Tissue Permeability of Drugs
| Home | | Biopharmaceutics and Pharmacokinetics |Chapter: Biopharmaceutics and Pharmacokinetics : Distribution of Drugs
Physicochemical Properties of the Drug, Physiological Barriers to Distribution of Drugs
TISSUE PERMEABILITY OF DRUGS
Of the several factors listed above, the two major rate-determining steps in the distribution of drugs are:
1. Rate of tissue permeation, and
2. Rate of blood perfusion.
If the blood flow to the entire body tissues were rapid and uniform, differences in the degree of distribution between tissues will be indicative of differences in the tissue penetrability of the drug and the process will be tissue permeation rate-limited. Tissue permeability of a drug depends upon the physicochemical properties of the drug as well as the physiological barriers that restrict diffusion of drug into tissues.
Physicochemical Properties of the Drug
Important physicochemical properties of drug that influence its distribution are molecular size, degree of ionisation, partition coefficient and stereochemical nature.
Almost all drugs having molecular weight less than 500 to 600 Daltons easily cross the capillary membrane to diffuse into the extracellular interstitial fluids. However, penetration of drugs from the extracellular fluid into the cells is a function of molecular size, ionisation constant and lipophilicity of the drug. Only small, water-soluble molecules and ions of size below 50 Daltons enter the cell through aqueous filled channels whereas those of larger size are restricted unless a specialized transport system exists for them.
The degree of ionisation of a drug is an important determinant in its tissue penetrability. The pH of the blood and the extravascular fluid also play a role in the ionisation and diffusion of drugs into cells. A drug that remains unionised at these pH values can permeate the cells relatively more rapidly. Since the blood and the ECF pH normally remain constant at 7.4, they do not have much of an influence on drug diffusion unless altered in conditions such as systemic acidosis or alkalosis.
Most drugs are either weak acids or weak bases and their degree of ionisation at plasma or ECF pH depends upon their pKa. All drugs that ionise at plasma pH (i.e. polar, hydrophilic drugs), cannot penetrate the lipoidal cell membrane and tissue permeability is the rate-limiting step in the distribution of such drugs. Only unionised drugs which are generally lipophilic, rapidly cross the cell membrane. Among the drugs that have same o/w partition coefficient but differ in the extent of ionisation at blood pH, the one that ionises to a lesser extent will have greater penetrability than that which ionises to a larger extent; for example, pentobarbital and salicylic acid have almost the same Ko/w but the former is more unionised at blood pH and therefore distributes rapidly. The influence of drug pKa and Ko/w on distribution is illustrated by the example that thiopental, a nonpolar, lipophilic drug, largely unionised at plasma pH, readily diffuses into the brain whereas penicillins which are polar, water-soluble and ionised at plasma pH do not cross the blood-brain barrier (Fig. 3.3).
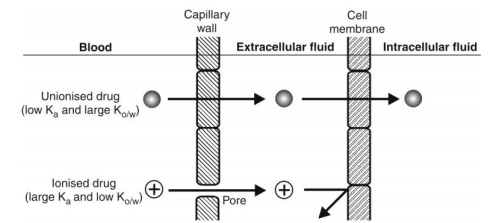
Fig. 3.3. Permeation of unionised and ionised drugs across the capillary and the cell membrane
Since the extent to which a drug exists in unionised form governs the distribution pattern, situations that result in alteration of blood pH affect such a pattern; for example, acidosis (metabolic or respiratory) results in decreased ionisation of acidic drugs and thus increased intracellular drug concentration and pharmacological action. Opposite is the influence of alkalosis. Sodium bicarbonate induced alkalosis is sometimes useful in the treatment of barbiturate (and other acidic drugs) poisoning to drive the drug out and prevent further entry into the CNS and promote their urinary excretion by favouring ionisation. Converse is true for basic drugs; acidosis favours extracellular whereas alkalosis, intracellular distribution.
In case of polar drugs where permeability is the rate-limiting step in the distribution, the driving force is the effective partition coefficient of drug. It is calculated by the following formula:
Effective Ko/w (Fraction unionised at pH 7.4) (Ko/w of unionised drug) (3.1)
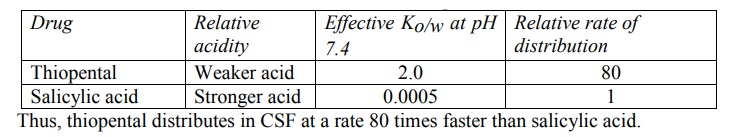
The extent to which the effective partition coefficient influences rapidity of drug distribution can be seen from the example given in Table 3.1.
TABLE 3.1.
