Fibrinolytics (Thrombolytics)
| Home | | Pharmacology |Chapter: Essential pharmacology : Drugs Affecting Coagulation, Bleeding And Thrombosis
These are drugs used to lyse thrombi/clot to recanalize occluded blood vessels (mainly coronary artery). They are curative rather than prophylactic; work by activating the natural fibrinolytic system
FIBRINOLYTICS
(Thrombolytics)
These are drugs used
to lyse thrombi/clot to recanalize occluded blood vessels (mainly coronary
artery). They are curative rather than prophylactic; work by activating the
natural fibrinolytic system (Fig. 44.3).
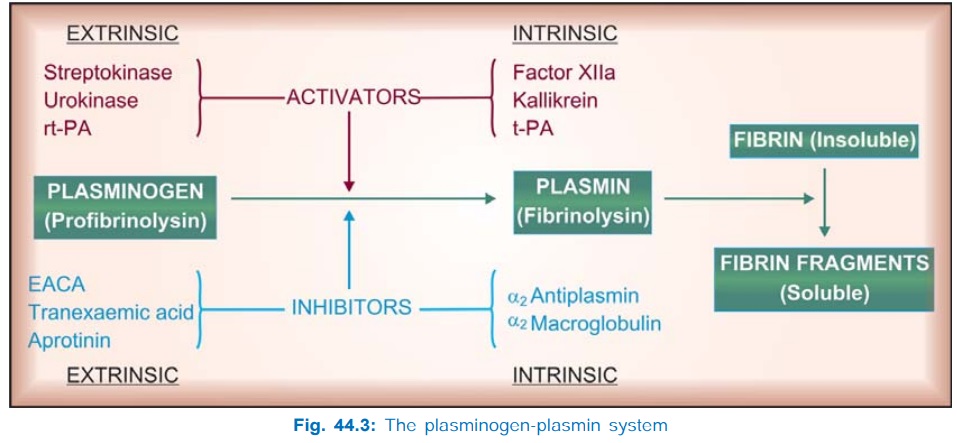
Haemostatic plug of
platelets formed at the site of injury to blood vessels is reinforced by fibrin
deposition to form a thrombus. Once repair is over, the fibrinolytic system is
activated to remove fibrin. The enzyme responsible for digesting fibrin is a serine
protease Plasmin generated from plasminogen by tissue plasminogen
activator (tPA), which is produced primarily by vascular endothelium.
Plasminogen circulates in plasma as well as remains bound to fibrin. The tPA
selectively activates fibrin bound plasminogen within the thrombus, and any plasmin
that leaks is inactivated by circulating antiplasmins. Fibrin bound plasmin is
not inactivated by antiplasmins because of common binding site for both fibrin
and antiplasmin.
When excessive amounts
of plasminogen are activated (by administered fibrinolytics), the α2 antiplasmin is
exhausted and active plasmin persists in plasma. Plasmin is a rather nonspecific
protease: degrades coagulation factors (including fibrinogen) and some other
plasma proteins as well. Thus, activation of circulating plasminogen induces a
lytic state whose major complication is haemorrhage. Even selective activation
of thrombus bound plasmin can cause bleeding by dissolving physiological
thrombi. In general, venous thrombi are lysed more easily than arterial, and recent
thrombi respond better: little effect on thrombi > 3 days old. The
clinically important fibrinolytics are:
Streptokinase Alteplase (rt-PA)
Urokinase Reteplase
Tenecteplase
Streptokinase (Stk)
It is obtained from β haemolytic Streptococci group C. It is inactive as
such: combines with circulating plasminogen to form an activator complex which
then causes limited proteolysis of other plasminogen molecules to plasmin.
Antistreptococcal antibodies present due to past infections inactivate
considerable fraction of the initial dose of Stk: a loading dose is necessary
in the beginning. Its t½ is estimated to be 30–80 min.
Streptokinase is
antigenic; can cause hypersensitivity reactions and anaphylaxis, especially
when used second time in a patient. Repeat doses are also less effective due to
neutralization by antibodies. Fever is common, hypotension and arrhythmias are
reported.
Because of the
availability of newer fibrinolytics which do not pose some of the above problems,
Stk is infrequently used now in developed countries. However, being the least
expensive, it is still widely used in India and other developing countries.
STREPTASE, (freeze
dried powder in vials) 2.5 lac, 7.5 lac and 15 lac IU/vial, ESKINASE, CARDIOSTREP
7.5 lac, 15 lac IU/vial.
For MI: 7.5–15 lac IU
infused i.v. over 1 hr.
For
deep vein thrombosis and pulmonary embolism: 2.5 lac IU loading dose over ½1 hr, followed by 1
lac IU/hr for 24 hr.
Urokinase
It is an enzyme
isolated from human urine; now prepared
from cultured human kidney cells, which activates plasminogen directly and has
a plasma t½ of 10–15 min. It is nonantigenic. Fever occurs during treatment,
but hypotension and allergic phenomena are rare. Indicated in patients in whom
streptokinase has been used for an earlier episode, use has now declined due to
introduction of newer fibrinolytics.
UROKINASE, KDUNASE,
2.5 lac, 5 lac, 10 lac IU per vial inj.
For MI: 2.5 lac IU i.v. over
10 min followed by 5 lac IU over next
60 min (stop in between if full recanalization occurs) or 6000 IU/min for upto
2 hr.
For venous thrombosis and pulmonary embolism: 4400 IU/kg over 10 min i.v. followed by 4400
IU/kg/hr for 12 hr.
