Oral Anticoagulants
| Home | | Pharmacology |Chapter: Essential pharmacology : Drugs Affecting Coagulation, Bleeding And Thrombosis
A haemorrhagic disease was described in cattle in 1924 which was due to feeding them on spoiled sweet clover hay. The disorder was found to be due to prothrombin deficiency and the toxic principle was identified as bis-hydroxycoumarin in 1939.
ORAL ANTICOAGULANTS
A haemorrhagic disease
was described in cattle in 1924 which was due to feeding them on spoiled sweet
clover hay. The disorder was found to be due to prothrombin deficiency and the
toxic principle was identified as bis-hydroxycoumarin in 1939. It was cured by
feeding alfalfa grass. First clinical use of bis-hydroxycoumarin was made in
1941 and many congeners were added later. Warfarin was initially used as rat poison;
demonstration of its safety led to clinical trial; it is now a commonly
employed oral anticoagulant.
Action And Mechanism
Warfarin and its congeners
act as anticoagulants only in vivo,
not in vitro. This is so because they
act indirectly by interfering with the synthesis of vit K dependent clotting
factors in liver. They apparently behave as competitive antagonists of vit K
and reduce the plasma levels of functional clotting factors in a dose-dependent
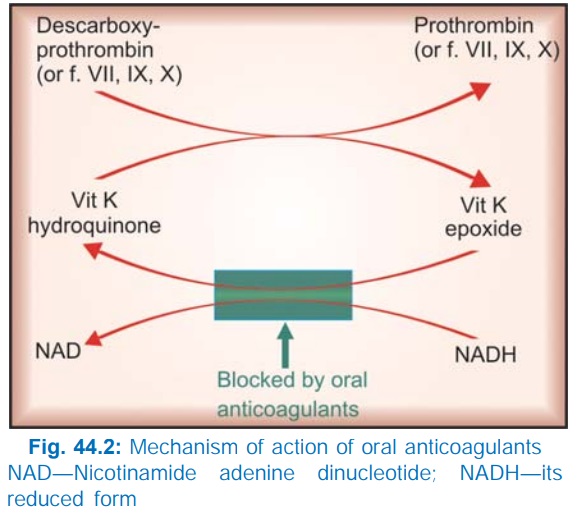
manner. In fact, they interfere with regeneration of the active hydroquinone
form of vit K (Fig. 44.2) which carries out the final step of γ carboxylating
glutamate residues of prothrombin and factors VII, IX and X. This carboxylation
is essential for the ability of the clotting factors to bind Ca2+ and to get
bound to phospholipid surfaces, necessary for coagulation sequence to proceed.
Factor VII has the
shortest plasma t½ (6 hr), its level falls first when warfarin is given,
followed by factor IX (t½ 24 hr), factor X (t½ 40 hr) and prothrombin (t½ 60
hr). Though the synthesis of clotting factors diminishes within 2–4 hours of
warfarin administration, anticoagulant effect develops gradually over the next
1–3 days as the levels of the clotting factors already present in
plasma decline
progressively. Thus, there is always a delay between administration of the drug
and the anticoagulant effect. Larger initial doses hasten the effect only
slightly.
Therapeutic effect occurs
when synthesis of clotting factors is reduced by 40–50%.
Protein C, protein S,
osteocalcin and some other proteins contain glutamate residues that require
vit. K dependent carboxylation. These are also inhibited by oral
anticoagulants, but density of adult bone is not affected, though new bone
formation may be depressed.
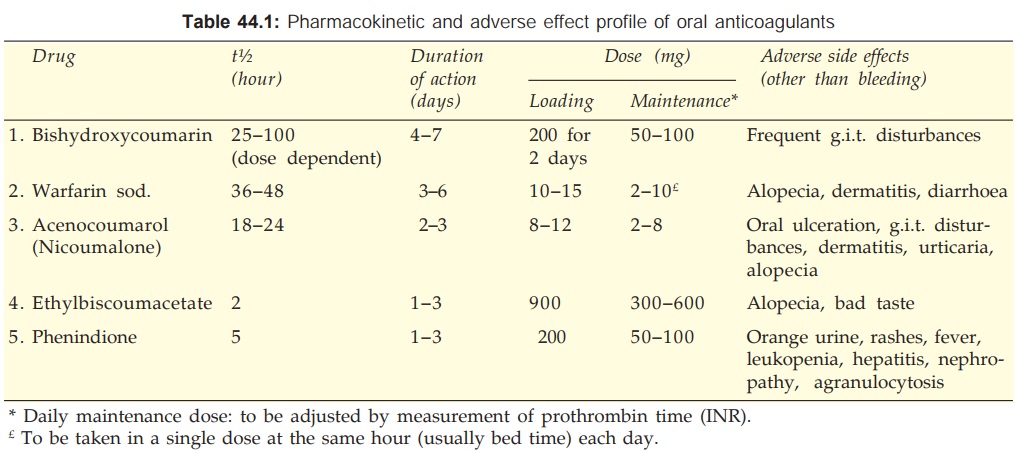
The differences between
different oral anticoagulants are primarily pharmacokinetic and in the adverse
side effects produced by them. These are summarized in Table 44.1.
