Italy
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: Other Databases in Europe for the Analytic Evaluation of Drug Effects
Similar to the Netherlands and the United Kingdom, GPs in Italy act as gatekeeper to second line health care and provide most of the medical care. Contrary to the Netherlands, however, feedback from specialists or hospitals to GPs is organized through the patient.
ITALY
HEALTH CARE
Similar
to the Netherlands and the United Kingdom, GPs in Italy act as gatekeeper to
second line health care and provide most of the medical care. Contrary to the
Netherlands, however, feedback from specialists or hospitals to GPs is
organized through the patient. In 2000, a GP database called the Health Search
database (HSD) was set up by the Italian College of General Practitioners. In
2004, the database was sold to the market research company Segedim that also
owns THIN and THALES, but research can still be conducted.
Italy
is rather unique in having a specific paedi-atric primary care system for
children between 0 and 14 years of age. A minority of children are cared for by
family physicians (mostly those 10 years and older), whereas there are almost
7000 paediatricians through-out the country associated with the NHS that gives
a flat fee for service per registered child to the paedi-atrician. Inscription
in the National Health system is compulsory for residents; thus, every child at
birth is referred to as a paediatrician associated with the NHS. All
consultations, prescriptions and examinations that are prescribed by the
paediatrician are free of charge to the patient; thus, there are no economic
constraints to attend medical care (Fornaro et
al., 1999). This unique feature of Italian health care has resulted in the
initiation of the PEDIANET database in 1999.
As
part of its tax funded health care for resi-dents, the Italian NHS provides
medical attendance and prescribed medicines. Typically, new and more expensive
drugs are preferred in Italy even when effective, safe and less expensive
alternatives are available. Prescribers had to face a highly dynamic
pharmaceutical market in which 30% of substances (among the 300 most sold)
changed every 5 years before the Drug Reform Act. After the Drug Reform Act in
1994, the reimbursement status of drugs is cate-gorized in three groups: class
A drugs are reimbursed completely, class B drugs require a small patient fee
dependent on age and exemption status and class C drugs are not reimbursed at
all (Rolle et al., 1995). An example
of class C drugs is sedatives.
Health
care is organized by regional and local health agencies (USSL) that use the
local and regional health care information systems (SISR) for planning of
resource utilization. Historically, the most impor-tant source for
pharmacoepidemiological studies in Italy have been the SISR that accumulate
data on all births, deaths, claims of dispensed drugs, hospi-talizations and
procedures that are reimbursed by the NHS through their local health units
(Caffari and Raschetti, 1991; Menniti-Ippolito et al., 1998; Degli Esposti et
al., 1999). The SISR of the Friuli-Venezia-Giulia (FVG) region in the
north-east of Italy has been used most frequently for the conduct of
pharmacoepidemiological research (Rossi et
al., 1991; Simon et al., 1994;
Garcìa-Rodriguez et al., 1998;
Castellsague et al., 1999).
Access
to regional SISR databases and especially original hospital records has become
more compli-cated with the European privacy legislations for exter-nal
organizations. Access to original hospital records for validation purposes is
not possible anymore. Local databases are more and more being used for the
conduct of epidemiological studies; an example is the database in Ravenna that
is commercially exploited (Degli Esposti et
al., 1999). The future will learn whether access to (combined) local
databases through key persons may be easier than formal access to regional
databases.
PEDIANET
Since
1999, the Società Servizi Telematici (So.Se.Te.) based in Padova is developing
a national database, called PEDIANET, which currently collects the clinical,
demographic and prescription data for approximately 180 000 children that have
provided informed consent and who are under the care of any of the 105 primary
care paediatricians (GP) that currently provide data to the database.
Data
are generated during routine patient care with the software JB 95® and are
stored in different files, which can be linked through a unique (anonymous)
numerical identifier. The identification file contains information on the
demographic data of the child and the eligibility status (registration status,
date of registration and date of death). The prescription file contains
information on all drugs (date of prescription, ATC code, product, quantity,
dosing regimen, legend duration, indication and reimbursement status) and
vaccinations that are prescribed by the paediatricians. Reasons for contact and
diagnoses (free text or coded by the ICD-9 system) are collected in the medical
file. In addition, the database contains information on referrals to
specialists, procedures, hospitalizations, medical examinations, health status
(according to the Guidelines of Health Supervision of the American Academy of
Paediatrics) and centile diagrams.
