SAR of Thiazide Diruretics
| Home | | Medicinal Chemistry |Chapter: Medicinal Chemistry : Diuretics
The 2nd position can tolerate the presence of small alkyl groups such as CH3.
SAR of Thiazide Diruretics The 2nd position can tolerate the presence of small alkyl groups such as CH3. Substituents with hydrophobic character in the 3rd position increases saluretic activity 1000 times. Substituents include –CH2Cl, –CHCl2, –CH2C6H5, –CH2S, –CH2 –C6H5. The increase in saluretic activity correlates with the lipid solubility. Saturation of double bond between the 3rd and 4th position of nucleus increases the diuretic activity approximately 3 fold to 10-fold. Example— Hydrochlorthiazide. Hydrogen atom at the 2nd position is more acidic due to the presence of neighbouring electron with-drawing the sulphone group. A free sulphamoyl or potentially free sulphamoyl group at 7th postion is essential for activity. N7-caproyl chlorthiazide is excreted as chorothiazide, the loss of sulphamoyl group eliminates the diuretic effect, but not the antihypertensive action, example, diazoxide. Direct substitution of the 4th, 5th, or 8th position with an ethyl group usually results in diminished diuretic activity. Substitution of the 6th position with an activating group is essential for diuretic activity. The substiutents include Cl, Br, and CF3 groups. The acidic protons make positive the formation of water-soluble sodium salt that can be used for intravenous administration of the diuretics.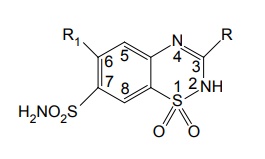
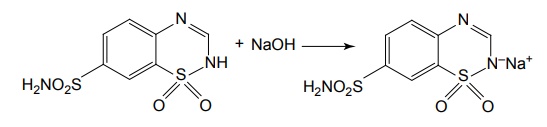
Related Topics
