Steric Features of Drugs
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Steric Features of Drugs
The potential biological activity of a targeted drug molecule solely depends on its physicochemical characteristics, and essentially comprises the nature and type of functional moieties and also the spatial arrangement of such groups in the molecules.
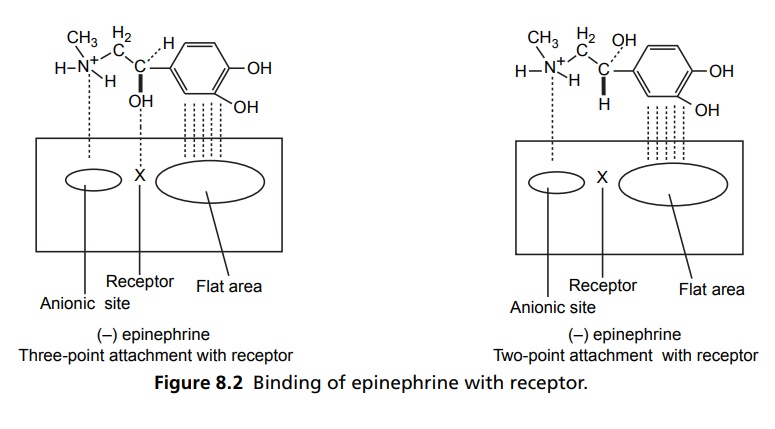
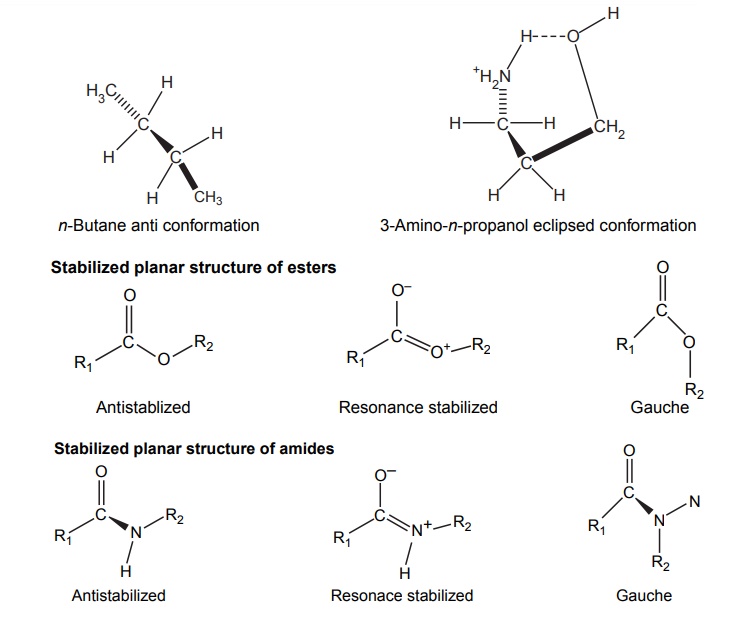
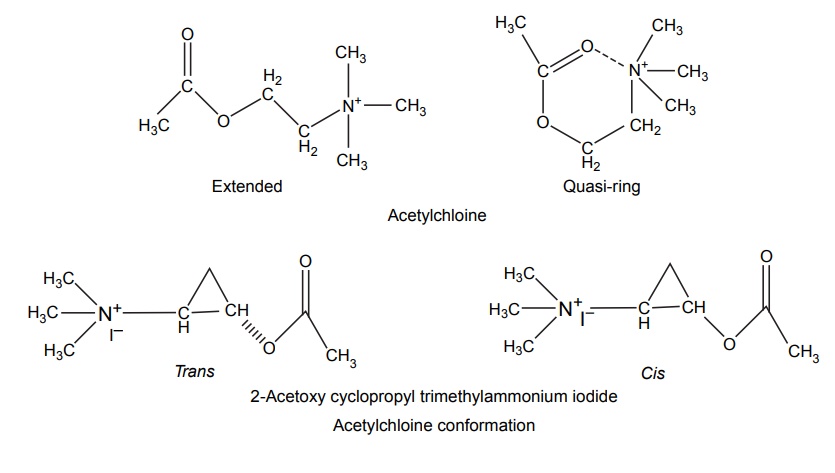
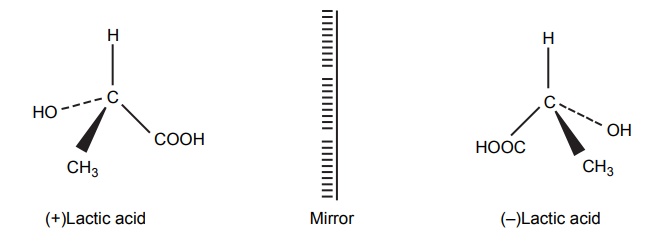
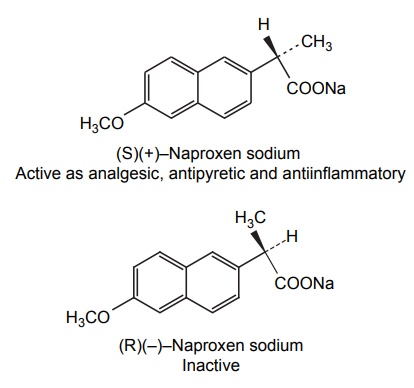
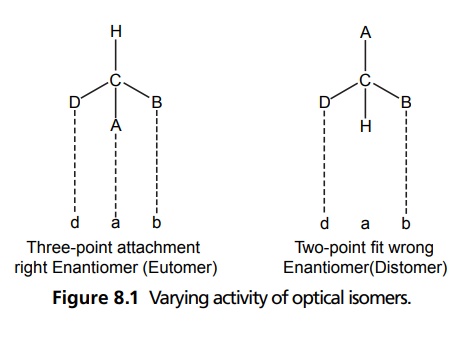
Steric Features of Drugs INTRODUCTION The potential biological activity of a targeted drug molecule solely depends on its physicochemical characteristics, and essentially comprises the nature and type of functional moieties and also the spatial arrangement of such groups in the molecules. Interestingly, the human body itself represents an asymmetric environment, wherein drug molecules interact with proteins and biological macromolecules (receptors). Hence, it is virtually important and necessary that the decisive functional moieties must be strategically located with respect to exact spatial region encircling the targeted drug molecule, so as to enable the crucial and productive bonding interactions particularly with the receptor, thereby potentially accomplishing the desired pharmacologic effect. It is, however, pertinent to state here that the right fitment of correct 3D-orientation of the functional moieties in a drug substance may ultimately result in the formation of an extremely viable and reasonably strong interaction with its receptor. Steric factors determined by the stereochemistry of the receptor site surface and that of the drug molecules are, therefore, of primary importance in determining the nature and the efficiency of the drug-receptor interaction. The drug must approach and fit closely into the receptor surface to evoke the pharmacological action. Hence, the drug must possess a high degree of structural specificity or stereo selectivity. Many drugs show stereo selectivity because mostly receptor binds are optically active biological macromolecules, such as protein, polynucleotide, or glycolipids. For example, diethyl stilboesterol exists in two fixed stereo isomeric forms and trans-diethylstilboestrol is oestrogenic, whereas cis-isomer is only 7% as active. In trans-diethylstilbostrol, resonance interaction and minimal stearic interference tend to hold the two aromatic ring and connecting ethylene carbon atom in the same plane. In geometric isomers, cisand trans-isomers differ in their physical and chemical properties. Therefore, distribution in the biological medium is different. Different arrangements in the space for atoms or groups in single bonds are called conformations. Rotations about the bonds allow interconversion of conformers (conformational isomers). The energy barrier between isomers is often high enough for their independent existence