Alkenes
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Functional Group Synthesis
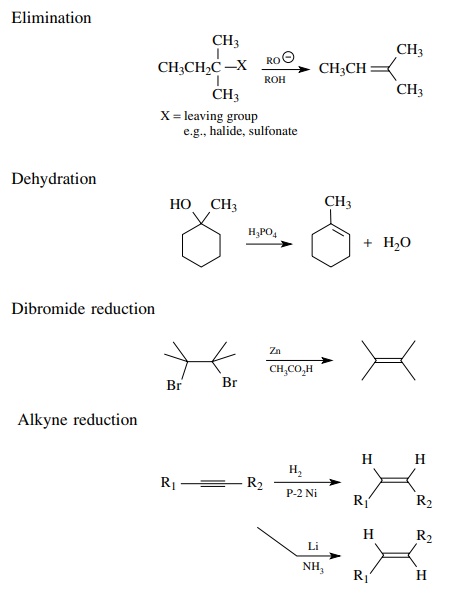
Alkenes are relatively low oxidation level hydrocarbons. The most common way to prepare alkenes is to carry out the elimination of a small molecule from between vicinal carbon atoms.
ALKENES
Traditional
preparations of alkenes include the following:
Alkenes
are relatively low oxidation level hydrocarbons. The most common way to prepare
alkenes is to carry out the elimination of a small molecule from between
vicinal carbon atoms. However, this is only a viable strategy if the
regiochemistry of elimination can be controlled. That is, traditional
dehydrohalo-genations or dehydrations often are regioselective but not
regiospecific, so that mixtures of structurally isomeric olefins are formed.
For example,
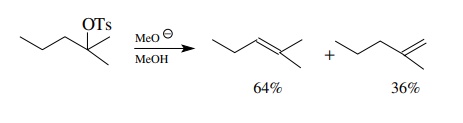
The
formation of regioisomers is due to the presence of several sets of
nonequiva-lent vicinal hydrogens of similar but not identical reactivity. The
resulting mixture of similar products must be separated if only one of the
regioisomers is desired. Since the alkene isomers are very similar in physical
properties, such separations can be very difficult and certainly are not
practical.
Several
strategies to control the elimination regiochemistry have been devel-oped.
These include placement of the leaving group, steric bulk of the base, and/or
establishment of thermodynamic control. By placing the leaving group at the end
of a chain, only terminal olefins can be produced by elimination because there
is only one set of vicinal hydrogens that can be removed by the base.
Diaz-abicycloundecane (DBU) is the base used in the example below. It is very
useful for promoting olefin-forming eliminations since it is a strong nitrogen
base which is also relatively nonnucleophilic.
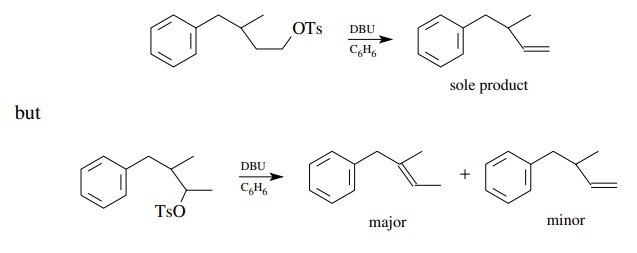
By
using very bulky alkoxide bases (t
-butoxide or amyloxide), attack of the base occurs at the least hindered
position — usually at the end of chains if possible. In this way the
regioselectivity of elimination is controlled by steric factors so that one
isomer is produced nearly exclusively.
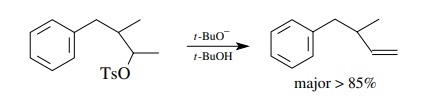
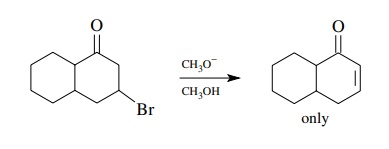
Finally,
when eliminations which give conjugated systems are possible, they are favored
significantly by the greater stability of the conjugated π system.
