Absorption of Drugs From Non-PER OS Extravascular Routes
| Home | | Biopharmaceutics and Pharmacokinetics |Chapter: Biopharmaceutics and Pharmacokinetics : Absorption of Drugs
Drug absorption from all non-oral extravascular sites is governed by the same factors that influence absorption from GIT viz. the physicochemical properties of drug, formulation factors, and anatomic, physiologic and pathologic characteristics of the patient.
ABSORPTION OF DRUGS FROM NON-PER OS EXTRAVASCULAR ROUTES
Drug absorption from all non-oral extravascular sites is governed by the same factors that influence absorption from GIT viz. the physicochemical properties of drug, formulation factors, and anatomic, physiologic and pathologic characteristics of the patient. This is so because the barrier to transport of drugs into the systemic circulation from all such sites is a lipoidal membrane similar to the GI barrier and the major mechanism in the absorption is passive diffusion. One of the major advantages of administering drugs by non-invasive transmucosal routes such as nasal, buccal, rectal, etc. is that greater systemic availability is attainable for drugs normally subjected to extensive presystemic elimination due to GI degradation and/or hepatic metabolism (Fig. 2.36). Moreover, peptide and protein drugs can also be delivered by such routes. Some of the more important biopharmaceutic and pharmacokinetic principles that must be considered for non-oral absorption will be discussed here.

Fig. 2.36. Various transmucosal non-invasive routes of drug administration to bypass presystemic elimination in GIT/liver
Buccal/Sublingual Administration
The two sites for oral mucosal delivery of drugs are:
1. Sublingual route: The drug is placed under the tongue and allowed to dissolve.
2. Buccal route: The medicament is placed between the cheek and the gum.
The barrier to drug absorption from these routes is the epithelium of oral mucosa. Passive diffusion is the major mechanism for absorption of most drugs; nutrients may be absorbed by carrier-mediated processes.
Some of the advantages of these routes are:
1. Rapid absorption and higher blood levels due to high vascularisation of the region and therefore particularly useful for administration of antianginal drugs.
2. No first-pass hepatic metabolism.
3. No degradation of drugs such as that encountered in the GIT
4. Presence of saliva facilitates both drug dissolution and its subsequent permeation by keeping the oral mucosa moist.
Notable factors to be considered in the oral mucosal delivery of drugs are:
1. Lipophilicity of drug: Slightly higher lipid solubility than that required for GI absorption is necessary for passive permeation.
2. Salivary secretion: In addition to high lipid solubility, the drug should be soluble in aqueous buccal fluids i.e. biphasic solubility of drug is necessary for absorption; absorption is delayed if the mouth is dry.
3. pH of the saliva: Usually around 6, the buccal pH favours absorption of drugs which remain unionised.
4. Binding to oral mucosa: Systemic availability of drugs that bind to oral mucosa is poor.
5. Storage compartment: For some drugs such as buprenorphine, a storage compartment in the buccal mucosa appears to exist which is responsible for the slow absorption of drugs.
6. Thickness of oral epithelium: Sublingual absorption is faster than buccal since the epithelium of former region is thinner and immersed in a larger volume of saliva.
Factors that limit drug administration by these routes are: limited mucosal surface area (thus only a small dose can be administered), concern for taste of the medicament and discomfort (since the region is highly innervated).
Examples of drugs administered by oral mucosal route include antianginals like nitrites and nitrates, antihypertensives like nifedipine, analgesics like morphine and bronchodilators like fenoterol. Certain steroids like oestradiol and peptides like oxytocin can also be administered. Apart from tablets, the drugs may be administered as a buccal spray especially to children. This newer approach to drug absorption from the oral cavity is called as translingual delivery e.g. nitroglycerin spray.
Rectal Administration
Despite its diminished popularity, the rectal route of drug administration is still an important route for children and old patients. The drugs may be administered as solutions (microenemas) or suppositories. Absorption is more rapid from solutions than from suppositories but is more variable in comparison to oral route. Irritating suppository bases such as PEG promotes defecation and drug loss. Presence of faecal matter retards drug absorption. Though highly vascularised, absorption is slower because of limited surface area. The pH of rectal fluids (around 8) also influences drug absorption according to pH-partition hypothesis. Absorption of drugs from the lower half of rectum bypasses presystemic hepatic metabolism. Drugs administered by this route include aspirin, paracetamol, theophylline, few barbiturates, etc.
