Imines and Imine Derivatives
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Functional Group Synthesis
Nitrogen analogs of both aldehydes and ketones which have the same oxidation level are imines and imine derivatives.
IMINES AND IMINE DERIVATIVES
Nitrogen
analogs of both aldehydes and ketones which have the same oxidation level are
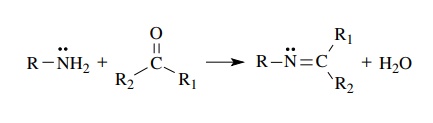
imines and imine derivatives. In almost every instance these compounds are
prepared by an exchange reaction between an amine derivative and a car-bonyl
compound. For simple imines (R =
alkyl, aryl) water removal by either a dehydrating agent (KOH or molecular
sieves) or azeotropic distillation is often employed to drive the reaction to
completion.
A
wide variety of other R groups attached to nitrogen can be used and all give
the C=N bond formation
with loss of water. However, most of these substitutions on the nitrogen atom
reduce the nucleophilicity of the amino group so acid catalysis is used to
facilitate the reaction. Many of these nitrogen derivatives of aldehydes and
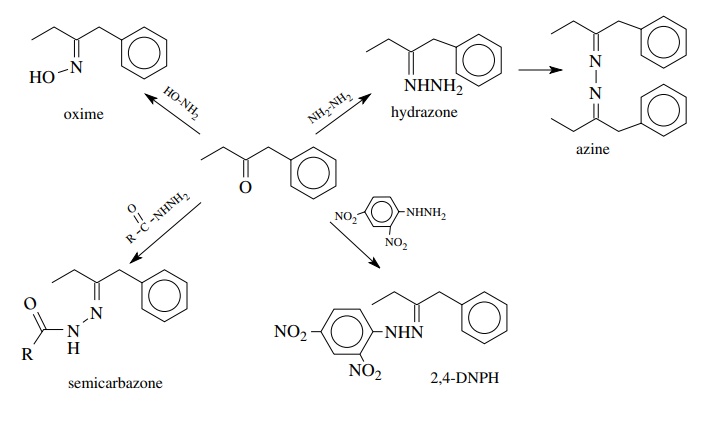
ketones have very interesting chemistry in their own right. These derivatives
also serve as a classic way to identify aldehydes and ketones. Many of these
derivatives tend to be solids with characteristic melting points. Thus
conversion of an aldehyde or ketone to a derivative, such as oxime or a
2,4-dinitrophenylhydrazone (2,4-DNPH) derivative, produces a solid with a
distinct melting point. Comparison of the melting point with the known value
can be used to confirm the structure of the aldehyde or ketone.