Classes of tetracyclines antibiotics
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Antibiotics
I. Natural tetracyclines II. Semisynthetic tetracyclines i. Methacycline ii. Doxycycline (Vibramycin) iii. Minocycline (Cynomycin, Minolox) iv. Rolitetracycline III. Pro-tetracyclines
Classes of tetracyclines antibiotics
Tetracycline
antibiotics
Tetracyclines
have a ring system of four linear annelated six-membered rings and are
characterized by a common octahydronaphthacenes skeleton. They are potent,
broad-spectrum antibacterial agents effective against gram-positive and
gram-negative aerobic and anaerobic bacteria. As a result, the tetracyclines
are drugs of choice or well-accepted alternatives for a variety of infectious
diseases. Among these, they also play a role in the treatment of sexually
transmitted and gonococcal diseases, urinary tract infections, bronchitis, and
sinusitis remain prominent.
The majority
of the marketed tetracyclines (tetracycline, chlorotetracycline,
oxytetracycline, and demeclocycline) are naturally occurring compounds obtained
by the fermentation of Streptomyces spp. broths.
The semisynthetic tetracyclines (methacycline, doxycycline, minocycline) have
the advantage of longer duration of antibacterial action. However, all these
tetracyclines exhibit a similar profile in terms of antibacterial potency. In
general, their activity encompasses many strains of gram-negative E. coli, Proteus, Klebsiella, Enterobacter,
Niesseria, and Serratia spp., as
well as gram-negative Streptococci and
Staphylococci of particular interest
is the potency of tetracylines against Haemophilus,
Legionella, Chlamydia, and Mycoplasma.
Classes of tetracyclines
I. Natrual
tetracyclines (biosynthetic)
II. Semisynthetic
tetracyclines
III. Protetracyclines
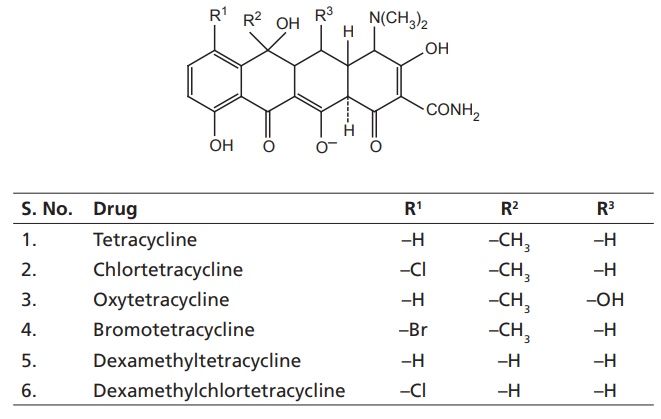
I. Natural tetracyclines
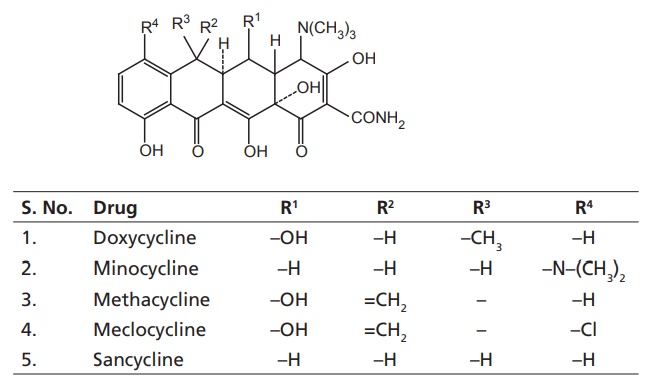
II. Semisynthetic tetracyclines
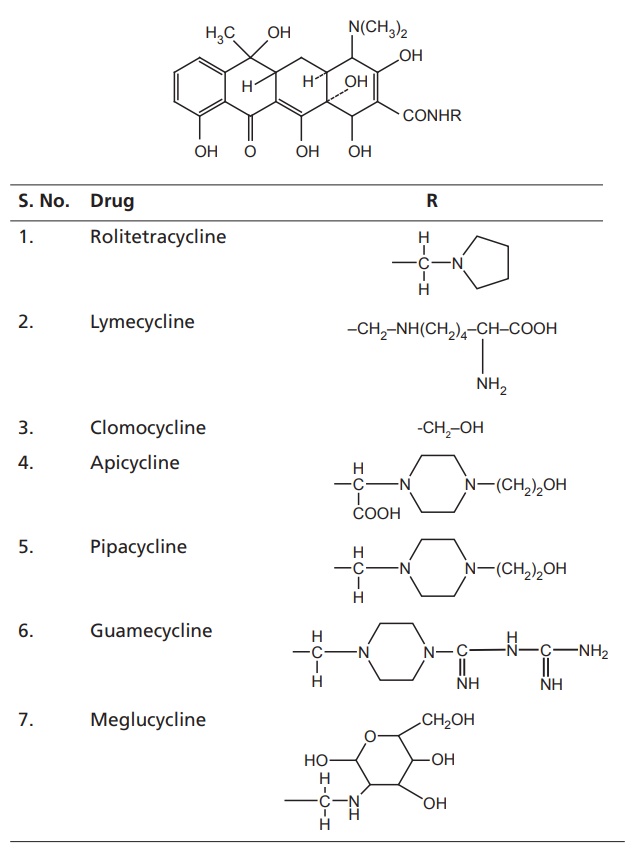
III. Pro-tetracyclines
I. Natural tetracyclines
General mode of action of tetracyclines: In bacterial protein synthesis, the messenger
RNA attaches itself to 30S ribosomes. The initiation complex of mRNA starts the
protein synthesis and polysome formation of the nascent peptide that is
attached to 50S ribosomes. Its specific tRNA transports the next amino acid to
the acceptor site of the ribosome, which is complementary to the base sequence
of the next mRNA codon. The nascent peptide is transferred to the newly
attached amino acid by peptide bond formation. Tetracyclines bind to 30S
ribosomes and the attachment of aminoacyl tRNA to mRNA ribosome complex is
interfered.
