Antibodies
| Home | | Anatomy and Physiology | | Anatomy and Physiology Health Education (APHE) |Chapter: Anatomy and Physiology for Health Professionals: Lymphatic System and Immunity
B cells divide and differentiate into plasma cells, pro-ducing antibodies or immunoglobulins (Igs) that react to destroy antigens or antigen-containing particles.
Antibodies
B cells divide and differentiate into plasma cells,
pro-ducing antibodies or immunoglobulins (Igs) that react to destroy
antigens or antigen-containing particles. This is called the humoral immune response. There are millions of different
types of T and B cells. Each variety originates from a single early cell to
form a clone of cells
identical to the original cell. Each variety has a certain antigen receptor
respond-ing to only a specific antigen. Humoral immu-nity is also called antibody-mediated immunity. Its name
comes from the antibodies that are present in the body fluids, which used to be
referred to as the body’s “humors.” Antibodies are produced by lymphocytes and
circulate in the blood and lymph, where they mostly bind to extracellular targets such as free
viruses, bacteria, and bacterial toxins. These targets are temporarily
inactivated, then marked for destruction by phagocytes or complement.
Antibod-ies released by plasma cells are involved in humoral immunity,
immediate hypersensitivity reactions, and autoimmune disorders.
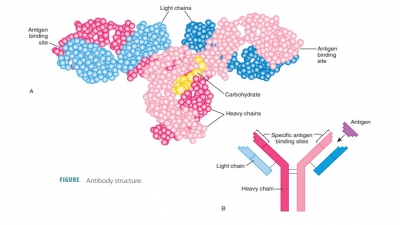
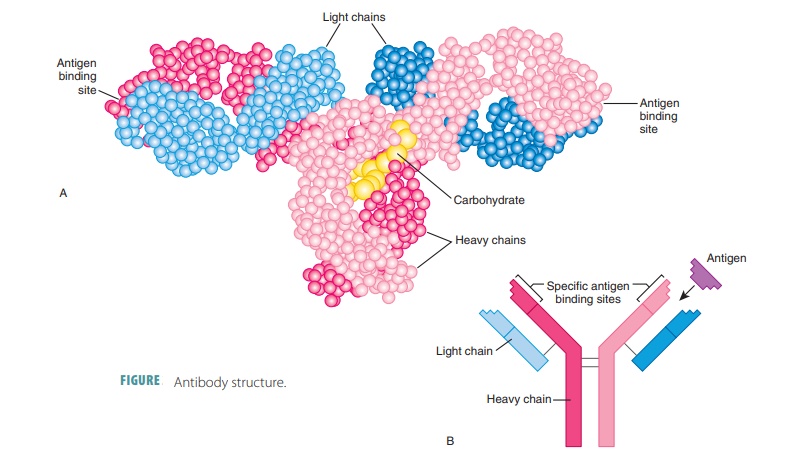
Structure of Antibodies
Each antibody has four looped polypeptide chains connected
by sulfur-to-sulfur or disulfide bonds. The chains form a molecule known as an antibodymonomer. This monomer has two
halves that areidentical. Overall, the antibody monomer is shaped like a “Y” or
a “T” (FIGURE 20-11).
The two heavy chains of
a monomer are identical to eachother. The other two light (L) chains are only half as long
as each H chain but are also identical to each other. Near their middles, the
heavy chains have a hinge region that
is flexible. On each chain the loopsare made up of disulfide bonds. These bonds
cause the intervening parts of the polypeptide chains to “loop out.”
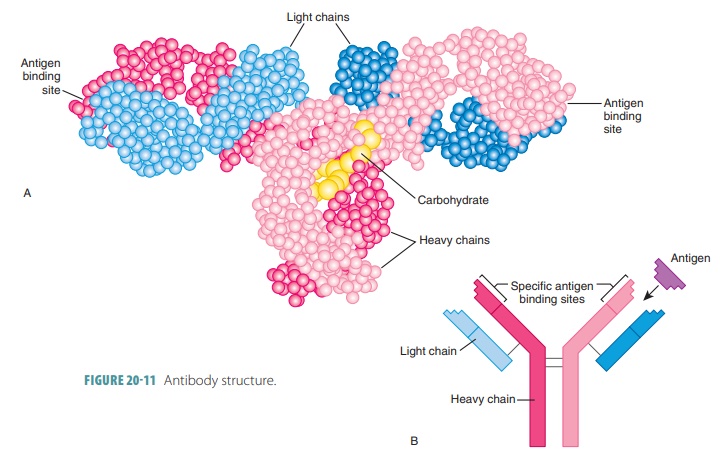
At the other end of each chain, there is a variable region at one end and a constant (C) region atthe other. Extremely
different V regions exist in anti-bodies that respond to different regions.