Topical Administration
Excluding the respiratory tract’s contact with the inhaled air, the skin is virtually the sole human surface directly interfacing the body with the external environment. It is the largest organ of the body weighing approximately 2 Kg and 2 m2 in area and receives about 1/3rd of total blood circulating through the body. Though tolerant to many chemicals, topically contacted xenobiotics can evoke both local and systemic effects. Majority of drugs applied topically are meant to exert their effect locally. When topically applied drugs are meant to exert their effects systemically, the mode of administration is called as percutaneous or transdermal delivery. Percutaneous absorption occurs only if the topically applied drug permeates the dermal capillaries and enters the blood stream.
Anatomically, the skin is made of 3 distinct layers—the epidermis, the dermis and the subcutaneous fat tissue. Epidermis is the nonvascular, multilayered outer region of the skin. The dermis or true skin is a highly vascular region; drugs permeating to this region are taken up into the systemic circulation and sink conditions are maintained.
The principal barrier to the entry of xenobiotics is the most superficial layer of epidermis called as stratum corneum. It is composed of dead, keratinised, metabolically inactive horny cells that act as the major rate-limiting barrier to passive diffusion of drugs. In order to act either locally or systemically, a topically applied drug may diffuse through the skin by hair follicles, sweat glands or sebaceous glands but permeation through the multiple lipid bilayers of stratum corneum is the dominant pathway though the rate is very slow. Several factors influence passive percutaneous absorption of drugs:
1. Thickness of stratum corneum: absorption is very slow from regions such as foot and palm where the skin has thickened stratum corneum.
2. Presence of hair follicles: absorption is rapid from regions where numerous hair follicles exist e.g. scalp.
3. Trauma: cuts, rashes, inflammation, mild burns or other conditions in which the stratum corneum is destroyed, promote drug absorption.
4. Hydration of skin: soaking the skin in water or occluding it by using emollients, plastic film or dressing, promote hydration of skin and drug absorption.
5. Environment humidity and temperature: higher humidity and temperature increase both the rate of hydration as well as local blood flow and hence drug absorption.
6. Age: gross histological changes take place as the skin ages. Aged skin is more prone to allergic and irritant effects of topically contacted chemicals as a result of hardening of blood vessels. Infants absorb drug through skin as efficiently as adults. Their ratio of surface area to body weight is 3 times that of adults; hence, systemic toxicity of topically applied drugs is of particular concern in infants.
7. Grooming: the frequency and vigour with which one bathes and the type of soap that is used also contribute to variability in drug absorption.
8. Exposure to chemicals: occupational exposure to solvents can accelerate shedding of epidermal cells and enhance drug absorption.
9. Vehicle or base: the vehicle in which the drug is incorporated influences drug absorption; the one in which the drug is dissolved rather than dispersed promotes absorption.
10. Permeation enhancers: incorporation of certain chemicals such as DMSO, propylene glycol, azone, etc. in the topical formulations aid drug penetration.
11. Chronic use of certain drugs: long term use of cortisol or keratolytics like salicylic acid results in enhanced drug penetration.
Drugs that are administered percutaneously include nitroglycerine, lidocaine, betamethasone, oestradiol, testosterone, etc. The route is particularly useful for drugs with low oral availability and short duration of action; the effect of the latter category of drugs is prolonged because percutaneous absorption is a slow process.
Certain ionic drugs are not absorbed transdermally despite the use of chemical penetration enhancers in the topical formulations. Percutaneous absorption of such drugs can be affected by novel techniques such as –
1. Iontophoresis – Iontophoresis drug delivery implies delivery of ionic drugs into the body by means of an electric current. An ionised drug in solution is placed on the skin and an electrical potential difference established thus driving the ions into the skin. Like electrical charges repel. Therefore, application of a positive current will drive positively charged drug molecules away from the electrode and into the tissues; similarly, a negative current will drive negatively charge ions into the tissues. Cortisol, methacholine, lidocaine, salicylates and peptides and proteins such as insulin have been delivered in this way.
