Formulation considerations
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Dosage forms - Suspensions
Suspensions are formulated to meet key quality requirements as outlined earlier.
Formulation
considerations
Suspensions
are formulated to meet key quality requirements as outlined earlier. Additional
formulation considerations for suspensions include managing the bitterness and
grittiness of the API, and dose volume. For example, a highly bitter API is
likely to impart an unpleasant taste to the suspension due to its solubility in
the suspension vehicle, even though this solubility may be extremely low. A
gritty particle shape of an API, such as needle-shaped crystals, is likely to
have poor mouthfeel unless the particle size of the suspension is reduced
significantly. In addition, reasonable dose volume for a patient is one
teaspoon (5 mL) or one tablespoon (15 mL) or other nondecimal multiples of
these measures. Total dose volume that may be administered per day is also
limited by the maximum allowed daily dose of other ingredients, such as the
artificial sweetener and the preservative.
Typically,
the following ingredients are used in suspensions.
1. Drug: A water-insoluble
drug is usually the dispersed phase in an
aqueous suspension. Drugs should be of a narrow particle size distri-bution
within the range of 1–50 μm.
2. Wetting agents: The surface of
dispersed drug particles can be either hydrophilic
or hydrophobic. Drugs with hydrophobic surfaces are usually difficult to
disperse in an aqueous medium. Wetting agents are surfactants that reduce the
surface tension of an aqueous medium and facilitate the wetting of hydrophobic
particles. Wetting agents adsorb onto the hydrophobic particle surface to
either partially coat the surface or form a complete monolayer. Examples of
typical wet-ting agents are sodium lauryl sulfate and polysorbate 80.
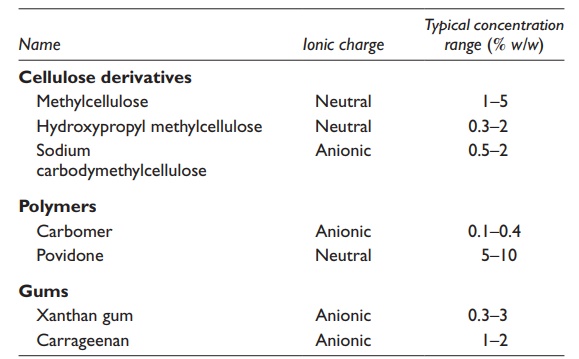
3. Suspending agents: Suspending agents
are hydrophilic colloids, such as
cellulose derivatives, acacia, and xanthan gum that are added to a suspension
to increase viscosity inhibit agglomeration, and decrease sedimentation. Suspending
agents may also interact with the sus-pended particle’s surface to facilitate
wetting and reduce the tendency to agglomerate upon interparticle collisions.
Typical suspending agents are listed in Table 16.1.
Although increasing the viscosity of a suspension improves its physical
stability, on oral administra-tion highly viscous suspensions may prolong
gastric emptying time, slow drug dissolution, and decrease the absorption rate.
Thus, the dose volume of a suspension and in
situ fluid viscosity on dilution of
Table 16.1 Commonly used suspending agents
4. Flocculating
agents:
Suspended particles that have high charge den-sity display deflocculation and
caking upon sedimentation. Such sus-pensions have high particle–particle
electrostatic repulsion and do not settle rapidly. Such suspensions are called
deflocculated because they display uniform distribution of particles without
any settling/ separation or flocculation for extended periods of time. However,
once they settle, they form a strong cake that is difficult to redisperse.
Addition of oppositely charged formulation ingredient(s) to such a suspension
results in partial neutralization of effective charge (i.e., zeta potential) on
the particles through the formation of the electri-cal double layer. This
results in suspended particles being weakly linked together in loose aggregates
or flocs. These flocs settle rap-idly but form large fluffy sediment, which is
easily redispersed. Such suspensions are called flocculated suspensions—and the
formulation ingredients that promote flocculation are called flocculating
agents. Flocculated suspensions are preferred over deflocculated systems to
enable rapid redispersibility upon shaking.
