Liquid–liquid and liquid-gas interfaces
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Interfacial phenomena
A liquid or a solid phase can be defined as a conglomeration of like mol-ecules, held together by intermolecular bonds that hold the molecules in association and proximity with each other.
Liquid–liquid and
liquid–gas interfaces
A
liquid or a solid phase can be defined as a conglomeration of like mol-ecules,
held together by intermolecular bonds that hold the molecules in association
and proximity with each other. The two phases—liquid and solid—differ in the
degree of order in the association of the molecules, with the solid phase being
more ordered than the liquid phase. Within the solid phase, the crystalline
phases are more ordered than the amorphous phases. The gas phase, on the other
hand, is the least ordered, with the molecules undergoing random Brownian
motion, independent of other molecules.
The
bonds that hold a phase together are van der Waals force, ionic, dipole, and
hydrogen bonds—depending on the atomic structure of the molecules of a phase.
For example, water molecules are held together pre-dominantly by hydrogen bond
and dipole forces, whereas octane molecules are held together by weak van der
Waals forces. The strength of inter-molecular forces of attraction and the
proximity of the molecules follow the general trend: solids > liquids >
gases. In the bulk of a phase, a mol-ecule is surrounded by other molecules of
the same type and encounters similar forces in all directions, which tend to
neutralize each other. At the interface, a molecule encounters directionally
different forces (Figure 8.1). Forces of
attraction between the molecules of the same type within a phase can be termed cohesive forces, and the resulting
phenomenon is termed cohesion.
Similarly, forces between the molecules of different types at the interface can be termed adhesive forces, and the resulting
phenomenon is termed adhesion.
At
the liquid–gas interface, cohesive forces are generally greater than adhesive
forces, leading to an inward pull on the molecules toward the bulk. This force
pulls and keeps the molecules of the interface together and tends to contract
the surface, resulting in minimization of the exposed surface area. Thus, a
liquid droplet tends to be spherical, since this shape
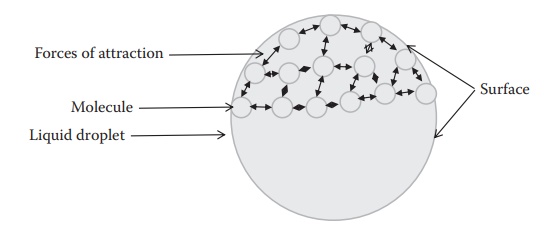
Figure 8.1 A liquid droplet depicted with some molecules (small spheres) with
mutual forces of attraction (depicted with arrows). The molecules at the
surface experience attractive forces from all directions, except at the
interface, leading to a pull toward the bulk of the liquid.
Expansion of surface
requires application of force. This force can be expressed in terms of surface
or interfacial tension.
Surface tension
Surface tension (γ) is the force per unit length that
must be applied in paral-lel to the
surface to expand the surface, counterbalancing the net inward pull. It has units of force per unit length, for example,
dyne/cm. Surface tension of a liquid film is commonly determined by creating a
film of the liquid in a horizontal bar apparatus (Figure
8.2) and pulling the film using standard weights until the film breaks.
Surface tension of the solution form-ing the film is a function of the force
that must be applied to break the film over the length of a movable bar in
contact with the film. Since the film has two liquid–gas interfaces (one above
and one below the plane of the bar), the total length of the contact is equal
to twice the length of the bar.
Thus,

where:
fb is the force required for breaking the film
L is the length of the
film or the movable bar
Surface
tension of a liquid is constant. Thus, this equation indicates that the amount
of force required to break the film is directly proportional to the
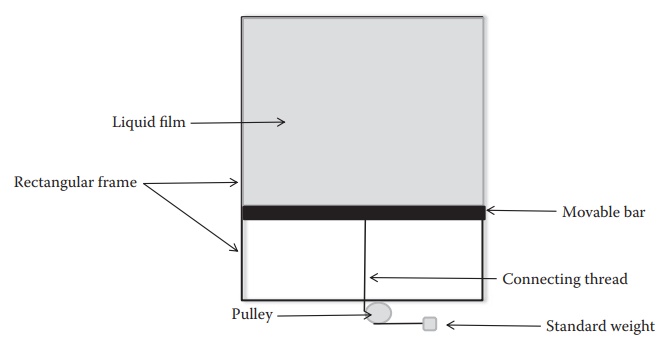
Figure 8.2 A simplistic representation of a rectangular block apparatus for
determining the surface tension of a liquid.