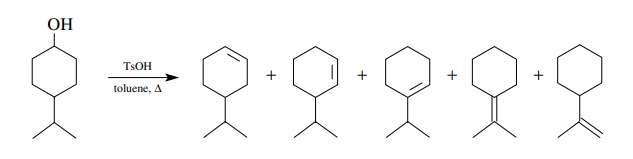
Dehydrations
produce olefins from alcohols by the acid-catalyzed elimination of a water
molecule from between two carbons. Acid-catalyzed dehydrations often give
mixtures of products because the intermediate carbocation is prone to cationic
rearrangements to more stable carbocations prior to formation of the olefin
product. Moreover, even when the intermediate carbocation is not subject to
skeletal rear-rangement, as in the case of tertiary alcohols, mixtures of
regioisomers are often produced during the loss of a proton from the
carbocation. As a consequence, the acid-catalyzed dehydration of alcohols is
generally not a viable synthetic method.
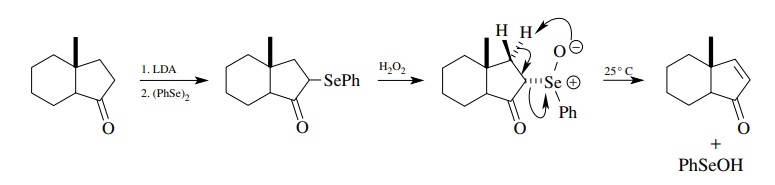
There
are many other methods for carrying out 1,2 eliminations to give olefins.
Several are particularly useful and widely used. Selenoxide eliminations are
fre-quently used to install the double bond of α, β -unsaturated
carbonyl compounds. They occur by concerted, cyclic, syn processes
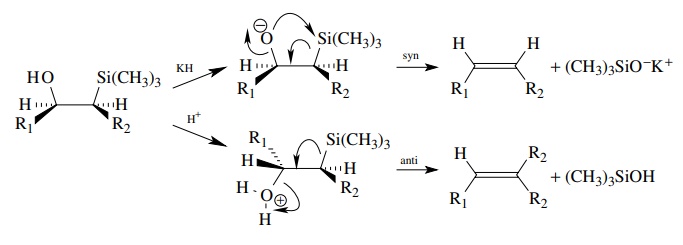
Silyloxide
eliminations (Petersen olefination) also proceed readily and regiospecifically
to give olefins. When base is used to produce the oxyanion, the elimination
occurs with syn stereochemistry. If an acid is used to promote the elimination,
it occurs in an anti fashion, leading to the opposite olefin stereochemistry.
This is a very useful way to generate either a Z or E olefin from the same
starting material.
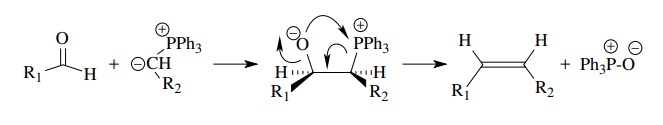
Similarly
phosphine oxide eliminations (Wittig reaction) occur very readily to give
olefins.
Both
of the latter two methods of elimination are part of a longer sequence of
reactions that produce olefins. Initial formation of a single bond to a
carbonyl carbon is followed by elimination to an alkene. Thus the alkene is a
condensation product of two smaller units. Schematically,
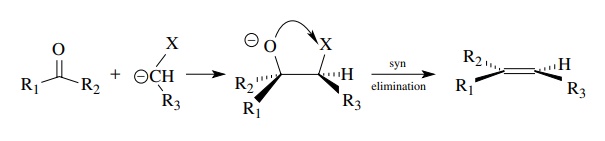
where
X is an element (Si or P) which can remove oxygen to the alkene. It should also
be noted that both anionic versions of these eliminations proceed with syn stereochemistry
between the oxyanion and the heteroatom; thus the stereochemistry of the
intermediate dictates the geometry of the olefin product.
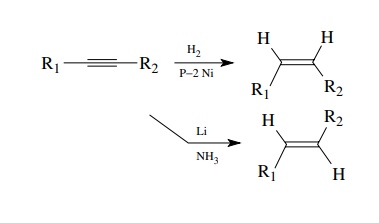
Alkenes
can also be produced effectively by the reduction of alkynes. The reduction can
be carried out stereospecifically to give either cis or trans olefins as
desired. This is a very useful method because of the stereocontrol. The P-2
nickel catalyst for the cis hydrogenation is produced in situ by the reduction
of Ni[II] acetate with sodium borohydride and the reaction is carried out at
atmospheric pressure making this a very simple method for the preparation of
cis olefins. The lithium in liquid ammonia reduction of alkynes to the trans
olefin is also very straightforward experimentally.