Distribution of Acidic Drugs in CSF
Stereochemical nature of drug will also influence the distribution characteristics especially when it has a tendency to interact with macromolecules like proteins. Tissue localisation of certain drugs may be an indication of stereoselectivity in drug distribution.
Physiological Barriers to Distribution of Drugs
A membrane (or a barrier) with special structural features can be a permeability restriction to distribution of drugs to some tissues. Some of the important simple and specialized physiological barriers are:
1. Simple capillary endothelial barrier
2. Simple cell membrane barrier
3. Blood-brain barrier
4. Blood-CSF barrier
5. Blood- placental barrier
6. Blood-testis barrier.
The Simple Capillary Endothelial Barrier: The membrane of capillaries that supply blood to most tissues is, practically speaking, not a barrier to moieties which we call drugs. Thus, all drugs, ionised or unionised, with a molecular size less than 600 Daltons, diffuse through the capillary endothelium and into the interstitial fluid. Only drugs bound to the blood components are restricted because of the large molecular size of the complex.
The Simple Cell Membrane Barrier: Once a drug diffuses from the capillary wall into the extracellular fluid, its further entry into cells of most tissues is limited by its permeability through the membrane that lines such cells. Such a simple cell membrane is similar to the lipoidal barrier in the GI absorption of drugs (discussed in chapter 2).
The physicochemical properties that influence permeation of drugs across such a barrier are summarized in Fig. 3.4.
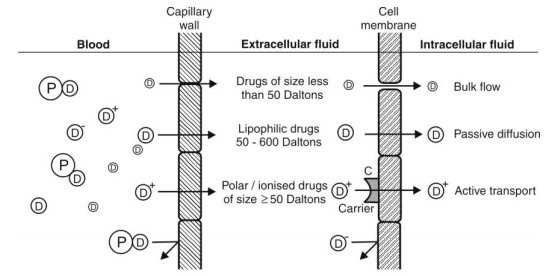
Fig. 3.4. Plasma membrane barrier and drug diffusion across it
Blood-Brain Barrier (BBB): Unlike the capillaries found in other parts of the body, the capillaries in the brain are highly specialized and much less permeable to water-soluble drugs. The brain capillaries consist of endothelial cells which are joined to one another by continuous tight intercellular junctions comprising what is called as the blood-brain barrier (Fig. 3.5). Moreover, the presence of special cells called as pericytes and astrocytes, which are the elements of the supporting tissue found at the base of endothelial membrane, form a solid envelope around the brain capillaries. As a result, the intercellular (paracellular) passage is blocked and for a drug to gain access from the capillary circulation into the brain, it has to pass through the cells (transcellular) rather than between them. (However, there are specific sites in the brain where the BBB does not exist, namely, the trigger area and the median hypothalamic eminence. Moreover, drugs administered intranasally may diffuse directly into the CNS because of the continuity between submucosal areas of the nose and the subarachnoid space of the olfactory lobe). There is also virtual absence of pinocytosis in brain.
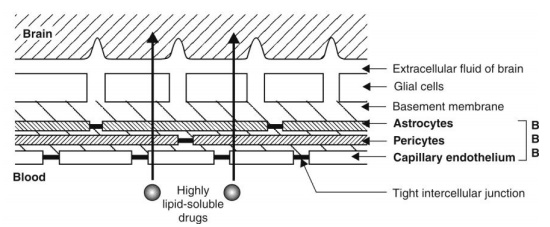
Fig. 3.5. Blood-brain barrier
A solute may thus gain access to brain via only one of two pathways:
1. Passive diffusion through the lipoidal barrier – which is restricted to small molecules (with a molecular weight less than a threshold of approximately 700 Daltons) having high o/w partition coefficient.
2. Active transport of essential nutrients such as sugars and amino acids. Thus, structurally similar foreign molecules can also penetrate the BBB by the same mechanism.
The effective partition coefficient of thiopental, a highly lipid soluble drug is 50 times that of pentobarbital and crosses the BBB much more rapidly. Most antibiotics such as penicillin which are polar, water-soluble and ionised at plasma pH, do not cross the BBB under normal circumstances.
The selective permeability of lipid soluble moieties through the BBB makes appropriate choice of a drug to treat CNS disorders an essential part of therapy; for example, Parkinsonism, a disease characterized by depletion of dopamine in the brain, cannot be treated by administration of dopamine as it does not cross the BBB. Hence, levodopa, which can penetrate the CNS where it is metabolised to dopamine, is used in its treatment. Targeting of polar drugs to brain in certain conditions such as tumour had always been a problem. Three different approaches have been utilized successfully to promote crossing the BBB by drugs:
i. Use of permeation enhancers such as dimethyl sulphoxide (DMSO).
ii. Osmotic disruption of the BBB by infusing internal carotid artery with mannitol.
iii. Use of dihydropyridine redox system as drug carriers to the brain.