2. Phonophoresis – Phonophoresis is defined as the movement of drug molecules through the skin under the influence of ultrasound.
Intramuscular Administration
Absorption of drugs from i.m. sites is relatively rapid but much slower in comparison to i.v. injections. Factors that determine rate of drug absorption from i.m. sites are:
1. Vascularity of the injection site: the decreasing order of blood flow rate to muscular tissues in which drugs are usually injected is:
Arm (deltoid) > Thigh (vastus lateralis) > Buttocks (gluteus maximus).
Since blood flow rate is often the rate-limiting step in absorption of drugs from i.m. sites, most rapid absorption is from deltoid muscles and slowest from gluteal region. The absorption rate decreases in circulatory disorders such as hypotension.
2. Lipid solubility and ionisation of drug: highly lipophilic drugs are absorbed rapidly by passive diffusion whereas hydrophilic and ionised drugs are slowly absorbed through capillary pores.
3. Molecular size of the drug: small molecules and ions gain direct access into capillaries through pores whereas macromolecules are taken up by the lymphatic system. There is some evidence that small peptides and fluids can cross the endothelial tissue of blood capillaries and lymph vessels by transport in small vesicles that cross the membrane, a process called as cytopemphis.
4. Volume of injection and drug concentration: a drug in concentrated injection and large volume is absorbed faster than when given in dilute form and small volume.
5. pH, composition and viscosity of injection vehicle: a solution of drug in acidic or alkaline pH (e.g. phenytoin, pH 12) or in a nonaqueous solvent such as propylene glycol or alcohol (e.g. digoxin) when injected intramuscularly result in precipitation of drug at the injection site followed by slow and prolonged absorption. Viscous vehicles such as vegetable oils also slow drug absorption. The principle can however be utilized to control rate of drug delivery.
Subcutaneous Administration
All factors that influence i.m. drug absorption are also applicable to absorption from subcutaneous site. Generally, absorption of drugs from a s.c. site is slower than that from i.m. sites due to poor perfusion, but this fact is of particular importance for the administration of drugs for which a rapid response is not desired and for drugs that degrade when taken orally e.g. insulin and sodium heparin. The rate of absorption of a drug from subcutaneous site can be increased in 2 ways:
1. Enhancing blood flow to the injection site: by massage, application of heat, co-administration of vasodilators locally, or by exercise, and
2. Increasing the drug-tissue contact area: by co-administering the enzyme hyaluronidase that breaks down the connective tissue and permits spreading of drug solution over a wide area.
Absorption can be slowed down by causing vasoconstriction through local cooling or co-injection of a vasoconstrictor like adrenaline or by immobilization of limb. Because of relatively slow drug absorption from s.c. tissues, the region is very popular for controlled release medication like implants.
Pulmonary Administration
In principle, all drugs intended for systemic effects can be administered by inhalation since the large surface area of the alveoli, high permeability of the alveolar epithelium and rich perfusion permit extremely rapid absorption just like exchange of gases between the blood and the inspired air. However, the route has been limited for administering drugs that affect pulmonary system such as bronchodilators (salbutamol), anti-inflammatory steroids (beclomethasone) and antiallergics (cromolyn). Lipid soluble drugs are rapidly absorbed by passive diffusion and polar drugs by pore transport. Absorption of drugs whose ionisation is pH sensitive is dependent upon pH of pulmonary fluids. The drugs are generally administered by inhalation either as gases (volatile/gaseous anaesthetics) or aerosol. In the latter case, drug delivery to lungs is largely dependent upon the particle size of the aerosolised droplets—particles larger than 10 microns impact on the mouth, throat or upper respiratory tract mucosa and do not reach the pulmonary tree whereas very small particles (0.6 microns) from which drug absorption is rapid, penetrate rapidly but are susceptible to easy exhalation. Sometimes, the patients’ inability to inhale a sufficient amount of drug limits drug delivery to lungs.
Intranasal Administration
The nasal route is becoming increasingly popular for systemic delivery especially of some peptide and protein drugs. Drug absorption from nasal mucosa is as rapid as observed after parenteral administration because of its rich vasculature and high permeability. The route is otherwise used for drugs to treat local symptoms like nasal congestion, rhinitis, etc.
