Solid-gas interface: Adsorption
| Home | | Pharmaceutical Drugs and Dosage | | Pharmaceutical Industrial Management |Chapter: Pharmaceutical Drugs and Dosage: Interfacial phenomena
If a solid comes in contact with a gas or a liquid, there is an accumula-tion of gas or liquid molecules at the interface. This phenomenon is known as adsorption.
Solid–gas interface
Adsorption
If
a solid comes in contact with a gas or a liquid, there is an accumula-tion of
gas or liquid molecules at the interface. This phenomenon is known as adsorption. Adsorption refers to the
surface binding of a liquid or gas molecule (adsorbate) onto a solid surface (adsorbent). Examples of adsor-bents are highly porous solids, such
as charcoal and silica gel, and finely divided powders, such as talc. Adsorbate
could be any molecule, such as a drug compound.
Removal
of the adsorbate from the adsorbent is known as desorption. A physically adsorbed gas may be desorbed from a solid
by increasing the temperature and reducing the pressure. Adsorption is a surface phenom-enon, distinct from absorption, which implies the
penetration through the solid surface into the core of the solid.
Factors affecting adsorption
The
degree of adsorption depends on the following:
·
The chemical nature of the adsorbent and the adsorbate.
Since adsorp-tion is a result of an adhesive process, whereby two types of
molecules interact with one another, the nature of the two types of molecules
will determine their attractive interactions.
·
Surface area of the adsorbent. Greater the surface area of
the adsor-bent, more the absolute amount of adsorbate that can be adsorbed. In
modeling the adsorption phenomenon, the amount of adsorbate per unit adsorbent
is usually calculated. In this scenario, the specific surface area (surface
area per unit mass) of the adsorbent plays a role in deter-mining the amount of
adsorbate per unit mass of the adsorbent. This
·
phenomenon indicates that a finely divided solid (of the
same mass as a coarse particulate solid) would adsorb greater amount of
adsorbate.
·
Temperature. Temperature increases molecular motion, and its
effect on adsorption depends on the relative change in the intermolecular
forces of attraction between the molecules of the two phases. Generally, an
increase in Brownian motion with increasing temperature reduces adsorption.
·
Partial pressure (gas) or concentration (liquid) of the
adsorbate. Generally, greater the solute (adsorbate) partial pressure or
concen-tration, greater the rate of adsorption.
Types of adsorption
Adsorption
can be physical or chemical in nature. Table 8.1
compares the characteristics of physical and chemical adsorption.
1. Physical adsorption
Physical
adsorption is rapid, nonspecific, and relatively weak. It is typically mediated
by weak noncovalent forces of attraction, such as van der Waals
Table 8.1 Characteristics of
physical and chemical adsorption
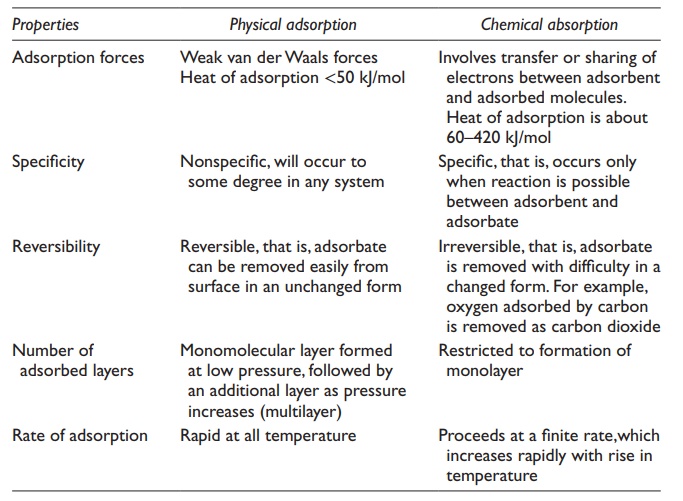
Physical adsorption is an exothermic process, since heat is
released with the formation of attractive interactions between mol-ecules of
the two phases. Physical adsorption may be associated with three phenomena:
·
Monolayer formation: Adsorption of a
solute on a solid surface leads to a
monolayer formation, as the solute occupies the available surface in a single
layer.
·
Multilayer formation: Surface adsorption
may continue into multilayer formation
if the adsorption is facilitated by the interactions of solute molecules with
other solute molecules (that are already adsorbed on the solid surface). Once
the monolayer formation is complete and the conditions (such as solute
concentration in the liquid or partial pressure of the gas) are supportive,
multimolecular adsorption may take place.
·
Condensation: The adsorbate may
condense in the pores or capillar-ies of the adsorbent, leading to changes in
the kinetics of the rate and the extent of adsorption.
