Introduction
| Home | | Inorganic Pharmaceutical Chemistry |Chapter: Essentials of Inorganic Chemistry : Introduction
Many metal ions play a vital role in living organisms. Metal ions are also involved in a variety of processes within the human body, such as the oxygen transport or the formation of the framework for our bones.
Introduction
Many metal ions play a vital role in living organisms. Metal
ions are also involved in a variety of processes within the human body, such as
the oxygen transport or the formation of the framework for our bones.
Haemoglobin is an iron-containing metalloprotein which carries oxygen from the
lungs to the various tis-sues around the human body. Calcium (Ca) ions are a
vital component of our bones. Elements such as copper (Cu), zinc (Zn) and
manganese (Mn) are essential for a variety of catalytic processes (Figure 1.1).
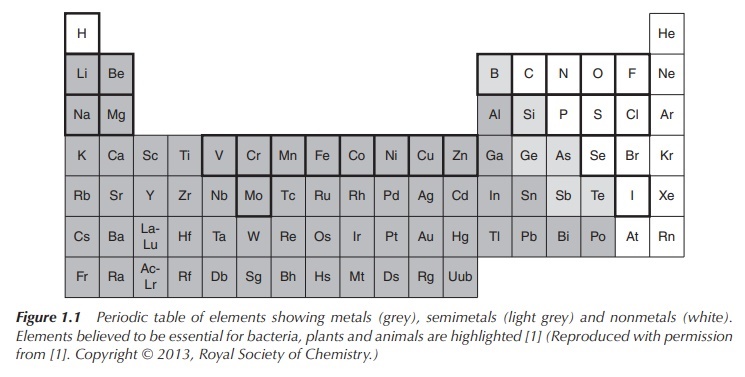
Nevertheless, metals are very often perceived as toxic elements.
Very often, the toxicity of a metal in a biological environment depends on the
concentration present in the living organism. Some metal ions are essential for
life, but concentrations too high can be highly toxic whilst too low
concentrations can lead to deficiency resulting in disturbed biological
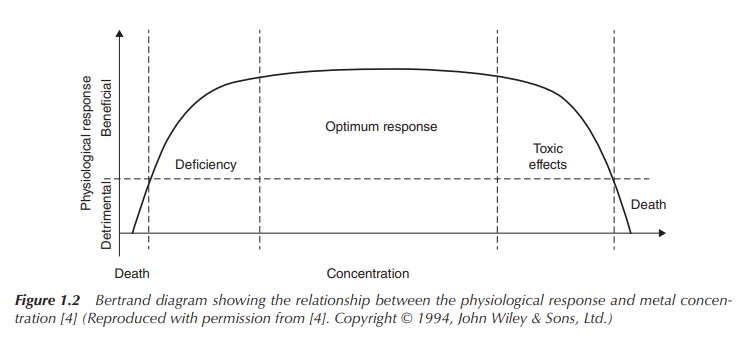
processes . The so-called Bertrand diagram visualises the rela-tionship between
the physiological response and the metal concentration. There are concentration
ranges that allow the optimum physiological response, whilst concentrations
above and below this range are detrimen-tal to life. The form of this diagram
can vary widely depending on the metal, and there are metals with no optimum
concentration range . Nevertheless, living organisms, including the human body,
have also found very sophisticated solutions to mask the toxicity of those
metals (Figure 1.2).
Researchers have questioned whether metal ions can and should be introduced into the human body arti-ficially and, if so, what the consequences are. Indeed, the use of metals and metal complexes for clinical applications gives access to a wide range of new treatment options.
Related Topics