Physicochemical properties: These are amphoteric due to the acidic and the
basic substituents, and have low solubility in water (0.5 mg/ml) with strong
acids and bases. They form water-soluble salts in each tetracycline, there are
three ionizable groups present: tricarbonyl methane moiety (pKa 3.3), phenol
diketone moiety (pKa 7.7), and ammonium cation moiety (pKa 9.7).
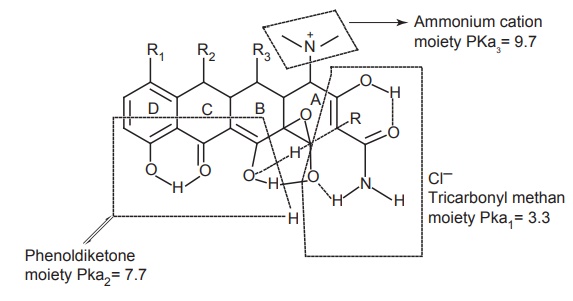
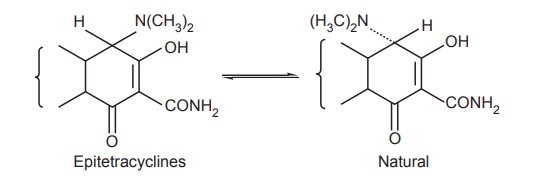
Effect of pH on tetracyclines: An interesting property of tetracyclines is
their ability to undergo epimerization at C-4 position in solutions of
intermediate pH range. These isomers are called epitetracyclines. Under the
influence of the acidic conditions, equilibrium is established in about a day
consisting of approximately equal amount of isomers. Epitetracyclines exhibit
much less activity than natural isomers.
Strong acids
and bases attack the tetracyclines having a hydroxyl group on C-6, causing a
loss in activity through the modification of C-ring. Strong acids produce
dehydration through a reaction involving the C-6 hydroxyl group and C-5a
hydrogen. The double bond formed between the positions C-5a and C-6 induces a
shift in the position of double bond between C-11a and C-12 to a position
between C-11 and C-11a forming the more energetically favoured resonance of the
naphthalene group found in the inactive anhydro tetracyclines.
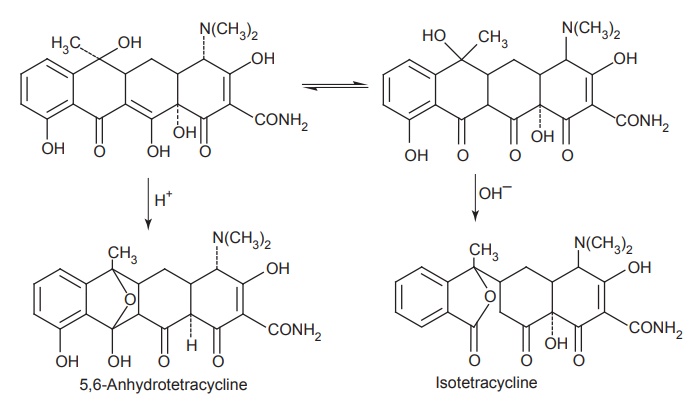
Bases
promote a reaction between the C-6 hydroxyl group and the ketone group at the
C-11 position, causing the bond between the C-11 and C-11a atoms to cleave and
to form the lactone ring found in the isotetracycline.
Effect of metals on tetracyclines: Tetracyclines form stable chelate complexes with
many metals, including calcium, magnesium, and iron. The chelates thus formed
are insoluble in water accounting for the impairment in absorption of most of
the tetracyclines in the presence of milk, calcium, magnesium, and aluminium
containing antacids and iron salts.
The affinity
of tetracyclines for calcium causes them to be laid down in newly formed bones
and teeth as tetracycline calcium orthophosphate complexes. Deposits of these
antibiotics in tooth cause yellow discolouration that darkens because of
photochemical reaction. Tetracyclines are distributed into the milk of
lactating mothers and also cross the placenta into the foetus. The possible
effect of these agents on bones and teeth of the child should be taken into
consideration before they are used during pregnancy or in children under eight
years of age.