2. Chemical adsorption (chemisorption)
Chemical
adsorption or chemisorption is an
irreversible process in which the adsorbent gets covalently linked to the
adsorbate by chemical bonds.
Chemisorption
is specific and may require activation energy. Therefore, this process is slow,
and only a monolayer may be formed.
Adsorption isotherms
An
adsorption isotherm is a graph that shows the amount of solute/adsorbate
adsorbed per unit mass of a solid/adsorbent as a function of the equilibrium
partial pressure (P) of the gaseous solute or the concentration (c) of the
sol-ute in the liquid at a constant temperature (thus, the term isotherm).
1. Type of isotherms
The
isotherms can generally be classified into five types [UK34](Figure 8.3):
·
Type I isotherms (e.g., ammonia on charcoal at 273 K) show a
fairly rapid rise in the amount of solute adsorbed with increasing pressure to
a limiting value. This phenomenon is due to the adsorption being restricted to
a monolayer.
·
Type II isotherms (e.g., nitrogen on silica gel at 77 K) are
frequently encountered and represent multilayer physical adsorption on
nonpo-rous solids. They are often referred to as sigmoid isotherms. These isotherms are characterized by rapid
solute adsorption to a limiting value, which sustains for certain increase in
the partial pressure of the solute. Thereafter, multilayer adsorption initiates
at an exponentially increasing rate.
·
Type III adsorption isotherm (e.g., bromide at 760°C or
iodine at 790°C on silica gel) shows large deviation from Langmuir model, no
flattish portion in the curve, and the formation of multilayer films.
·
Isotherm IV is typical of adsorption onto porous solids and
involves the formation of a monolayer, which is followed by multilayer
forma-tion. An asymptote toward a limiting value is observed after each
additional layer formation.
·
Type V isotherm is similar to a type III isotherm in terms
of the initial rate of solute adsorption increasing exponentially with solute
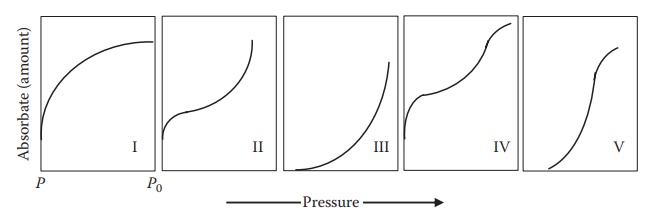
Figure 8.3 Types of adsorption isotherms.
concentration
or partial pressure. This behavior is seen in relatively few instances in which
the heat of adsorption of the solute in the first layer is less than the latent
heat of condensation of successive layers. This promotes more rapid deposition
of subsequent layers of adsorbed solute over the previous layer. Type III
isotherm does not involve an eventual asymptote toward a limiting value, while
type V isotherm does.
2. Modeling isothermal adsorption
Adsorption
of a solute on a solid substrate at constant temperature (i.e., isothermal conditions) is a kinetic and
a thermodynamic equilibrium phe-nomenon that can be described with the help of
empirical or semiempirical equations. Modeling adsorption helps us understand a
system and builds predictive ability to interpret the implications of changing
system variables on the amount of free versus adsorbed solute. For example, in
the case of drug adsorption on activated charcoal for preventing drug
absorption into the systemic circulation after an oral overdose, the modeling
of adsorp-tion isotherm enables simulation of absorption and pharmacokinetics
of the drug in the presence and absence of charcoal and the effect of different
quantities of drug and charcoal. This can help determine the required dose of
charcoal for a given drug overdose. In addition, modeling the adsorp-tion data
can be used to generate information about the system that would otherwise be
unavailable. For example, gas adsorption on a solid substrate is used to
quantify the specific surface area of a solid.
Isothermal
adsorption can be modeled by using Freundlich, Langmuir, or BET equations.
2.1 Freundlich adsorption isotherm
Some
cases of isothermal adsorption of a gas on a solid can be explained by the
empirical Freundlich equation (Figure 8.4a).
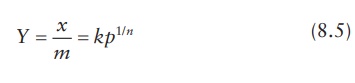
where:
Y is the mass ratio of
the adsorbent on the adsorbate, given by the ratio of the mass of gas (x)
adsorbed per unit mass (m) of
adsorbent at the partial pressure of gas (p).
The
k and n are constants for a particular system at a constant temperature.
