Regulatory Initiative
| Home | | Pharmaceutical Technology |Chapter: Pharmaceutical Engineering: Quality by Design
In recent years several regulatory agencies have indicated their desire to have Quality by Design tools adopted.
REGULATORY INITIATIVE
In
recent years several regulatory agencies have indicated their desire to have
Quality by Design tools adopted to justify the various processes employed in
the production of a pharmaceutical product to manage risk associated with the
quality of the product. Notable among these are the guidances of the U.S. Food
and Drug Administration and the relevant guidances (Q8–10) of the
Interna-tional Conference on Harmonization of Technical Requirements for
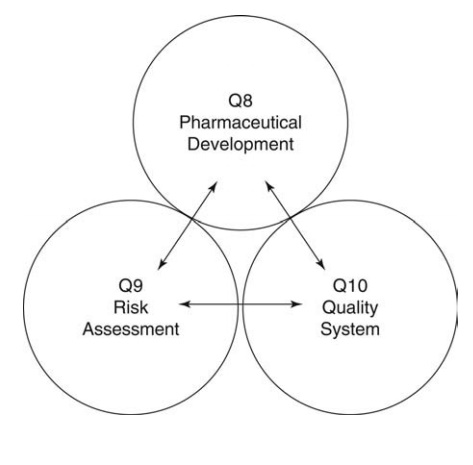
Registration of Pharmaceuticals for Human Use (ICH). Figure 17.1 depicts the
relationships beween the ICH quality chapters and their intent to (i) use
quality systems, such as process analytical technology or measurement and
control of operating var-iables, in (ii) risk analyses, which establishes
priority of these measurements through (iii) statistically designed experiments
in product development that identifies variable parameters and acceptable
variances with respect to mea-sured and monitored phenomena.
Consideration
of each of these items allows process space to be defined. That is the range of
input variable control that is required to minimally impact on the quality of
the product and thereby ensure the uniformity and reprodu-cibility of the final
product derived from the various processes employed.
Quality
by design is a systematic approach to development that begins with predefined
objectives and emphasizes product and process understanding and process control
on the basis of sound science and quality risk management (Pharmaceutical
development Annex to ICH, Q8, 2007).
Quality
by Design is
·
scientific, risk-based, holistic and proactive approach to
pharmaceutical development;
·
deliberate design effort from product conception through
commercializa-tion; and
·
full understanding of how product attributes and process
relate to product performance.