Two mechanisms for drug transport across the nasal mucosa have been suggested—
· A faster rate that is dependent upon drug lipophilicity, and
· A slower rate which is dependent upon drug molecular weight.
In case of lipophilic drugs, rapid absorption by diffusion is observed up to 400 Daltons and satisfactory absorption up to 1000 Daltons. By use of permeability enhancers such as surfactants, even a drug with molecular weight of 6000 Daltons shows reasonable bioavailability. Peptides up to 10 amino acids appear to be able to penetrate nasal mucosa e.g. vasopressin, oxytocin, calcitonin and buserelin. For polar compounds primarily absorbed by pore transport, an upper threshold of 200 Daltons is the limiting factor. Other factors that may influence nasal permeation of drugs include pH of nasal secretions (5.5 to 6.5) and its viscosity, and pathological conditions such as common cold and rhinitis. Drugs known to influence cleansing function of nasal cilia should not be administered by this route.
Intraocular Administration
Topical application of drugs to the eyes is mainly meant for local effects such as mydriasis, miosis, anaesthesia or treatment of infections, glaucoma, etc. Sterile aqueous solutions of drugs are widely used ophthalmic formulations and administered in the conjunctival cul-de-sac. The barrier to intraocular penetration of drugs is the cornea which possesses both hydrophilic and lipophilic characteristics. Thus, for optimum intraocular permeation, drugs should possess biphasic solubility. The pH of lachrymal fluid influences absorption of weak electrolytes such as pilocarpine. On the other hand, pH of the formulation influences lachrymal output—higher pH decreases tear flow and promotes drug absorption whereas lower pH solutions increase lachrymation and subsequent drug loss due to drainage. Rate of blinking also influences drainage loss. The volume of fluid instilled into the eyes also affects bioavailability and effectiveness of the drug. Normally, the human eye can hold around 10 µl of fluid; hence, instillation of small volume of drug solution in concentrated form increases its effectiveness than when administered in large volume in dilute form. Viscosity imparters in the formulation increase bioavailability by prolonging drug’s contact time with the eye. Oily solutions, ointments and gels show sustained drug action for the same reason. Sometimes systemic absorption of a drug with low therapeutic index such as timolol may precipitate undesirable toxic effects. Systemic entry of drugs occur by way of absorption into lachrymal duct which drains lachrymal fluid into the nasal cavity and finally into the GIT. This can be prevented by simple eyelid closure or naso-lachrymal occlusion by pressing the fingertip to the inside corner of the eye after drug instillation.
Vaginal Administration
Drugs meant for intravaginal application are generally intended to act locally in the treatment of bacterial or fungal infections or prevent conception. The route is now used for systemic delivery of contraceptives and other steroids, without the disadvantage of first-pass metabolism. Controlled delivery and termination of drug action when desired, is possible with this route. Factors that may influence drug absorption from intravaginal site include pH of lumen fluids (4 to 5), vaginal secretions and the microorganisms present in the vaginal lumen which may metabolise the drug.
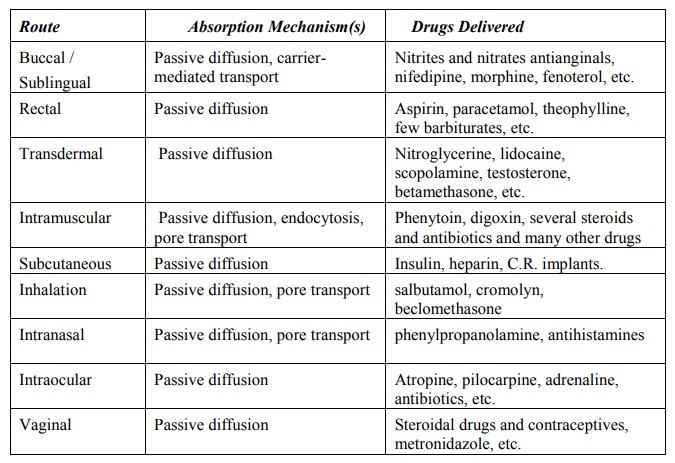
A summary of mechanisms and drugs absorbed from various non-invasive routes (other than the GI) is given in Table 2.10.
TABLE 2.10.
Absorption of Drugs from Non per os Extravascular Routes
Related Topics
