Bioreactor Design
| Home | | Pharmaceutical Technology |Chapter: Pharmaceutical Engineering: Bioprocessing
A bioreactor is a device within which biochemical transformations are caused by the action of enzymes or living cells.
BIOREACTOR DESIGN
Background
A
bioreactor is a device within which biochemical transformations are caused by
the action of enzymes or living cells. The simple method of shaking cells in a
flask to enhance oxygenation through the liquid surface and to aid mass
transfer of nutrients without cell damage has to be scaled up for industrial
processing.
The
use of biotechnology in the manufacture of pharmaceuticals is of increasing
interest. Consequently, these techniques require attention in the planning of
unit processes.
Bioprocessing
can be considered in terms of small-scale bioreactors, or fermenters, and the
translation of such processes into large-scale economically viable production
operations (Hofmann, 1992; Tatterson, 1994). Bioprocessing is by no means a new
field. The topicality of this subject is due to the increasing interest in the
use of isolated cells and microorganisms as manufacturing tools. It might well
be argued that the technology was developed millennia ago for the purposes of
wine and beer production. More recently, the use of attenuated microorganisms
or isolated antigenic materials for vaccination resulted in fur-ther
developments. In the last decade, interest in genetic engineering and
manipulation of the genetic code of certain microorganisms has produced a
revolution in pharmaceutical manufacturing.
The
major difference between a biotechnological process and other phar-maceutical
manufacturing operations is the need for a bioreactor (Fig. 16.2). A bioreactor
may be required to produce expressed proteins utilizing bacteria, yeast,
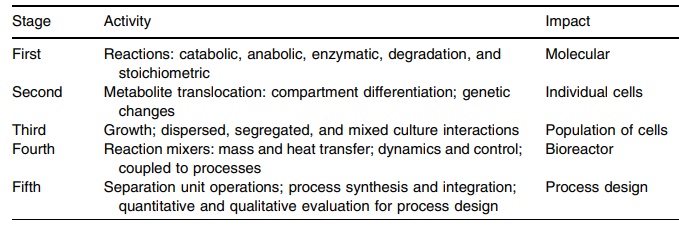
insect, or mammalian cells. Table 16.3 illustrates the various processes
(Prokop and Bajpai, 1991). It would be difficult to describe the various
bioreactor elements and their permutations. Some of the simplest examples of
bioreactors are shown in Figure 16.3.
Some
important factors in bioreactor design are (i) sterility, (ii) broth rheology,
(iii) mass transfer, (iv) mixing, (v) heat transfer, (vi) suspension
homogenization, and (vii) shear sensitivity of microorganisms. The importance
of these design considerations depends on the nature of the biological systems
considered.
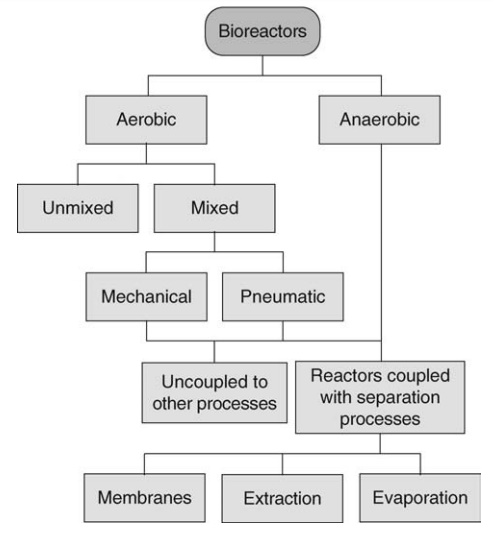
FIGURE 16.2 Types of bioreactors.
TABLE 16.3 Biotechnological Processing
Rheology
The
presence of organized structures in the form of mycelial cells or bio-po1ymers
tends to induce non-Newtonian properties in broth. The power law of plastic
systems (Martin, 1993) may be employed to describe broth rheology. The
viscosity and shear rate are related to the concentration of cell mass in the
system. These correlations are species specific and depend on the stage of
growth in the cell cycle.
