Homolytic Bond Making and Bond Breaking
| Home | | Organic Chemistry |Chapter: Organic Chemistry : Curved-Arrow Notation
Heterolytic processes make up a large proportion of organic transformations because most bonds are somewhat polarized.
HOMOLYTIC BOND MAKING AND BOND
BREAKING
Heterolytic
processes make up a large proportion of organic transformations because most
bonds are somewhat polarized. Heterolytic cleavage is merely an increase of
this polarity to the limit at which there is no bond remaining; that is,
electron movement follows in the direction established by the bond polarity to
give a cation – anion pair.
If
a bond is particularly weak and/or nonpolar, bond cleavage can occur by a
nonpolar or homolytic process. One electron of the shared pair goes with each
of the two bonded atoms. Bond breaking then is the movement of single electrons
rather than electron pairs and is indicated in curved-arrow notation as
“half-headed” arrows. Homolytic cleavage of a bond does not result in the
formation of charge but does result in the formation of unpaired electron
intermediates called free radicals. Free radicals normally have seven electrons
in the valence shell and as a consequence are very reactive intermediates.
Common examples of compounds which undergo homolytic bond cleavages include
halogens (Br2, Cl2, F2), peroxides (R–O–O–R),
and azocompounds (R–N=N–R).
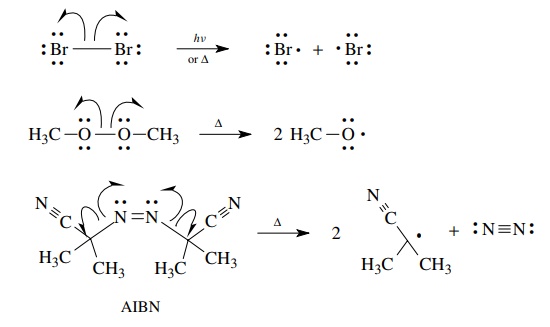
All
of these free-radical precursors are characterized by relatively weak, nonpolar
bonds which, upon heating, break to give free-radical intermediates. Free
radicals are very reactive and proceed to products by a variety of
one-electron, or homolytic, reactions.
Homolytic
bond formation can occur when two free-radical species contact each other. Each
has an available unpaired electron, and if these two electrons are shared, a
new bond will result.
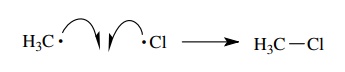
This
is simply the reverse of the homolytic cleavage. It is a very exothermic
process (by the amount equal to the energy of the bond being formed), and it
occurs at a very fast rate.
Homolytic
bond formation can also occur by the reaction of a free radical with a bonded
pair of electrons. Two common examples of this behavior are hydrogen (or other
atom) abstraction reactions and free-radical addition to double bonds. Atom
abstraction reactions take place by the interaction of a free radical with a σ-bonded
atom. One electron of the σ bond
pairs with the unpaired electron of the free radical to produce a new bond. The
remaining electron of the σ bond
remains on the fragment from which the atom has been abstracted and produces a
new free-radical species. This process is energetically driven by bond
strengths; that is, atom abstraction only occurs if the bond that is formed is
stronger than the one that is broken. In the example of hydrogen abstraction
shown below, a phenyl radical readily abstracts a benzylic hydrogen from
toluene to give benzene plus the benzyl free radical because the aromatic C–H
bond (103 kcal/mol) that is formed is appreciably stronger than the benzylic
C–H bond (85 kcal/mol) that is broken.
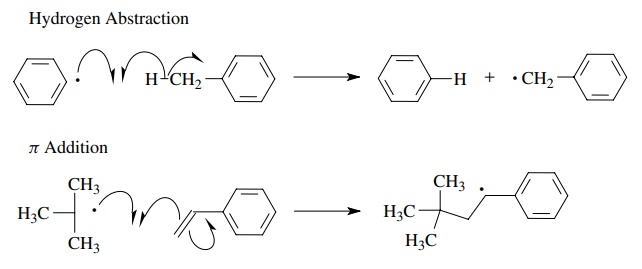
Addition
to π bonds is a second very common
reaction of free radicals. Inter-action of the free radical with the π-electron pair causes one of the π electrons to pair up with the unpaired
electron of the free radical to produce a new bond to one of the π-bonded atoms. The remaining π electron is now unpaired and thus
forms a new free-radical species. The process is often very favorable since the
new σ bond (70 – 90 kcal/mol) formed
in the addition process is normally much stronger than the π bond (60 kcal/mol) which is broken in the reaction. In the above
example a new carbon – carbon σ bond
is formed by free-radical addition to produce a new carbon-centered free
radical; however, a wide variety of other free-radical species add readily to
olefins.
Related Topics
