Other Methods of Drying
| Home | | Pharmaceutical Technology |Chapter: Pharmaceutical Engineering: Drying
Apart from specialized dryers using infrared or dielectric heating, the chief method of passing heat into a drying solid, other than from a hot airstream, is by conduction from a heated surface.
OTHER METHODS OF DRYING
Apart
from specialized dryers using infrared or dielectric heating, the chief method
of passing heat into a drying solid, other than from a hot airstream, is by
conduction from a heated surface. When a wet solid is placed in contact with a
hot surface, subsequent events depend on the temperature of the surface
relative to the boiling point of the liquid, the nature of the solid, and the
method of heating the surface. It is assumed here that the temperature of the
surface is not hot enough for convective boiling to take place.
Consider
first a cake of finely divided solids saturated with water. A temperature gradient
will be established through the cake and evaporation from the free surface will
take place at a rate governed entirely by the rate of heat
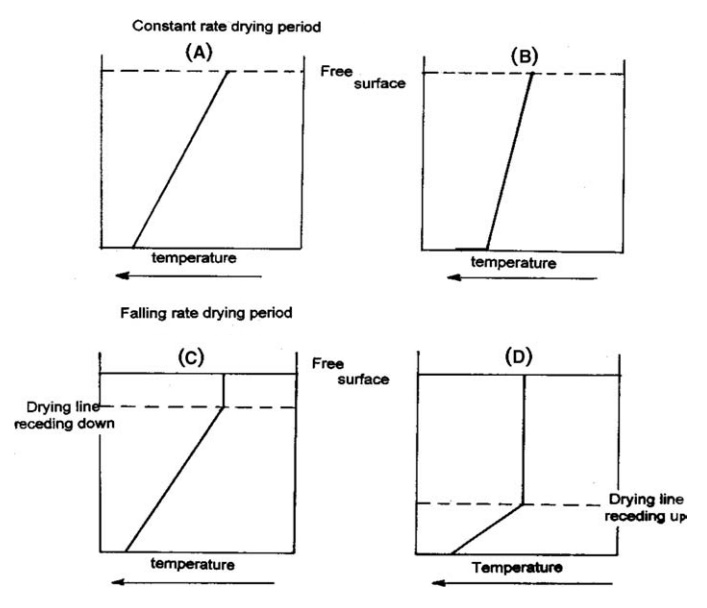
FIGURE 7.6 Drying by conduction of heat from a heated surface.
During this period, the rate of evaporation and the temperature of a particular
layer of cake will be approximately constant. This will continue until
capillary forces are unable to transfer liquid to the free surface at the
required rate. The temperature gradients during this period are given in Figure
7.6A and B for conditions in which the shelf temperature is below and above the
boiling point of the liquid, respectively.
With
a comparatively low heat flux, so that the partially dried cake can conduct
heat away from the hot surface at the required rate, the free surface will dry
and a fictitious drying line will recede slowly into the cake, the vapor
diffusing through the dry cake to the free surface. The temperature gradient
during this falling rate period is shown in Figure 7.6C. If the heat flux is
high, the point at which mobile water can no longer reach the surface is marked
by the onset of drying in a layer adjacent to the hot surface, and vapor is
forced through the wet cake above. As the solid dries, its temperature
increases and a temperature gradient is established through the dry solids to
the drying line that is receding upward. This is shown in Figure 7.6D. The free
surface of the solid appears wet and is at a constant temperature. These
conditions are destroyed when the drying line reaches the surface.
In
either case, a low and falling rate of drying will persist as the absorbed
water is removed. In this form of drying, the heat treatment received by the
solid is not uniform but depends on its position in the cake.
A
hot surface may also be used to dry solutions, such as milk or plant extracts,
which do not readily give porous, crystalline solids on concentration. Apart
from an initial constant rate period, when heat transfer is mainly con-vective,
drying periods are ill defined. As concentration proceeds, the liquor becomes
more viscous and heat transfer is mainly by conduction. Large volume changes
occur between initial and final stages. It is possible to dry thin films of
solution to a solid film, but if deeper layers are taken, a skin is frequently
formed at the free surface that is almost impervious to the vapor. Frothing and
drying to a porous, friable structure will then occur. This may also happen if,
during the upward recession of the drying line, the material above is too
viscous to allow the escape of vapor.
Related Topics