Recemic
Warfarin sod. It is the most popular oral anticoagulant.
The commercial preparation of warfarin is a mixture of R (dextrorotatory) and S
(levorotatory) enantiomers. The S form is more potent and is metabolized
relatively faster by ring oxidation, while R form is less potent and degraded
by side chain reduction. Both are partially conjugated with glucuronic acid and
undergo some enterohepatic circulation; finally excreted in urine.
Warfarin is rapidly
and completely absorbed from intestines and is 99% plasma protein bound. It
crosses placenta and is secreted in milk; however, quantity of active form is
generally insufficient to affect the suckling infant.
UNIWARFIN 1, 2, 5 mg tabs;
WARF5: 5 mg tab.
Bishydroxycoumarin
(Dicumarol) It is slowly and unpredictably absorbed
orally. Its metabolism is dose dependent—t½ is prolonged at higher doses. Has
poor g.i. tolerance.
DICOUMAROL 50 mg tab.
Acenocoumarol (Nicoumalone) The t½ of acenocoumarol as such is 8 hours, but an
active metabolite is produced so that overall t½ is about 24 hours. Acts more
rapidly.
ACITROM, 1, 2, 4 mg
tabs.
Ethyl biscoumacetate It has a rapid and
brief action; occasionally used to
initiate therapy, but difficult to maintain.
Phenindione It produces more serious nonhaemorrhagic toxic effects: should not be used.
DINDEVAN 50 mg tab.
Adverse Effects
Bleeding as a result
of extension of the desired pharmacological action is the most important
problem: ecchymosis, epistaxis, hematuria, bleeding in the g.i.t. Intracranial
or other internal haemorrhages may be fatal. This is more likely if therapy is
not properly monitored or interacting drugs/contraindications are present.
Treatment: of bleeding due to
oral anticoagulants consists of:
·
Withhold the anticoagulant.
· Give fresh blood transfusion: supplies
clotting factors and replenishes lost blood. Alternatively fresh frozen plasma
may be used as a source of clotting factors.
· Give vit K1—specific antidote, but it takes 6–24 hours for
the clotting factors to be resynthesized and released in blood after vit K
administration.
Adverse effects unrelated to anticoagulation are given in Table
44.1. Cutaneous necrosis is a rare complication that can occur with any oral anticoagulant.
Phenindione produces serious toxicity; should not be used
(though still available).
Warfarin and acenocoumarol are considered to be the most
suitable and better tolerated drugs.
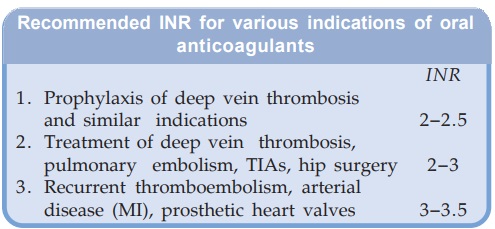
Dose Regulation
The dose of oral
anticoagulant must be individualised by repeated measurement of prothrombin time; the aim is to
achieve a therapeutic effect without unduly increasing the chances of bleeding.
The optimum ratio of
PT during treatment to the normal value (of the testing laboratory) has been
defined for various indications. But this value differs depending on whether
rabbit brain or human brain thromboplastin (Tp) has been used for the test. A
standardized system called the International Normalized Ratio (INR) based on the
use of human brain Tp has been developed by WHO and adopted in all countries.
Factors enhancing effect of oral anticoagulants
are:
·
Debility, malnutrition, malabsorption and
prolonged antibiotic therapy: the supply of vit K to liver is reduced in these
conditions.
·
Liver disease, chronic alcoholism: synthesis
of clotting factors may be deficient.
·
Hyperthyroidism: the clotting factors are
degraded faster.
·
Newborns: have low levels of vit K and
clotting factors (there should be no need of these drugs in neonates anyway).
