Antibacterial Sulphonamides
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Antibacterial Sulphonamides
The term sulphonamides are employed as a generic name for the derivatives of para amino benzene sulphonamide (sulphanilamide).
Antibacterial Sulphonamides
INTRODUCTION
The term
sulphonamides are employed as a generic name for the derivatives of para amino
benzene sulphonamide (sulphanilamide). The sulphonamide drugs were the first
effective chemotherapeutic agents to be
employed systemically for the prevention and treatment of bacterial infections
in humans. The sulphonamides are bacteriostatic antibiotics with a wide
spectrum action against most gram-positive bacteria and many gram-negative
organisms. Actually it was found to be the metabolic product of Prontosil,
which is responsible for antibacterial activity, and this has given the
initiation to develop sulphonamides as antibacterial agents.
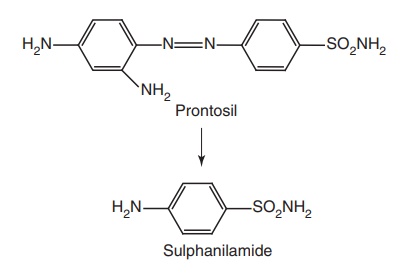
Sulphonamides
are total synthetic substances that are produced by relatively simple chemical
synthesis. The advent of penicillin and, subsequently of other antibiotics has
diminished the usefulness of sulphonamides. Antimicrobial compounds contain
sulphonamide (SO2NH2) group. This group (SO2NH2)
is also present in other compounds, such as antidiabetic agents (e.g.
Tolubutamide), diuretics (e.g. chlorthiazide and its congeners, furosemide, and
acetazolamide), and anticonvulsants such as sulthiame. The sulphonamides exists
as white powder, mildly acidic in character, and they form water-soluble salts
with bases. The pH of sodium salts with some exception, for example, sodium
sulphacetamide, is very high when given intramuscular (IM), the marked
alkalinity causes damage to the tissues.
Microorganisms
that may be susceptible in vitro to sulphonamides include Streptococcus pyogens, Streptococcus pneumoniae, Haemophilus influenzae,
H. ducreyi, Nocardia, Actinomyces, Calymmatobacterium granulomatis, and
Chlamydia trachomatis. The minimal inhibitory concentration ranges from 0.1
μg/ml for C. trachomatis to 4–64
μg/ml for E. coli. Sulphonamides are
selective drugs used to treat urinary tract infections, bacterial respiratory
infections, and gastrointestinal (GI) infections.
Mode of
action: Sulphonamides are
structure analogues and competitive antagonists of para-amino benzoic acid
(PABA). They inhibit dihydropteroate synthetase, the bacterial enzyme
responsible for the incorporation of PABA into dihydropteric acid, and it is
the intermediate precursor of folic acid. Synergistic effect is obtained by a
combination of trimethoprim. The compound trimethoprim is a potent and
selective inhibitor of microbial dihydrofolate reductase, the enzyme that
reduces dihydrofolate to tetrahydrofolate. The simultaneous administration of
sulphonamide and trimethoprim blocks the pathway of cell-wall synthesis
sequentially.
Related Topics