5. Preservatives: Preservatives are
often added in aqueous suspensions because
suspending agents and sweeteners are good media for micro-bial growth. Some
preservatives are ionic, such as sodium benzoate, and
Table 16.2 Commonly used antimicrobial preservatives
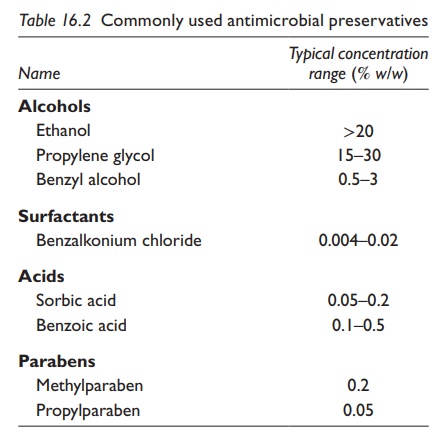
Thus, effective aqueous concen-tration of the
preservative must be monitored and controlled. Solvents, such as alcohols,
glycerin, and propylene glycol, may also have some preservative effect
depending on their concentration. Typical microbial preservatives are listed in
Table 16.2.
6. Sweeteners,
flavors, and colorants: Sweeteners are often added to sus-pensions to reduce any
unpleasant taste of the partially dissolved drug and to improve palatability in
general. Examples include sorbitol, corn syrup, sucrose, saccharin, acesulfame,
and aspartame. Flavors are added to enhance patient’s acceptance of the
product. Colorants are added to provide a more esthetic appearance to the final
product. Choice of col-orant is usually tied to the choice of flavor, and their
choices are also linked to the patient population, such as age group and
geographic region, and the therapeutic need. For example, red colorant is
usually used with strawberry flavor for pediatric formulations.
Table 16.3 shows two examples of suspension formulations.
One is benzoyl peroxide topical suspension, which is used for treating mild to
moderate acne. The other is triamcinolone diacetate parenteral suspension,
which is used for treating allergic disorders.
Table 16.3 Examples of suspension formulations
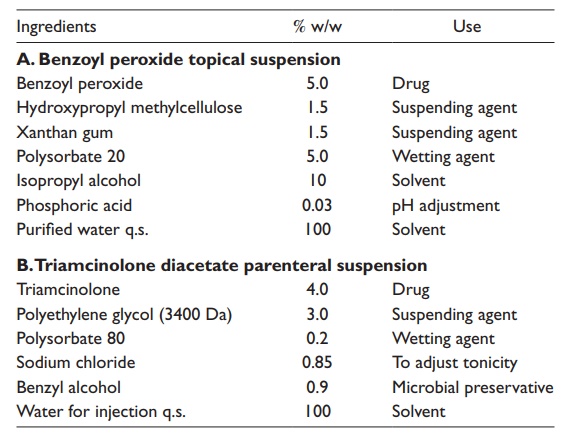
Flocculation
The
large surface area of the suspended fine particles is associated with high
surface-free energy that makes the system thermodynamically unsta-ble.
Generation of fine particles by milling, which is commonly used for
pharmaceutical suspensions, generates particles with higher energy per unit
surface area than the parent, unmilled API crystals. This is attributed to the
preferred orientation of functional groups of a molecule during crys-tallization.
During crystallization, hydrophobic regions of an API, which interact less
favorably with the solvent of crystallization, get embedded on the inside of
the crystal structure, whereas hydrophilic regions are exposed to the surface.
When such a material is milled, the inner hydrophobic sur-face gets exposed.
This is a high-energy surface in an aqueous environ-ment because it resists
interaction with water and has the propensity to self-aggregate to decrease the
total surface area and surface-free energy.
Stabilization
of high-energy fine particles can be accomplished through the use of suspending
agents that increase solution viscosity, reduce surface tension, and/or coat
the surface of the dispersed particles. In addition, the use of formulation
ingredients, such as hydrophilic polymers, that facilitate the formation of
lose associations of dispersed particles through the forma-tion of relatively
weak bonds with each other, can contribute to a phenom-enon called
flocculation.
