Non-Newtonian flow
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Rheology
Most pharmaceutical fluids, such as colloidal dispersions, emulsions, liquid suspensions, and ointments, do not follow Newton’s law of flow.
Non-Newtonian flow
Most
pharmaceutical fluids, such as colloidal dispersions, emulsions, liquid
suspensions, and ointments, do not follow Newton’s law of flow. The vis-cosity
of the fluid varies with the rate of shear. Depending on how viscosity changes
with shear, there are three general types of non-Newtonian flow behaviors:
plastic, pseudoplastic, and dilatant (Figure 12.1b–d).
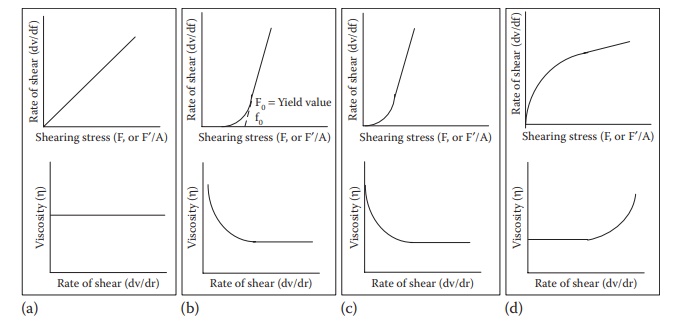
Figure 12.1 Plots of rate of shear and viscosity as a function of shearing stress for (a) Newtonian, (b) plastic, (c) pseudoplastic, and (d) dilatant flows.
Plastic flow
Substances
that undergo plastic flow are called Bingham bodies; they are defined as
substances that exhibit a yield value as the point at which plas-tic flow curve
intersects shearing stress axis (Figure 12.1b).
Plastic flow is associated with, for example, the presence of flocculated
particles in con-centrated suspensions. Flocculated solids are light, fluffy
conglomerates of adjacent particles held together by weak van der Waals forces.
A certain shearing stress must be exceeded in order to break up van der Waals
forces, which is the yield value, f0. The yield value is an
indicator of flocculation. Higher the yield value, greater the degree of
flocculation.
A
plastic system resembles a Newtonian system at shear stresses below the yield
value. The characteristics of plastic flow can be summarized as follows:
·
Plastic flow does not begin until a shearing stress,
corresponding to a yield value, f, is exceeded.
·
The curve intersects the shearing stress axis but does not
cross through the origin.
·
The materials are said to be elastic at shear stresses below the yield value.
Pseudoplastic (shear-thinning) flow
Pseudoplastic
flow is characterized by decrease in viscosity with increas-ing shear stress.
This leads to increasing rate of shear (flow) for the same amount of change in
the shear stress as the shear stress levels are increased. Thus, these fluids
tend to flow more easily with increasing shear stress and flow and are thus
called shear-thinning fluids. The
molecular origin of the shear-thinning behavior of fluids is in preferential
alignment of the mol-ecules of solution during flow, such that intermolecular
cohesion forces are reduced as the rate of shear and the rate of flow increase.
Thus, shear thin-ning occurs when molecules align themselves during flow, such
that they slip and slide past each other.
Linear
polymers in solution exhibit pseudoplastic flow. A large number of
pharmaceutical products, including natural and synthetic gums (e.g., liquid
dispersions of tragacanth, sodium alginate, methyl cellulose, and sodium
carboxymethylcellulose), exhibit pseudoplastic flow properties.
The
characteristics of pseudoplastic flow materials can be summarized as follows:
·
Pseudoplastic substances begin to flow when a shearing
stress is applied; therefore, they exhibit no, or very low, yield value. Thus,
the shear stress—shear rate profile does cross the origin (Figure 12.1c).
·
Viscosity of a pseudoplastic substance decreases with
increasing shear stress and shear rate.
Dilatant (shear-thickening) flow
Dilatant
flow is characterized by an increase in viscosity with increasing shear stress.
This leads to decreasing rate of shear (i.e., reduced flow and higher
viscosity) for the same amount of change in the shear stress as the shear
stress levels are increased. Thus, these fluids tend to get more viscous and thicker and flow with greater difficulty
with increasing shear stress and flow; they are thus called shear-thickening fluids. The molecular
origin of the shear-thickening behavior of fluids is in entanglement with
increasing attractive intermolecular interactions among the molecules of
solution dur-ing flow, such that the intermolecular cohesion forces increase as
the rate of shear and the rate of flow increase. Thus, shear thickening occurs
when mol-ecules get entangled, swell, or otherwise align themselves during
flow, such that the intermolecular forces of attraction are higher as the flow
increases.
Generally,
dilatant solutions are those that exhibit an increase in solute volume when
sheared. This leads to the reduction in free,
or unbound, solvent volume between the solute molecules. Thus, the viscosity
increases with shear rate. Dilatant systems are usually suspensions with a high
per-centage (≥ 50% w/w) of dispersed small, deflocculated particles. These
sys-tems exhibit an increase in resistance to flow with increasing rates of
shear.
The
characteristics of dilatant flow materials can be summarized as follows:
·
Solutes in dilatant solutions increase in volume when
sheared. When the stress is removed, the dilatant system returns to its
original state of fluidity.
·
Viscosity increases with increasing shear rate.
In
concentrated suspensions, particles are closely packed, with the inter-particle
void volume being at a minimum at rest. Nevertheless, the amount of vehicle in
the suspension is sufficient to fill this volume and to allow the particles to
move relative to one another at low rates of shear. Dilatant sus-pensions can
be poured from a bottle, since these are reasonably fluid under these
conditions. The bulk of the system dilates (expands) with increase in shear
stress. The particles in a deflocculated suspension take an open form of
packing; that is, each particle is detached from every other particle,
occu-pying its own space and interacting with solvent molecules to its full
surface. Thus, the solvent-filled interparticle void volume is lower for
deflocculated suspensions. Accordingly, resistance to flow increases with an
increase in flow, because the particles no longer completely get wetted or
lubricated by the vehicle. Eventually, the suspension will set up as a firm
paste.
The
Newton’s flow equation described earlier, t
= η * D, where, D is the rate of flow
and t is the applied stress, can be
modified to:
t = η* D*eN
For
Newtonian fluids, N = 0 and the
expression eN = 1, thus
yielding the expression for Newtonian flow. For dilatant or shear-thickening
fluids, 0 < N < 1. As dilatancy
increases, the shear rate response to shear stress decreases exponentially;
thus, N decreases as dilatancy
increases for shear-thickening fluids.
Related Topics
