Anaphylactic Reactions
| Home | | Pharmacovigilance |Chapter: Pharmacovigilance: Anaesthetic Adverse Drug Reactions
In Australia, the reported incidence of anaphylac-tic reactions was between 1 in 10 000 and 1 in 20 000 anaesthetics (Fisher and Baldo, 1994).
ANAPHYLACTIC REACTIONS
In
Australia, the reported incidence of anaphylac-tic reactions was between 1 in
10 000 and 1 in 20 000 anaesthetics (Fisher and Baldo, 1994). Since 1984 in
France, there has been an epidemiological study of suspected anaphylactic
reactions occurring during anaesthesia. Initially, routine allergic
assess-ments focused on IgE-dependent immune mecha-nisms. These investigations
were skin tests combined with the identification of specific antibodies in the
serum. A spectrum of tests has now been described starting at the time of the
event with estimation of plasma histamines (these are of low specificity),
tryptase (with a half life of 2 hours and occasional false negative tests) and
specific IgE and skin tests 6 weeks later (AAGBI, 2003; Mertes and Laxenaire,
2002).
The
most common drugs implicated in these type of reactions were the neuromuscular
blocking agents with an incidence of 1 in 6500 anaesthetics compared with an
overall incidence of 1 in 13 000 (Laxenaire, 1999). The specific substances
identified in this multi-centre outpatient study as possible causes of allergic
phenomenon and that were associated with posi-tive allergy tests were
neuromuscular blocking drugs (62%), latex (17%), antibiotics (8%), hypnotics
(5%), colloid solutions (3%) and opioids (3%). Anaphylac-tic reactions to local
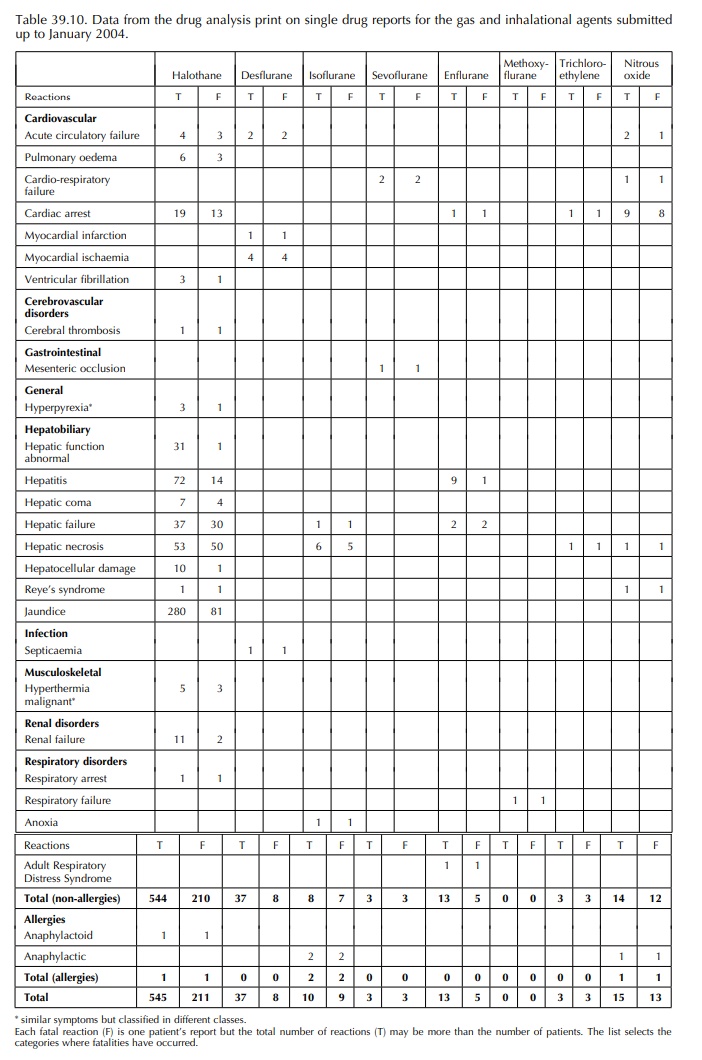
anaesthetic drugs are considered to be rare. The AAGBI report (2003) was unable
to identify allergic reactions to inhalational agents but a few have been
reported in the United Kingdom (Table 39.10) through yellow forms onto the
database of the MHRA.