Flocculation is the formation of
loose, light, and fluffy flocs (associa-tions of particles) held together by
weak van der Waals forces. In contrast, particles in deflocculated suspensions
tend to exhibit strong interparticle
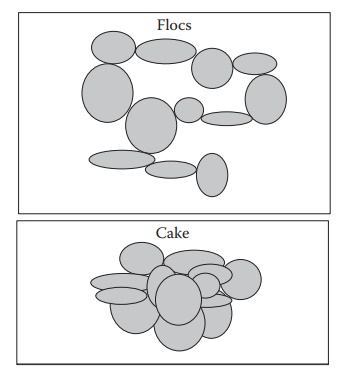
Figure 16.1 Formation of flocs and cake in pharmaceutical suspensions. Suspensions
often form loose networks of flocs that settle rapidly, do not form cakes, and
are easy to suspend. However, settling and aggregation may result in the
formation of cakes that is difficult to resuspend.
Aggregation
occurs in a compact cake situation, that is, growth and fusing together of
crystals in the pre-cipitates to form a solid cake. Figure
16.1 illustrates the difference between flocs and cake in pharmaceutical
suspensions.
Forces
at the surface of the dispersed particles affect the degree of floc-culation
and agglomeration in a suspension. Generally, the forces of attrac-tion are of
the van der Waals type, whereas the repulsive forces arise from the interaction
of the electric double layers surrounding each particle (Figure 16.2). When the repulsion energy is high,
collision of the particles is opposed. The system remains deflocculated.
However, when sedimen-tation is complete, the particles form a close-packed and
strongly bound structure. Those particles lowest in the sediment are gradually
pressed together by the weight of the ones above. The repulsive energy barrier
is thus overcome, allowing the particles to come into close contact with each
other. The reduced bonding potential energy at a critical interparticulate
distance allows the forces of attraction to dominate and caking to occur (Figure 16.3).
To
resuspend and redisperse caked particles in a suspension, it is neces-sary to
overcome the high-energy barrier. Since this is not easily achieved by
agitation, the particles tend to remain strongly attracted to each other and
form a hard cake. When the particles are flocculated, the particles equilibrate
in the second energy minimum, which is at a distance of separa-tion of
~1000–2000 Å—sufficient to form the loosely structural flocs.
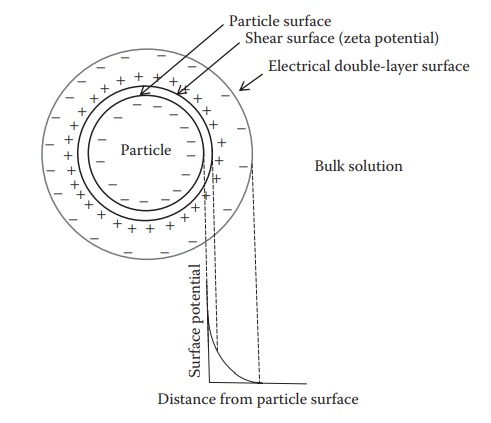
Figure 16.2 Illustration of particle surface charge and zeta potential on the
surface of a particle. Interparticle interactions in a suspension are
determined by the zeta poten-tial, the net charge at the end of an electrical
double layer on the particle surface. This electrical double layer is formed by
the selective adsorption of oppositely charged ionic species in solution to an
electrostatically charged particle surface.
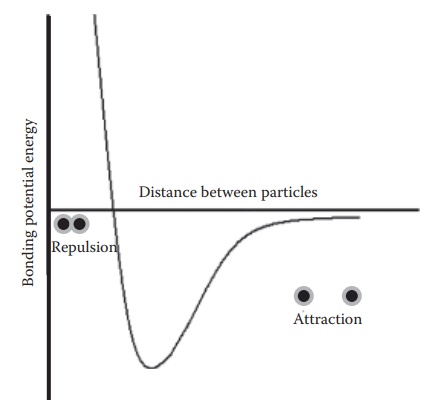
Figure 16.3 Bonding potential energy between particles as a function of distance, in the absence of surface charge. The forces of attraction between particles are dependent on the distance between the particles and are maximized at an optimum distance. Caking in a suspension is facilitated if interparticulate distance allows the forces of attraction to dominate and form strong bonds.