and reaction. Differences in the reactivity of functional groups or interaction with biological receptors may be due to differences in steric requirements of the receptors. Open chains of atoms form an important part of many of the drug molecules. Energy barrier to the free rotations of the chains are present because of the interactions of nonbonded atoms, for example, the atoms tend to position themselves in space so that they occupy staggered positions with no two atoms directly facing each other (eclipsed). Nonbonded interactions in polymethylene chains tend to favour the most extended anticonformations, although some of the partially extended gauche conformations also exist. The conformational isomers show significant differences in biological activities. The potential interaction energy of trimethyl ammonium ion and acetoxy group is lowest in the staggered (also called, though erroneously, trans or transoid) conformation, and highest when the two groups are eclipses (cis or cisoid conformation). It has been suggested that acetylcholine interacts with the muscarinic receptor in fully extended staggered conformation and interacts with nicotinic receptor in folded (gauche) conformation. To study the relationship between the possible conformations of rigid analogues of acetylcholine and their biological effects conformationally rigid analogues of acetylcholine have been used. The cis and trans isomers of 2-acetoxy cyclopropyl trimethyl ammonium iodide are two such compounds. The (+) – trans-isomer in which the quaternary nitrogen atom and acetoxy groups are held apart in a shape approximating that of extended conformation of acetylcholine was found to be almost equipped with acetylcholine at the muscarnic receptor, but shows little nicotinic activity. It is easily hydrolyzed by acetyl cholinesterase. In contrast (+) – cis-isomer showed practically no activity at the nicotinic or muscarinic receptor. The results indicate that acetylcholine assumes staggered conformation at the muscarinic receptors. Optical isomers were further categorized as enantiomers and diastereoisomers. If four different atoms or groups are attached at the four corners of a regular tetrahedron, then the molecule is asymmetric and can exist in two forms. The three-dimensional structure cannot be superimposed on each other, and hence, are different, even though they represent the same structural arrangement of atoms. They bear a relationship to each other corresponding to what exists between an object and its mirror image. Mirror image molecules are not superimposable and are called enantiomers. A tetrahedral carbon atom carrying four groups that are all different, therefore, must invariably constitute a centre of asymmetry and permits two arrangements of the groups in space. This asymmetry calls for the existence of two isomers identical in all respects except optical properties. For example, lactic acid— 2-hydroxy propanoic acid. A chiral compound containing one asymmetric centre has