–reduction enzyme system or the system which they inhibit. At similar
potentials, the semiquinone concentration would be maximal, and thus,
facilitate or compete with the electron transfers in the enzyme system
involved.
For example, it has
been suggested that the semiquinone of chlorpromazine is responsible for the
inhibition of certain oxidoreductase in vitro and some of the biological
activities of phenothiazines correlates with the formation of their
semiquinones in vivo.
