Tissue Binding of Drugs (Tissue Localization of Drugs)
| Home | | Biopharmaceutics and Pharmacokinetics |Chapter: Biopharmaceutics and Pharmacokinetics : Protein Binding of Drugs
The body tissues, apart from HSA, comprise 40% of the body weight which is 100 times that of HSA. Hence, tissue-drug binding is much more significant than thought to be.
TISSUE BINDING OF DRUGS (TISSUE LOCALIZATION OF DRUGS)
The body tissues, apart from HSA, comprise 40% of
the body weight which is 100 times that of HSA. Hence, tissue-drug binding is
much more significant than thought to be.
A drug can bind to one or more of the several
tissue components. Tissue-drug binding is important in distribution from two
viewpoints:
1. It increases the apparent
volume of distribution of drugs in contrast to plasma protein binding which
decreases it. This is because the parameter is related to the ratio of amount
of drug in the body to the plasma concentration of free drug and the latter is
decreased under conditions of extensive tissue binding of drugs.
2. Tissue-drug binding results in
localization of a drug at a specific site in the body (with a subsequent
increase in biological half-life). This is more so because a number of drugs
bind irreversibly with the tissues
(contrast to plasma protein-drug binding); for example, oxidation products of
paracetamol, phenacetin, chloroform, carbon tetrachloride and bromobenzene bind
covalently to hepatic tissues.
Factors influencing localization of drugs in
tissues include lipophilicity and structural features of the drug, perfusion
rate, pH differences, etc. Extensive tissue-drug binding suggests that a tissue
can act as the storage site for
drugs. Drugs that bind to both tissue and plasma components result in
competition between drug binding sites.
For majority of drugs that bind to extravascular
tissues, the order of binding is:
Liver > Kidney > Lung >
Muscles
Several examples of extravascular tissue-drug
binding are:
1. Liver: As stated earlier, epoxides of a
number of halogenated hydrocarbons and paracetamol
bind irreversibly to liver tissues resulting in hepatotoxicity.
2. Lungs: Basic drugs like imipramine,
chlorpromazine and antihistamines accumulate in lungs.
3. Kidneys: Metallothionin, a protein present
in kidneys, binds to heavy metals such as
lead, mercury, and cadmium and results in their renal accumulation and
toxicity.
4. Skin: Chloroquine and phenothiazines
accumulate in skin by interacting with melanin.
5. Eyes: The retinal pigments of the eye
also contain melanin. Binding of chloroquine and phenothiazines to it is responsible for retinopathy.
6. Hairs: Arsenicals, chloroquine and
phenothiazines are reported to deposit in hair shafts.
7. Bones: Tetracycline is a well-known
example of a drug that binds to bones and teeth. Administration of this antibiotic to infants or children during
odontogenesis results in permanent brown-yellow discoloration of teeth. Lead is
known to replace calcium from bones and cause their brittleness.
8. Fats: Lipophilic drugs such as
thiopental and the pesticide DDT accumulate in adipose tissues by partitioning into it. However, high o/w
partition coefficient is not the only criteria for adipose distribution of
drugs since several highly lipophilic (more than thiopental) basic drugs like
imipramine and chlorpromazine are not localized in fats. The poor perfusion of
adipose could be the reason for such an ambiguity. Reports have stated that
adipose localization of drugs is a result of binding competition between
adipose and non-adipose tissues (lean tissues like muscles, skin and viscera)
and not partitioning.
9. Nucleic Acids: Molecular components of cells
such as DNA interact strongly with drugs
like chloroquine and quinacrine resulting in distortion of its double helical
structure.
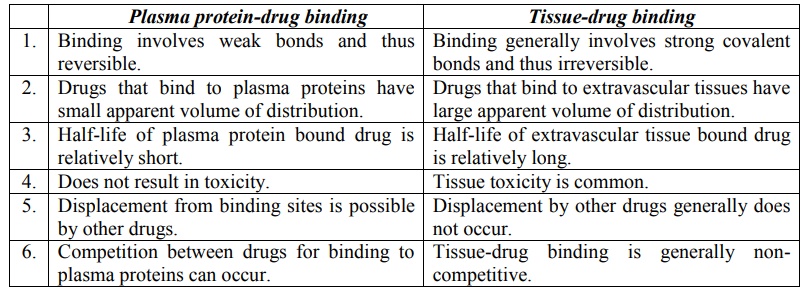
Table 4.2 compares plasma protein-drug binding and
tissue-drug binding.
TABLE 4.2
Comparison Between Plasma Protein-Drug Binding and Tissue-Drug Binding
Related Topics