The
database is suitable for both retrospective inspection of routinely collected
data and for prospec-tive data collection (outcomes and indirect costs of
disease) (Menniti-Ippolito et al.,
2000; Nicolosi et al., 2003; Sturkenboom
et al., 2005a). Data access is
possible after approval of the protocol by the Scien-tific Board.
HEALTH SEARCH DATABASE
The
HSD was set up by the Italian College of General Practitioners in 1998
(Cricelli et al., 2003) and is a
longitudinal observational database that contains data from computer-based
patient records of a selected group of GPs throughout Italy, who voluntarily
chose to supply data to the database and to follow courses on data
entry/collection. The HSD currently contains information from over 561 GPs who
cover a total source population of about 800 000 persons since 2001. Each GP
undergoes formal training for data entry and use standard software to record
data. Data are subject to a range of quality checks. Any vari-ations within agreed
ranges are investigated and submitted to each participating GP. Physicians who
fail to meet standard quality criteria are not consid-ered for epidemiological
studies. Currently, 320 GPs with a representative population (around 500 000)
are judged up to a standard, and their data can be used for scientific
research.
The
database contains identification information (age, sex, patient identification
and GP registration information), which is linked to prescription informa-tion,
clinical events and diagnoses, hospital admis-sion and causes of death. All
diagnoses are coded according to the ICD-9-CM. Drug names are coded according
to the ATC classification.
Studies
on disease prevalence and incidence as well as drug utilization and adherence
have been conducted (Cricelli et al.,
2003; Filippi et al., 2003a,b;
2004a,b; 2005a–c; Mazzaglia et al.,
2003; Sturkenboom et al., 2005b;
Galatti et al., 2006).
GENERAL CONSIDERATIONS
The
use of automated data for the conduct of obser-vational epidemiological
research has been heavily discussed in the past and will continue to be
discussed even more so if researchers are using the same or simi-lar data. A
good example are the conflicting results of two studies on the risk of venous
thromboembolism in women using third-generation oral contraceptives that were
both conducted in the GPRD (Farmer et al.,
2000; Kaye et al., 2000). Despite the
controversies that may arise between persons with conflicting inter-est
(researchers, producers, prescribers and patients), there is no doubt that the
use of automated linkage or GP data has proven its value in
pharmacoepidemi-ology. The exploitation of the GPRD by the MHRA, the
sublicensing of the GPRD to different research groups and companies and the
interest of various regulatory agencies such as the EMEA in the use of
automated databases clearly demonstrate the need for longitudinal medical
databases to anticipate, evaluate and assess the use, the cost, the positive
and adverse effects of drugs.
Large
automated databases have given us the opportunity to study the rare and common
effects of (in)frequently used drugs. Good examples are the studies conducted
with the PDNJ database on the teratogenic effects of specific drugs (Nielsen et al., 1999, 2001; Sorensen et al., 1999, 2000; Thul-strup et al., 1999; Fonager et al., 2000; Larsen et al., 2000). Owing to the tax funded
health care struc-tures in many European countries, it is possible to conduct
population-based studies that do not suffer from potential socio-economic
selection biases that may occur with the health maintenance organization
databases in the United States. In addition, the longitu-dinal prospective
collection of routine care data elim-inates recall errors that have plagued so
many ad hoc case–control studies in the past.
Challenges
remain the validation of outcomes, misclassification of exposure and the
adequate control of confounding by indication, severity and contra-indication.
The extent of these potential problems depends on the type of database. Record
linkage databases such as PHARMO, FVG, OPED, PDNJ and also MEMO usually contain
only data on hospitaliza-tions and (reimbursed) drug use. Important
confound-ing factors such as the indication of drug use, body mass index (BMI),
smoking, family history and minor medical problems cannot easily be assessed
and adjusted for. The validation of outcomes with original charts has become
more difficult due to the current privacy regulations.
General
practitioners databases like IPCI, Health Search and PEDIANET and more famously
GPRD and Mediplus have fewer disadvantages than record linkage systems due to
both the nature of the data and the fact that data are collected directly from
the individual health care provider. The latter simpli-fies not only the access
to original data but also the inclusion of project-specific modules in the
soft-ware. In these databases, it would be possible to conduct randomized
database studies as was recently attempted in the IPCI database (Mosis et al., 2005a,b). Randomized database
studies may bridge the gap between randomized trials and observational studies
and deal with confounding by indication.