Caking
is undesirable, since a caked dispersed phase is difficult to redisperse.
Flocculating agents can prevent caking, whereas deflocculat-ing agents increase
the tendency to cake. To convert a suspension from a deflocculated to a
flocculated state, the following flocculating agents are often used:
1. Electrolytes: Electrolytes act
as flocculating agents by reducing the electric
barrier between the particles. The addition of an inorganic electrolyte to an
aqueous suspension alters the zeta potential of the dispersed particles. Flocculation
occurs when the zeta potential is lowered sufficiently. The most widely used
electrolytes include sodium salts of acetate, phosphate, and citrate.
2. Surfactants: Ionic surfactants
may also cause flocculation by neutral-ization of the charge on each particle.
An example of ionic surfactant is sodium lauryl sulfate.
3. Hydrophilic
polymers:
Particles coated with hydrophilic polymers are less prone to caking than uncoated particles. Especially for
particles that lack strong electrostatic surface charge, using nonionic
hydro-philic polymers, which act as protective colloids, can contribute to
flocculation. These polymers exhibit pseudoplastic (i.e., shear-thinning,
viscosity reduces upon exposure to shear) flow in solution. This prop-erty
serves to promote physical stability within the suspension by maintaining high
viscosity when the suspension is stagnant, whereas allowing easy pourability by
reduction of viscosity when the suspen-sion is mixed.
Starch,
alginates, cellulose polymers (sodium carboxymethylcellulose), gum
(tragacanth), carbomers, and silicates are examples of polymeric flocculat-ing
agents. Their linear branched chain molecules form a gel-like network within
the system, thus increasing the viscosity of the aqueous vehicle, and become adsorbed
on the surfaces of the dispersed particles, thus acting as protective colloids.
Whether
a suspension is flocculated or deflocculated depends on the relative magnitudes
of the electrostatic forces of repulsion and the forces of attraction between the
particles. When zeta potential is
relatively high, the repulsive forces usually exceed the attractive forces.
Consequently, dis-persed particles remain as discrete units and settle slowly.
The suspension is deflocculated. The slow rate of settling prevents the
entrapment of liquid within the sediment, which thus becomes compacted and can
be very dif-ficult to redisperse. This phenomenon is called caking.
Flocculated
systems form lose sediments, which are easily redispersible. However, the
sedimentation rate is faster than deflocculated suspensions. Association of
particles with each other in a flocculated system leads to a rapid rate of
sedimentation because each unit is composed of many indi-vidual particles and
is, therefore, larger. Supernatant of a deflocculated
Table 16.4 Properties of flocculated and deflocculated suspensions
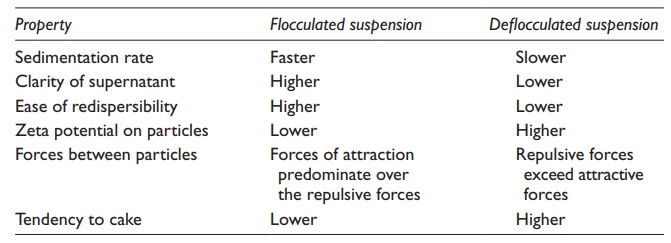
In con-trast, the
supernatant of a flocculated system quickly becomes clear as the flocs,
composed of lose agglomerates of particles of all sizes, settle rap-idly. If
the sedimentation rate is too fast, the dispensed dose may not be accurate.
Therefore, an optimum suspension formulation should only be partially
flocculated. In addition, viscosity is controlled so that the sedi-mentation
rate is minimized. Controlled flocculation is usually achieved by a combination
of particle size control, the use of electrolytes to control zeta potential,
and the addition of polymers to enable the formation of weak networks in
solution that entangle and form weak bonds between the dispersed particles.