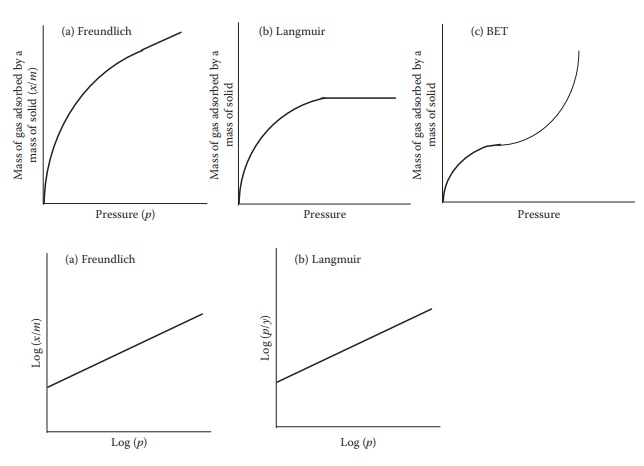
Figure 8.4 Plots showing (a) Freundlich, (b) Langmuir, and (c) BET
isotherms.
The
Freundlich isotherm, thus, states dependence of the mass of gas adsorbed on the
partial pressure of gas with nonlinear kinetics, which depends on the specific
combination of the adsorbent, the adsorbate, and the environment. Thus, the
constants k and n depend not only on the sub-strate (adsorbate) and the gas
(adsorbent) but also on the system (environ-ment, such as other constituents).
The
above equation can be written logarithmically as:
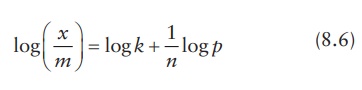
A
plot of log (x/m) against log p yields a straight line, with slope
1/n and intercept log k. This allows the experimental
determination of the constants for a given system.
Freundlich
isotherm models multilayer adsorption and mostly represents physical adsorption
that does not reach saturation.
2.2 Langmuir adsorption isotherm
Langmuir
developed an equation based on the theory that the molecules or atoms of gas
are adsorbed on active sites of the solid to form a layer one-molecule thick
(monolayer) (Figure 8.4b). Langmuir adsorption
isotherm predicts not only a dependence on the partial pressure of gas (p) but also saturable kinetics of the
overall rate of adsorption (K), which
is defined as the ratio of the forward (adsorption) reaction rate constant (ka) to the reverse
(desorption) reaction rate constant (kd).
Thus, for the adsorption reaction,
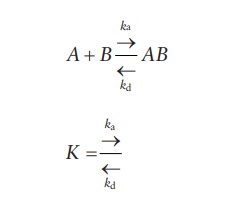
Langmuir
adsorption isotherm predicts that:
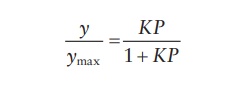
where,
y is the number of available surface
adsorption or binding sites occupied by the adsorbent, also expressed as the
mass of gas adsorbed per unit mass of adsorbent, and ymax is the total number of surface adsorp-tion or
binding sites on the adsorbate, also expressed as the maximum mass of gas that
a unit mass of adsorbent can absorb when monolayer is complete.
Therefore,
the Langmuir adsorption isotherm predicts that y never exceeds ymax,
even as the rates of forward, adsorption, reaction reach but never exceed 1.
The
simplified equation of Langmuir isotherm is:
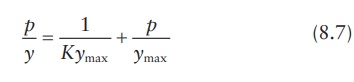
A
plot of p/y against p yields a straight line, with 1/ymax as the slope and 1/Kymax as the intercept. This allows the experimental estimation of the values of ymax and K.
Langmuir
adsorption isotherm is often indicative of chemisorption and has the following
characteristics:
·
Adsorption is localized to the active regions on the
surface, and only monolayer adsorption takes place.
·
Heat of adsorption is independent of surface coverage,
indicating that all molecules being adsorbed experience the same attractive
force, independent of the neighboring adsorbed molecules.
2.3 BET adsorption isotherm
The
BET adsorption isotherm models multilayer gas adsorption and assumes that the
forces involved in physical adsorption are the same as those responsible for
the condensation of the adsorbate.
The
BET equation relates the partial pressure of gas (P) with the relative proportion of the adsorbed molecules (Y/Ym) by the equation:
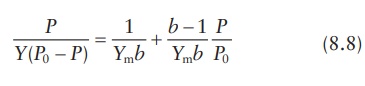
where:
p is the partial
pressure of adsorbate
y is the mass of
adsorbate per unit mass of adsorbent
P0 is the vapor pressure of adsorbate when the adsorbent is
saturated with adsorbate molecules
Ym is the maximum quantity of adsorbate adsorbed per unit mass
of the adsorbent
b is the constant
proportional to the difference between the heat of adsorption of the gas in the first layer and the latent heat of
con-densation in the successive layers
The
BET isotherms occur when gases undergo physical adsorption onto nonporous
solids to form a monolayer, followed multilayer formation. The BET isotherms
have a sigmoidal shape (Figure 8.4c) and
represent type II isotherms.
Related Topics