Alteplase (recombinant tissue plasminogen activator (rtPA)
Produced by recombinant DNA technology from human tissue culture, it
specifically activates gel phase plasminogen already bound to fibrin, and has
little action on circulating plasminogen. It is rapidly cleared by liver and
has a plasma t½ of 4–8 min. Because of the short t½, it needs to be given by
slow i.v. infusion and often requires heparin coadministration. It is
nonantigenic, but nausea, mild hypotension and fever may occur. It is
expensive.
ACTILYSE 50 mg vial
with 50 ml solvent water.
For MI: 15 mg i.v. bolus
injection followed by 50 mg over 30
min, then 35 mg over the next 1 hr.
For pulmonary embolism: 100 mg i.v. infused
over 2 hr.
Reteplase
It is a modified form
of rtPA that is longer acting, but somewhat
less specific for fibrin bound plasminogen. The longer duration of action enables
bolus dose administration (10 mg over 10 min repeated after 30 min).
Tenecteplase
It is a mutant variant
of rtPA with higher fibrin selectivity and
longer duration of action. A single i.v. bolus dose (0.5 mg/kg) or split into two
doses 30 min apart is given.
The clinical efficacy
and risk of bleeding with reteplase and tenecteplase are similar to alteplase.
Uses Of Fibrinolytics
1. Acute
Myocardial Infarction is the chief indication. Fibrinolytics are an alternative
first line approach to emergency percutaneous coronary intervention (PCI) with
stent placement. Recanalization of thrombosed coronary artery has been achieved
in 50–90% cases. Time lag in starting the infusion is critical for reducing
area of necrosis, preserving ventricular function and reducing mortality. The
benefits of i.v. thrombolytic therapy have been established by large randomised
studies. Aspirin with or without heparin is generally started concurrently or
soon after thrombolysis to prevent re-occlusion.
Alteplase has advantages over streptokinase, including higher
thrombolytic efficacy. However, incidence of haemorrhage is not lower; may even
be higher. Its stronger lytic effect on physiological haemostatic plugs may
compensate for the lesser systemic fibrinolytic state.
Fibrinolytic therapy has also been used in unstable angina,
because many such patients have coronary thrombi.
2. Deep Vein Thrombosis in leg, pelvis, shoulder etc.; up to 60% patients can be successfully
treated. Thrombolytics can decrease subsequent pain and swelling, but the main
advantage is preservation of venous valves and may be a reduced risk of
pulmonary embolism, though at the risk of haemorrhage. Comparable results have
been obtained with Stk, urokinase and rtPA.
3. Pulmonary Embolism: Fibrinolytic therapy
is
indicated
in large, lifethreatening pulmonary embolism. The lung function may be better
preserved, but reduction in mortality is not established.
4. Peripheral
Arterial Occlusion: Fibrinolytics recanalise ~40% limb artery occlusions, especially
those treated within 72 hr. However, it is indicated only when surgical
thrombectomy is not possible. Regional intraarterial fibrinolytics have been
used for limb arteries with greater success. Peripheral arterial thrombolysis
is followed by shortterm heparin and long-term aspirin therapy.
Fibrinolytics have no role in chronic peripheral vascular
diseases.
5. Stroke: Thrombolytic therapy
of ischaemic stroke is
controversial. Trials showing improved neurological outcome with no change in
mortality, as well as those finding significant risk of intracranial
haemorrhage and increased mortality are on record. No net benefit was concluded
by the ATLANTIS trial in patients treated at 3–5 hours of stroke onset.
However, rtPA is approved for use in ischaemic stroke, and current opinion
supports use of i.v. alteplase in carefully selected patients who can be
treated within 3 hours of onset, and in whom intracranial haemorrhage is ruled
out along with all risk factors for bleeding.
Evaluation
All patients with ST
segment elevation myocardial
infarction (STEMI) are candidates for reperfusion therapy. Both shortterm and long-term
outcome is determined by early restoration of flow in the occluded artery,
regardless of whether it is achieved by thrombolysis or by PCI. Best results
are obtained if perfusion can be restored within the first hour (the golden
hour). While the efficacy of fibrinolytics in dissolving the thrombus diminishes
with passage of time (little benefit after 6 hours of MI onset), reperfusion by
PCI is not as much affected by the time lapse. Thrombolysis may be favoured if
it can be started within 1–2 hours of onset. After 3 hours, PCI is favoured.
Moreover, PCI has the advantage of lower bleeding risk, higher grade of flow in
the reperfused artery and reduction in the rate of nonfatal recurrent MI
compared to thrombolysis. As such, wherever available, PCI is being used in
preference. Presence of risk factors for bleeding also favour PCI. However, the
overall 6 month mortality has not been found to differ between either mode of
reperfusion.
Invasive procedures,
such as cardiac catheterization, should be avoided in patients who are to be
given thrombolytics, because risk of bleeding is increased. With concurrent use
of heparin, major bleeding (including intracranial haemorrhage) occurs in 2–4%
patients. The incidence of bleeding is almost similar with Stk, urokinase and
rtPA. Analysis of recent trials has shown that exclusion of heparin reduces
bleeding, and that heparin affords no extra benefit over fibrinolytic + aspirin.
Another analysis has shown that efficacy of Stk and rtPA in MI is similar, but
certain other features favour the newer thrombolytics.
Thrombolytic therapy requires careful patient selection. It is
contraindicated in all situations where the risk of bleeding is increased, such
as— recent trauma, surgery, biopsies, haemorrhagic stroke or peptic ulcer,
severe hypertension, aneurysms, bleeding disorders, diabetes, acute
pancreatitis, etc. Its use in retinal vessel occlusion has been abandoned.
Related Topics