two enantiomers. Although each enantiomer has identical chemical and physical properties, they may have different physiological activities such as the interaction with receptor, metabolism, and protein binding. Examples of enantiomers possessing varying biological activity are the following: Many optical isomers exhibits variation in the intensity of their biological properties. For example, (–)Hyoscyamine is 15–20 times more active as a mydriatic than (+) Hyoscyamine. (–)Hyoscine is 16–18 times as active as (+) hyoscine. (–)Epinephrine is 12–15 times more active as vasoconstrictor than (+) epinephrine. (–)Isoprenaline is 800 times more active bronchodilator than (+) isoprenaline. (+)Nor homoepinephrine is 160 times more active as a pressor than (–) nor homoepinephrine. (+)Amino acids are sweet, whereas (–) –amino acids are either sweetless or bitter. (+)Ascorbic acid has good antiscorbutic properties, whereas (–) ascorbic acid has none. (S) Thalidomide is more teratogenic than (R) thalidomide. Tartaric acid is an example of a compound having two similar asymmetric carbon atoms since each carbon atom has attached hydrogen atom, a hydroxyl group, a carboxyl group (CH OH COOH group). If we represent the configurations of two mirror images’ about each asymmetric carbon atom, then the structures are represented as enantiomers of dand l-tartaric acid. A third structure is possible, which possess a plane of symmetry (achiral) and is, therefore, optically inactive. Such molecules are designated as mesoforms. The relationship of stereoisomers (1) and (2) are enantiomers, but (1) and (3) are not enatiomers, they are called diastereomers. These stereoisomers are not mirror images, unlike enantiomers, but diastereomers possess different physical and chemical properties. Examples of diastereomers possessing varying activity are the following: The difference is due to the interaction of asymmetric carbon atom of the molecule with stereo specific receptors. According to Easson–Stedman hypothesis, if binding ions are specific for one enantiomer, then a threepoint attachment must occur between the enantiomer and the asymmetric surface of the receptor, since only one of the entantiomers will fit, and the other one is only capable of a two-point attachment as shown in Figure 8.1. The enantiomer (Fig. 8.2) that has a high affinity for receptor is called eutomer, whereas the one with a lower affinity is called distomer. The ratio of activity of the eutomer and distomer is called eudismic ratio. Examples of Easson–Stedaman principle: For epinephrine, the benzene ring, benzylic hydroxyl, and protonated amine binds with the hydrophobic or aromatic region, anionic site, and hydrogen bonding centre of the receptor.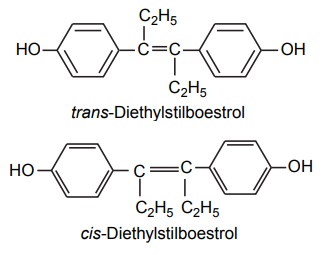
Conformational Isomers
Optical Isomers
ENANTIOMERS
DIASTEREOMERS
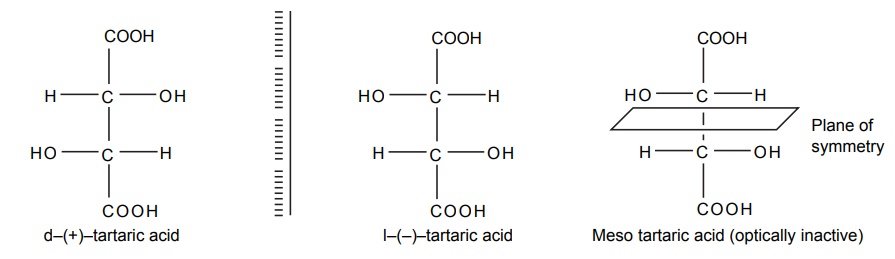
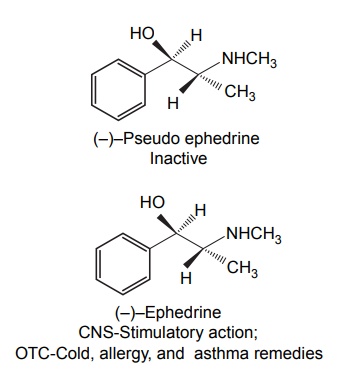
Reasons Behind Varying Activity of Optical Isomers
BINDING OF ENANTIOMERS TO RECEPTORS