II. Semisynthetic tetracyclines
i. Methacycline
Synthesis
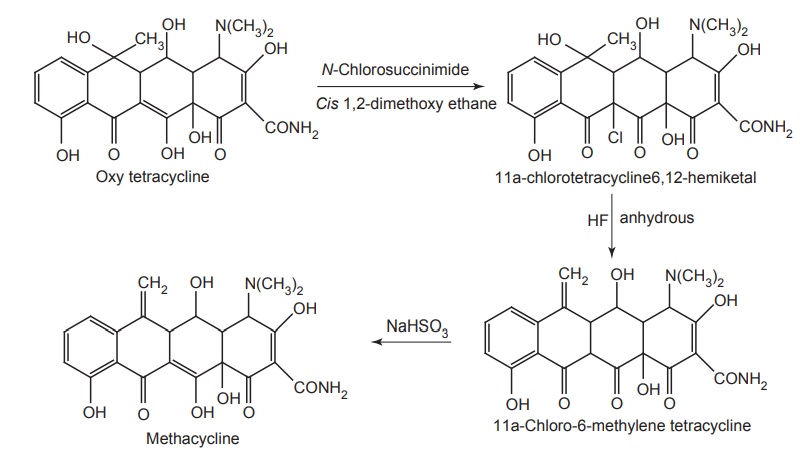
Properties and uses: Methacycline is a yellow crystalline powder,
sparingly soluble in water. It is obtained by the chemical modification of
oxytetracycline. It has an antibiotic spectrum similar to tetracyclines, but
greater potency; about 600 mg of methacycline is equivalent to 1 g of
tetracycline.
ii. Doxycycline (Vibramycin)
Synthesis

Properties and uses: It was first obtained in small yields by a
chemical transformation of oxytetracycline. The 6α-methyl epimer is more than
three times as active as its β epimer.
Dose: In adults, the oral dosage is 100 mg every 12 h.
Dosage forms: Doxcycline HCl capsules I.P., Doxcycline HCl tablets I.P.
iii. Minocycline (Cynomycin, Minolox)

Synthesis
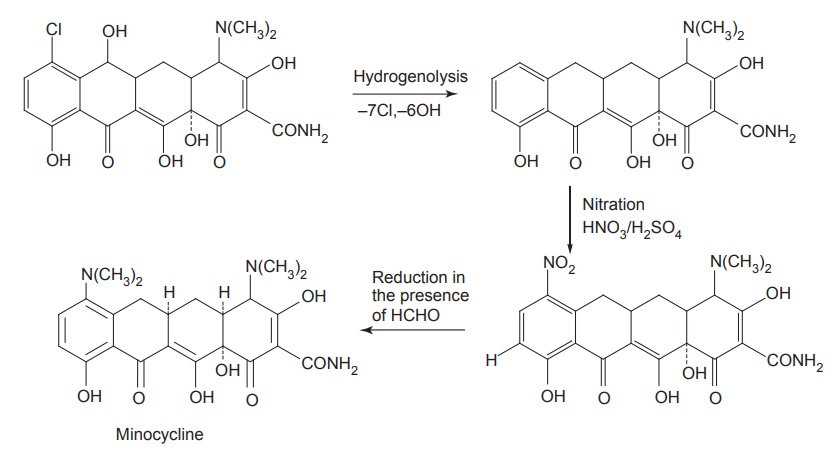
Properties and uses: It is a yellow crystalline powder with slightly
bitter taste, soluble in water. It is very active against gram-positive
bacteria. It is especially effective against Mycobacterium marinum. As a prophylactic against streptococcal
infections, it is the drug of choice. It lacks the 6-hydroxyl group, therefore,
it is stable to acids and does not dehydrate or rearrange to anhydro or lactone
forms.
Dose: The dose orally for adults is 200 mg.
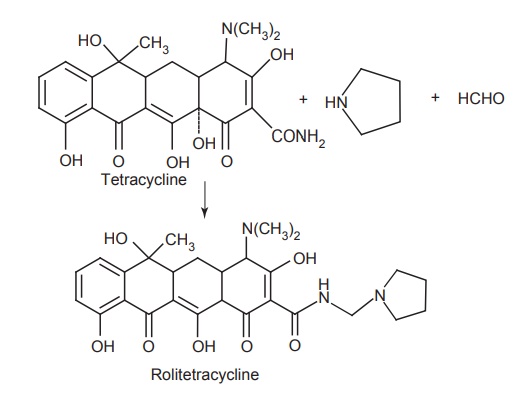
iv. Rolitetracycline
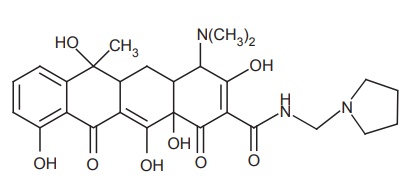
Synthesis
Related Topics