However, their C regions are nearly the same, or identical, in all antibodies
of the same class. The V regions of the heavy and light chains make up an antigen-bindingsite that has a shape fitting
a certain antigenic deter-minant. As a result, in each monomer arm, each
antibody monomer has two of these antigen-binding regions.
The stem of the antibody monomer is formed by two C regions, which determine the class of antibody. They also have common functions in all antibodies. The C regions are the effector regions of the antibody. They determine the body’s cells and chemicals to which the antibody may bind. They also determine how the anti-body class eliminates antigens. Some antibodies circu-late in the blood, whereas others are found mostly in body secretions. Others fix complement, and still oth-ers can cross the barrier of the placenta. Many other functions exist.
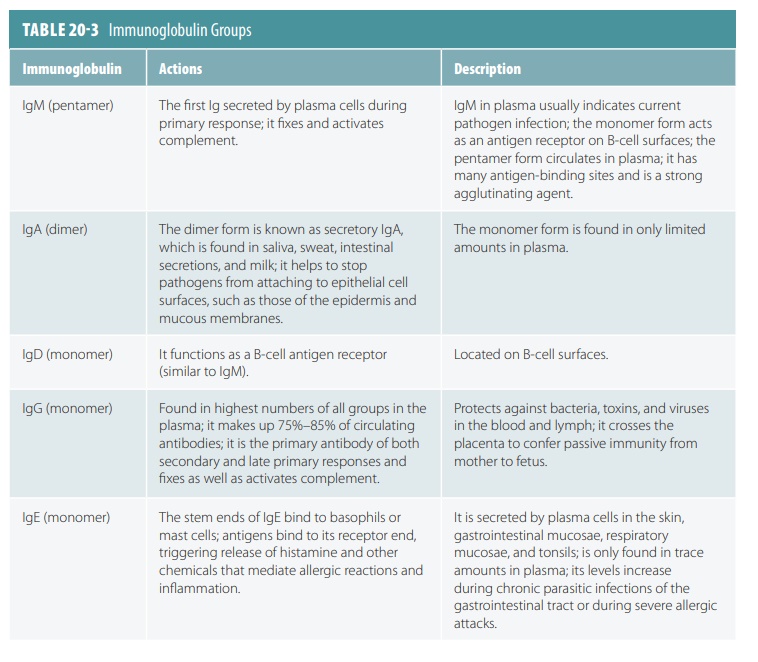
Immunoglobulin Groups
The major five groups of immunoglobulins are IgM, IgA, IgD,
IgG, and IgE. These groups are defined according to the basis of the C regions
in the heavy chains. TABLE
20-3 explains each class of immunoglob-ulins. Compared with the
others, IgM in the blood plasma is much larger. It is made up of five monomers or Y-shaped units that are
linked, forming a pentamer, which has
five parts. A monomer with one part, or a dimer
with two parts, may be formed by IgA, whichis found in tears, saliva,
sweat, and breast milk. The other immunoglobulins all are monomers, with
simi-lar Y-shaped structures. The most abundant antibody in body secretions is
IgA.
Antibody Targets and Functions
Antibodies commonly attack antigens directly, activate
complement, or stimulate inflammation.
They combine with antigens, causing clumping or agglutination or forming insoluble substances,
which is known as precipitation.
Phagocytosis then can occur moreeasily. Sometimes antibodies neutralize the
toxic effects of antigens. Complement activation is gener-ally more important
in protecting against infection than direct antibody attack, however. Antigens
that are close together may result in antibodies binding to antigenic
determinant sites on two separate antigens. The antibodies can then link many
antigens together, creating a three-dimensional immune complex.
When certain antibodies combine with antigens, they trigger
many reactions that lead to the activation of the complement proteins. Effects
include coating the antigen–antibody complexes or opsonization, attract-ing macrophages and neutrophils in a process
called chemotaxis,
making the complexes more susceptibleto phagocytosis, clumping antigen-bearing
cells, rup-turing foreign cell membranes via lysis, and altering viral molecular
structures to make them harmless. In all types of macrophages, phagocytosis
begins with adhe-sion, which is the
attachment of a phagocyte to its target.
1. Describe the common effects of antibodies.
2. Contrast the basic differences between T cells and B cells.
3. Explain the five immunoglobulin groups.
4. Which antibody is secreted first in a primary immune
response?
Related Topics