Interfacial tension
Interfacial tension is the force per
unit length that must be applied in
parallel to the interface to expand the interface, counterbalancing the net inward pull of the two phases. While
the term surface tension is reserved for liquid–gas and solid–gas
interfaces, the term interfacial tension
is com-monly used for liquid–liquid interfaces. Interfacial tension has the
same symbol (γ) and units
(dyne/cm) as surface tension and is derived similarly from the amount of force
required to create new interface. Subscripts are commonly used to distinguish
between different interfacial tensions. For example, γ L/L is the interfacial
tension between two liquids (designated “L”), and γ L/V is the surface
tension between a liquid and its vapor (designated “V”) in the gas phase.
Usually,
the interfacial tension (liquid–liquid)
of a hydrophilic liquid is less than its surface
tension (liquid–vapor). This is because the adhesive forces between two
liquid phases forming an interface are generally higher than those between a
liquid and a gas phase. For example, at ~20°C, the interfacial tension between
water and carbon tetrachloride is 45 mN/m, while the surface tension of water
is 72.8 mN/m.
Factors affecting surface tension
Surface
tension is measured with devices known as tensiometers.
These devices measure the force by which a surface is held together while the
force is applied on the surface to expand it. The methods for surface ten-sion
measurement include the du Nouy method (maximum pull on a rod or plate immersed
in a liquid), du Nouy ring method (maximum down-ward force on a ring pulled
through the liquid–air interface), Wilhelmy plate method (downward force on a
plate lowered to the surface of the liquid), and pendant drop method (shape of
the drop at the tip of needle by optical imaging). All of these methods measure
the inherent force within a liquid that resists the growth or expansion of its
surface. Factors affecting this force, or the surface tension, of a liquid
include the following:
·
Nature of the liquid: Greater the
cohesive forces between the molecules of
a liquid, higher its surface tension. Thus, the surface tension of water (72.8
mN/m at 20°C) is higher than that of methanol (22.7 mN/m). Mixing of the two
miscible solvents leads to an intermediate surface tension. For example, a 7.5%
solution of methanol in water has a surface tension of 60.9 mN/m.
·
Temperature: Surface tension of
most liquids decreases linearly with an
increase in temperature. This is because of greater Brownian motion of
individual molecules that leads to reduction in the inter-molecular attractive
forces and, thus, the reduced inward pull
of the molecules on the surface.
Surface free energy
Surface
free energy of a liquid is defined as the work required for increasing the
surface area. Surface free energy (W)
and surface tension (γ)
are related by:
W=γΔA (8.2)
Where,
W is the work done, or the surface
free energy (ergs) input, required to increase the surface by an area ∆ A (cm 2) for a liquid that has the
surface tension γ (dynes/cm).
Surface
free energy represents the amount of energy put into the system per unit
increase in surface area. Thermodynamically, surface free energy represents the
Gibbs free energy at constant temperature and pressure.
W= ∆G= γ∆A
(8.3)
Thus,
surface tension (γ) can be represented
as the increment in Gibbs free energy per unit area.
γ =
∂G/∂A (8.4)
Example 1: If the
length of the bar (Figure 8.2) is 5 cm and the
mass required to break a liquid film is 0.5 g, what is the surface tension of the
soap solution? What is the work required to pull the wire down 1 cm?
Since
γ = fb/2L
∴ γ = (0.50 g × 981 cm/s2)/10 cm = 49
dyn/cm In addition,
W = γΔA
∴ W = 49 dyn/cm ×
10 cm2 = 490 ergs.
Related Topics