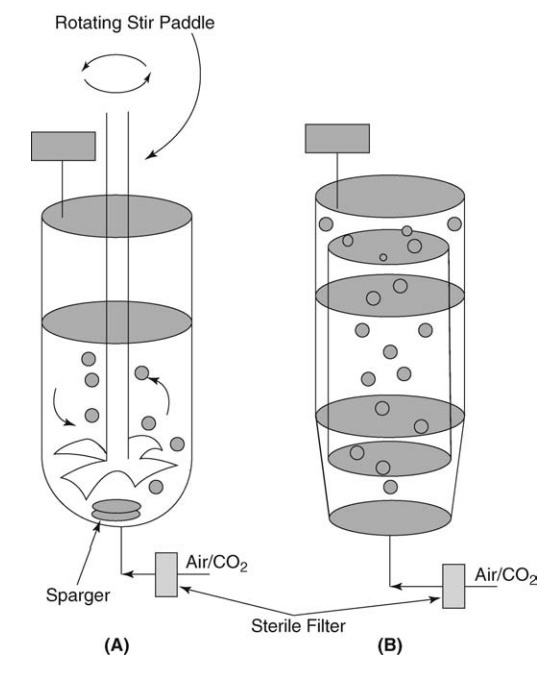
FIGURE 16.3 Bioreactors: (A) stirred tank reactor and (B) airlift fermenter.
Mass Transfer
Although
all nutrient, waste product, and cell integrity issues in growth may be
considered in terms of mass transfer, the most notable of these is oxygen
transfer for aerobic growth. A maximum uptake rate of oxygen exists for any
system, and the design should be based on an understanding of this limitation.
Also the oxygen uptake rate of cells shows a saturation dependence on dissolved
oxygen concentration (CL). Assuming a pseudo–steady state of
dissolved oxygen con-centration, a design value of gas-liquid mass transfer
coefficient, kLα for a biological
system can be specified for a specific reactor as
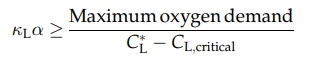
The
term “critical” refers to the point in the oxygen uptake rate versus dissolved
oxygen concentration plot (CL) at which saturation is achieved and
no further oxygen can be dissolved. The gas-liquid mass transfer coefficient
often changes during the course of fermentation because of changes in broth
rheology or through additives, such as antifoaming agents.
Mixing
Concentration
and temperature are influenced by mixing in bioreactors. Total homogeneity
within a system is rarely, if ever, achieved and local variations in mixing
within vessels may affect growth, metabolism, or other molecular expression
phenomena. Operating conditions influence terminal mixing time (time to reach
designated variability associated with complete mixing) and mean circulation
time (time to circulate through specific region once). Charac-terization of
mixing times and the influence of geometric features of reactors under
different operating conditions and scales of operation (bench, pilot, and full
scale) are important if efficiency (time and cost) is to be optimized.
Heat Transfer
Heat
is dissipated mainly by convection across the walls of the jacket or coils. In
aerated systems, metabolic heat production is correlated with oxygen uptake
rate. The maximum metabolic load should be considered in design calculations as
in gas-liquid oxygen transfer. Handbook values are available for heat transfer
on the jacket side, vessel side, and in tubes. In general, heat transfer
becomes a problem only in very large scale operations and in dense microbial
populations, which are frequent with recombinant cells. In other cases,
gas-liquid mass transfer and mixing are the major concerns.
Shear
Agitation
is required to maintain suspensions of the cells. Agitated bioreactors are
designed to maintain complete suspension (no cell mass at the bottom of the
reactor) or a homogeneous suspension. These terms imply stable flocculations
(aggregates) in suspension or homogeneous cell distribution throughout the
suspension.
The
mechanism of shear damage to the cells is not clear. Mycelial or protozoan
cells exhibit shear rate–limited growth, and cell damage has been monitored by
analyzing the concentration of low-molecular-weight nucleotides in the culture
broth.