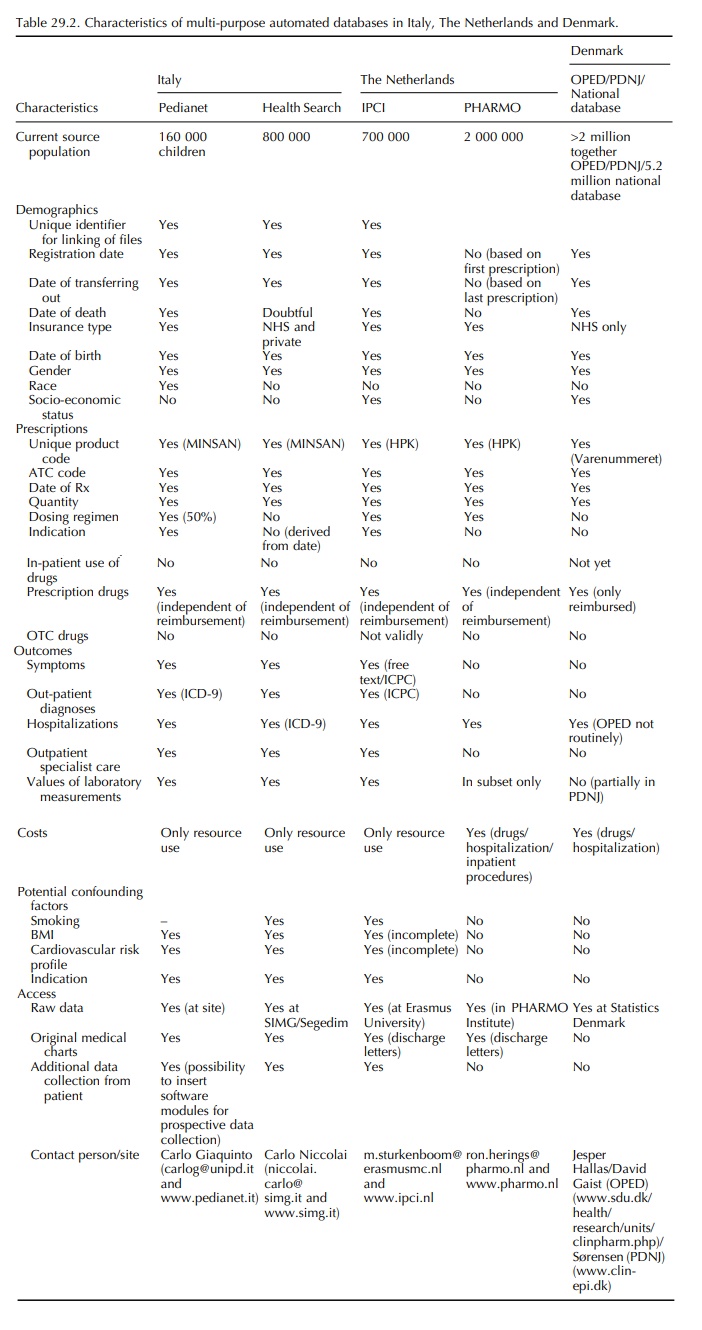
The unification of Europe and the increased computerization of health care are promising future perspectives. Initiatives have been taken to further link hospitalization and pharmacy claims data to GP records, and we may soon expect databases also in other countries (Sweden and Spain). Now that we move from data scarcity to an era of data abundance, it will be possible to choose a database that is tailored for the research question at hand. Table 29.2 may offer an aid in comparing the available databases. As researchers, we may want to unite forces. Effort should be put on the organization of multi-national database stud-ies that have advantages in size but also in variability of drug use, allowing for the full evaluation of drug-and dose-specific risks and comparisons between coun-tries. One such attempt is the pharmacoepidemiological studies as part of the TEDDY project, an EU network of excellence aiming at drug development for the young (www.teddynoe.org). In this network, GPRD, Mediplus, IPCI and PEDIANET data are combined to assess drug safety issues in children.
Related Topics