Factors decreasing effect of oral anticoagulants are:
·
Pregnancy: plasma level of clotting factors is
higher.
·
Nephrotic syndrome: drug bound to plasma
protein is lost in urine.
·
Genetic warfarin resistance: the affinity of
warfarin (as well as of vit K epoxide) to bind to the reductase enzyme, which
generates the active vit K hydroquinone, is low. Dose of oral anticoagulant is
4–5 times higher.
Contraindications
All contraindications
to heparin apply to these
drugs as well. Factors which enhance the effect of oral anticoagulants (see above) should also be taken into
consideration.
Oral anticoagulants
should not be used during pregnancy. Warfarin given in early pregnancy
increases birth defects, especially skeletal abnormalities: foetal warfarin syndrome—hypoplasia
of nose, eye socket, hand bones, and growth retardation. Given later in pregnancy,
it can cause CNS defects, foetal haemorrhage, foetal death and accentuates neonatal
hypoprothrombinemia.
Drug Interactions
A large number of
drugs interact with oral
anticoagulants at pharmacokinetic or pharmacodynamic level, and either enhance
or depress their effect. These interactions are clinically important (may be
fatal if bleeding occurs) and may involve more than one mechanism; the exact
mechanism of an interaction is not always definable.
A. Enhanced
Anticoagulant Action
1. Broad spectrum antibiotics,
inhibit gut flora and reduce vit K production.
2. Newer cephalosporins
(cefamandole, moxalactam, cefoperazone) cause hypo-prothrombinaemia by the same
mechanism as warfarin —additive action.
3. Aspirin: inhibits
platelet aggregation and causes g.i. bleeding—this may be hazardous in
anticoagulated patients. High doses of salicylates have synergistic hypo-prothrombinemic
action and also displace warfarin from protein binding site.
4. Long acting
sulfonamides, indomethacin, phenytoin and probenecid: displace warfarin from
plasma protein binding.
5. Chloramphenicol,
erythromycin, celecoxib, cimetidine, allopurinol, amiodarone and metronidazole:
inhibit warfarin metabolism.
6. Tolbutamide and phenytoin:
inhibit warfarin metabolism and vice
versa.
7. Liquid paraffin
(habitual use): reduces vit K absorption.
B. Reduced
Anticoagulant Action
1. Barbiturates (but not
benzodiazepines), rifampin and griseofulvin induce the metabolism of oral
anticoagulants. The dose of anticoagulant determined during therapy with these
drugs would be higher: if the same is continued after withdrawing the inducer—
marked hypoprothrombinemia can occur— fatal bleeding is on record.
2. Oral contraceptives:
increase blood levels of clotting factors.
Uses Of Anticoagulants
The aim of using
anticoagulants is to prevent thrombus extension and embolic complications by
reducing the rate of fibrin formation. They do not dissolve already formed
clot, but prevent recurrences. Heparin is utilized for rapid and shortlived
action, while oral anticoagulants are suitable for maintenance therapy.
Generally, the two are started together; heparin is discontinued after 4–7 days
when warfarin has taken effect.
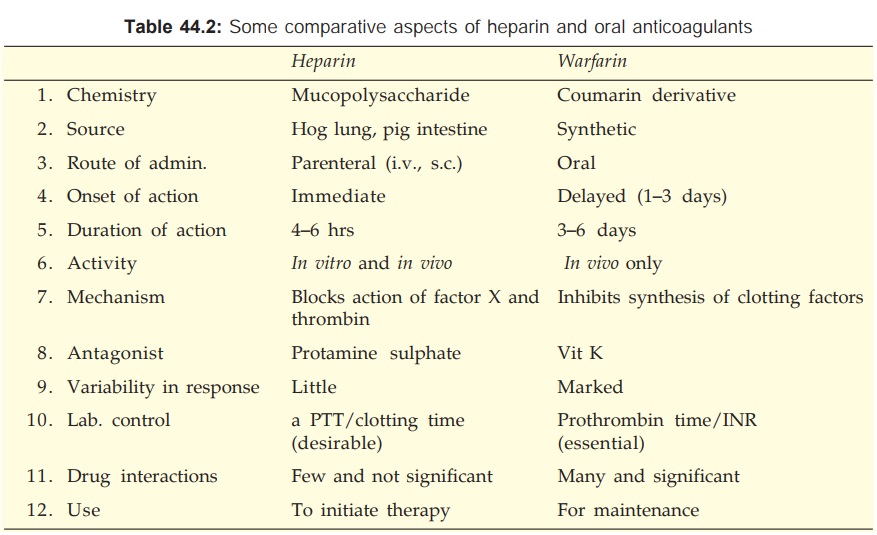
The important features
of heparin and oral anticoagulants are compared in Table 44.2.