In the latter case, the lipid soluble dihydropyridine is linked as a carrier to the polar drug to form a prodrug that readily crosses the BBB. In the brain, the CNS enzymes oxidize the dihydropyridine moiety to the polar pyridinium ion form that cannot diffuse back out of the brain. As a result, the drug gets trapped in the CNS. Such a redox system has been used to deliver steroidal drugs to the brain.
Blood-Cerebrospinal Fluid Barrier: The cerebrospinal fluid (CSF) is formed mainly by the choroid plexus of the lateral, third and fourth ventricles and is similar in composition to the ECF of brain. The capillary endothelium that lines the choroid plexus have open junctions or gaps and drugs can flow freely into the extracellular space between the capillary wall and the choroidal cells. However, the choroidal cells are joined to each other by tight junctions forming the blood-CSF barrier which has permeability characteristics similar to that of the BBB (Fig. 3.6).
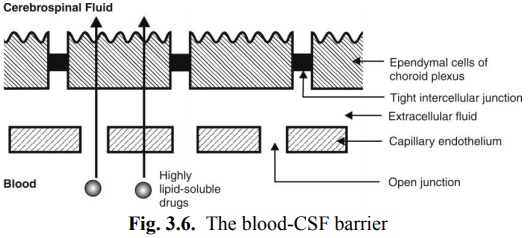
As in the case of BBB, only highly lipid soluble drugs can cross the blood-CSF barrier with relative ease whereas moderately lipid soluble and partially ionised drugs permeate slowly. A drug that enters the CSF slowly cannot achieve a high concentration as the bulk flow of CSF continuously removes the drug. For any given drug, its concentration in the brain will always be higher than in the CSF.
Although the mechanisms for diffusion of drugs into the CNS and CSF are similar, the degree of uptake may vary significantly. In some cases, CSF drug concentration may be higher than its cerebral concentration e.g. sulphamethoxazole and trimethoprim, and vice versa in other cases, e.g. certain β-blockers.
Blood-Placental Barrier: The maternal and the foetal blood vessels are separated by a number of tissue layers made of foetal trophoblast basement membrane and the endothelium which together constitute the placental barrier. The flow of blood in the maternal and the foetal blood vessels is shown in Fig. 3.7.
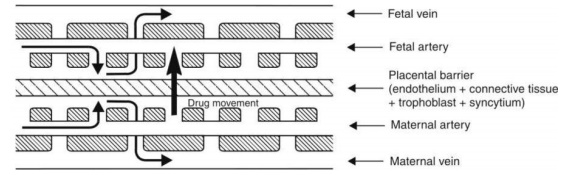
Fig. 3.7. Placental barrier and blood flow across it
The human placental barrier has a mean thickness of 25 microns in early pregnancy that reduces to 2 microns at full term which however does not reduce its effectiveness. Many drugs having molecular weight less than 1000 Daltons and moderate to high lipid solubility e.g. ethanol, sulphonamides, barbiturates, gaseous anaesthetics, steroids, narcotic analgesics, anticonvulsants and some antibiotics, cross the barrier by simple diffusion quite rapidly. This shows that the placental barrier is not as effective a barrier as BBB. Nutrients essential for the foetal growth are transported by carrier-mediated processes. Immunoglobulins are transported by endocytosis.
An agent that causes toxic effects on foetus is called as teratogen. Teratogenecity is defined as foetal abnormalities caused by administration of drugs during pregnancy. Drugs can affect the foetus at 3 stages as shown in table 3.2.
TABLE 3.2.
Stages during which teratogens show foetal abnormalities
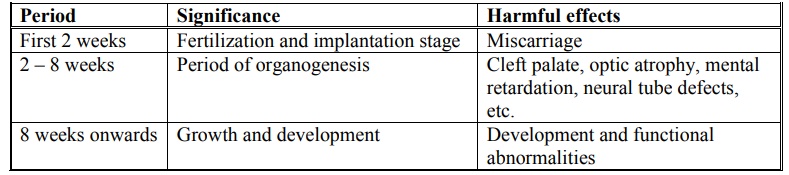
It is always better to restrict all drugs during pregnancy because of the uncertainty of their hazardous effects.
Blood-Testis Barrier: This barrier is located not at the capillary endothelium level but at sertoli-sertoli cell junction. It is the tight junctions between the neighbouring sertoli cells that act as the blood-testis barrier. This barrier restricts the passage of drugs to spermatocytes and spermatids.
Related Topics