Fisher
and colleagues have identified the presenting clinical features of anaphylaxis
during anaesthesia in 555 patients (Whittington and Fisher, 1998). In order of
frequency they are; no pulse, difficulty inflating the lungs, flushing, oxygen
desaturation, cough, rash, dysrrhythmias, urticaria and oedema. The
cardiovas-cular system is most often destabilised, and cardio-vascular collapse
may be the only feature, leading to misdiagnosis. Many factors can influence
sever-ity of the reaction. These are asthma, beta-adrenergic blockade and
neuraxial anaesthesia where there may be compromise of the sympathetic nervous
system. During the reaction, there was cardiovascular collapse (88%),
bronchospasm (37%), cutaneous signs such as erythema, urticaria, rash (in over
70%), oedema (33% including generalised and pulmonary) and gastroin-testinal
effects (7%). The wide range of clinical symptoms and signs may generate
diagnostic diffi-culties given the timing of the event and the range of drugs
used. The recommendations of the AAGBI (2003) include immediate management
(depending on the severity), immediate and late investigations and centralised
reporting.
A
diagnosis of an anaphylactoid reaction to anaes-thetic drugs may be difficult
to establish. First, many drugs are often delivered simultaneously; second,
skin testing may not be sensitive and third, the hetero-geneous nature of the
signs may delay or obscure the diagnosis. An important observation has been
that the severity of the reaction does not estab-lish the diagnosis. Although
most anaphylactic reac-tions were severe (88%) and often life-threatening (65%)
some cases were only mild (Mertes, Laxenaire and Alla, 2003) and may be
indistinguishable from anaphylactoid reactions without adequate diagnostic
investigation.
The
allergic reaction can be activated by the binding of antigens to the drugs. For
neuromuscular blocking drugs, the main antigenic determinants are substituted
ammonium ions. Most neuromuscular blocking drugs contain two similar quaternary
ammonium ions, and the distance between them is relevant to the chemical
structure of the antibodies. Flexibility in the molecule also confers
sensitivity to these effects as demon-strated by suxamethonium compared with
pancuro-nium. For thiopentone, two antigenic determinants have been identified,
one on position 5 of the pyrim-idine ring nucleus and the other in the thiol
region (Baldo, Fisher and Harle, 1991). It should be recog-nised that
antibodies to neuromuscular blocking drugs can persist for a long time.
The
risk factors for allergic reactions have been listed as gender, age, atopy and
allergy history (Mertes and Laxenaire, 2002). Reactions to anaesthetic drugs
are more common in females than males even when the gender ratio of
anaesthetised patients is taken into account. Age was only identified as a
factor for latex allergies, but allergies to anaesthetic drugs overall are
reported at all ages from neonates to the very elderly. Atopy has long been
considered a risk especially where there is a risk for histamine release, for
example neuromuscular blocking drugs (such as atracurium and mivacurium) or
where drugs have a food component. For example, the propofol formula-tion
contains egg lecithin and soybean oil, so its use is contraindicated in
patients with hypersensitivities to these components (Hofer et al., 2003). Interestingly, Mertes and
Laxenaire (2002) consider that previous drug exposure does not appear to be a
risk but a docu-mented reaction to a specific anaesthetic drug partic-ularly
the muscle relaxants is a positive risk factor. In addition, the high incidence
of cross-reactions leads to a recommendation of caution between muscle
relax-ants (Matthey et al., 2000).
Their advice in the context of a previous allergy to a neuromuscular blocking
drug is to check for cross-reactivity before anaesthetic administration. There
is no evidence for generalised screening before surgery but, given the
importance of a positive history of adverse drug reaction, primary prevention
and accurate documentation is essential.
Although the majority of adverse drug reactions to anaesthetic drugs occur at the time of anaesth-esia, there are many reported delayed reactions after general anaesthesia. These include exfoliative derma-titis, Stevens–Johnson syndrome and other events (Fisher and Baldeo, 1993).
Related Topics