Deep Vein Thrombosis And Pulmonary Embolism
Because venous thrombi
are mainly fibrin thrombi,
anticoagulants are expected to be highly effective. The best evidence of
efficacy of anticoagulants comes from treatment and prevention of venous
thrombosis and pulmonary embolism. Prophylaxis is recommended for all high risk
patients including bedridden, old, postoperative, postpartum, poststroke and
leg fracture patients. When deep vein thrombosis/ pulmonary embolism has
occurred, immediate heparin followed by warfarin therapy should be instituted.
Three months anticoagulant therapy (continued further if risk factor persists)
has been recommended by American College of Chest Physicians (2001).
Introduction of low
dose heparin prophylaxis for patients undergoing elective surgery has
considerably reduced the incidence of leg vein thrombosis and pulmonary
embolism in the postoperative period. It has been extended to other situations
needing prolonged immobilization. It is based on the premise that inhibition of
small amount of activated factor X prevents further amplification of active
products—particularly thrombin. This is the regimen of choice: does not need
laboratory monitoring; spontaneous bleeding does not occur. LMW heparin is
being preferred for this purpose. Anticoagulants are of little value in chronic
peripheral vascular diseases.
Myocardial Infarction (MI)
Arterial thrombi are mainly platelet thrombi; anticoagulants
are of questionable value. Their use in acute MI has declined. They do not
alter immediate mortality of MI. It was hoped that anticoagulants will prevent
extension of the thrombus and ward off a recurrent attack. This has not been
supported by the collected statistics. They may benefit by preventing mural
thrombi at the site of infarction and venous thrombi in leg veins. Thus, anticoagulants
may be given for a short period till patient becomes ambulatory. For secondary
prophylaxis against a subsequent attack— anticoagulants are inferior to
antiplatelet drugs.
Heparin (i.v.) for 2–8 days followed by oral anticoagulants for
3 months or low dose s.c. heparin are generally given after recanalization of coronary
artery by fibrinolytic therapy. Heparin is also used during coronary
angioplasty and stent placement.
Unstable Angina
Short-term use of
heparin has reduced the
occurrence of MI in unstable angina patients; aspirin is equally effective.
Current recommendation
is to use aspirin + heparin followed by warfarin.
Rheumatic Heart
Disease; Atrial Fibrillation (AF)
Warfarin/low dose
heparin/low dose aspirin are effective
in preventing stroke (due to embolism from fibrillating atria). The ‘Stroke
prevention in Atrial Fibrillation’ trial and a meta-analysis have shown
warfarin to be more effective than aspirin. Current guideline is to give warfarin
to a target INR of 2–3 in AF patients with high risk for stroke (elderly, heart
failure, etc.), and to reserve aspirin for low risk patients or for those
unable to take warfarin. Anticoagulants are given for 3–4 weeks before and
after attempting conversion of AF to sinus rhythm.
Cerebrovascular Disease
Anticoagulants are of little value in cerebral thrombosis.
They have been used with the aim of preventing clot propagation, but all the
trials conducted, including International Stroke Trial (IST), have failed to
demonstrate significant benefit. Neurological sequelae are similar whether they
are used or not. Moreover, in the initial stages it is difficult to rule out
cerebral haemorrhage (unless CAT scan is done) in which they can be
devastating. They may be used in cerebral embolism, because showers of emboli
are often recurrent and can be prevented by anticoagulants. A late start (after
one week) anticoagulant therapy is advocated by many in case of large embolic
stroke. Oral anticoagulants may be beneficial in transient ischaemic attacks
(TIAs), but antiplatelet drugs are simpler to use and probably better.
Vascular Surgery, Prosthetic Heart Valves, Retinal Vessel Thrombosis, Extracorporeal Circulation, Haemodialysis
Anticoagulants are indicated along
with antiplatelet drugs for prevention of thromboembolism.
Heparin flushes (200 U in 2 ml) every 4–8 hr are used to keep
patent long-term intravascular cannulae/catheters.
Defibrination
Syndrome
‘Disseminated intravascular coagulation’ occurs in abruptio
placentae and other obstetric conditions, certain malignancies and infections.
The coagulation factors get consumed for the formation of intravascular
microclots and blood is incoagulable. Heparin paradoxically checks bleeding in
such patients by preserving the clotting factors. However, in some cases
heparin may aggravate bleeding.
Related Topics