Differences between flocculated and deflocculated suspensions are summarized in
Table 16.4.
Quantitating the degree of flocculation
Sedimentation
studies can quantitatively define the sedimentation volume and degree of
flocculation/deflocculation of a system. As illustrated in Figure 16.4, the
sedimentation volume, F, is defined as the ratio of the final volume of the sediment, Vu, to the original volume of
the suspension, V0 before
settling. Thus,
F = Vu/V0
The
sedimentation volume can have values from less than 1 (particle set-tling) to
greater than 1 (particle swelling). It is usually less than 1. That is, the
final volume of sediment is smaller than the original volume of suspen-sion.
Particle swelling can occur for a freshly prepared suspension that has not been
allowed enough time to equilibrate to fully hydrate all solid com-ponents. If
the volume of sediment in a flocculated suspension is equal to the original
volume of suspension, then F = 1.
Such a product is believed to be in flocculation equilibrium and shows no clear
supernatant on standing.
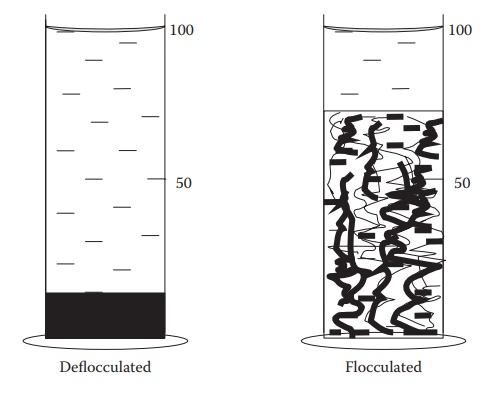
Figure 16.4 A schematic illustrating the differences in the sediment quality and
volume between a deflocculated and a flocculated suspension. On placing
undisturbed in a measuring cylinder, an equal volume of a flocculated
suspension forms a larger mass of a more porous sediment than a deflocculated
suspension.
Physics of particle sedimentation: Stokes’ law
The
control of sedimentation rate of dispersed particles is required to ensure
uniform dosing of a pharmaceutical system. Sedimentation of a disperse system
depends on the motion of the particles, which may be thermally or
gravitationally induced. If a suspended particle is sufficiently small in size,
random Brownian motion dominates over unidirectional gravitational pull. When
the radius of the suspended particles is increased, Brownian motion becomes
less important and sedimentation becomes dominant. These larger particles,
therefore, settle gradually under gravitational forces. Stokes’ law describes
the sedimentation of suspended particles in suspensions:
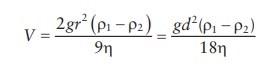
where:
V is the velocity of
sedimentation
r is the particle
radius
d is the particle
diameter
ρ1 and ρ2 are the densities of the particles
and dispersion medium, respectively
g is the acceleration
of gravity
η is the viscosity of the medium
As
the diameter is squared in Stokes’ law, a reduction in particle size by ½ will
reduce the sedimentation rate by (½)2 or a factor of 4. Thus,
particle size control is an important element in the formulation of stable
suspensions.
In
addition, doubling the viscosity of a suspension will decrease the
sedimentation rate by a factor of 2. Increasing the viscosity of a suspen-sion
reduces the rate of settling of dispersed particles, changes the flow
properties of a suspension, and affects the spreading qualities of a lotion.
Viscosity can be increased by the addition of hydrophilic polymers or gums that
act as suspending agents. An ideal suspending agent should have a high
viscosity at negligible shear and should be free flowing during agitation,
pouring, and spreading. A suspending agent that is thixotropic as well as
pseudoplastic should prove to be useful since it forms a gel on standing and
becomes fluid when distributed.
Stokes’
law further indicates that if the difference in density between the suspended
particle and the suspension medium can be matched, the sedimentation rate would
be reduced to zero. Density of a drug particle is an inherent property of the
crystal structure and packing, and may not be altered readily. However, the
density of the suspension medium can be increased by increasing the solids
content of the liquid or solution. This, however, needs to be balanced with an
increase in solution viscosity and reduction of pourability.
