Molecular Diffusion in Gases
| Home | | Pharmaceutical Technology |Chapter: Pharmaceutical Engineering: Mass Transfer
Transport of material in stagnant fluids or across the streamlines of a fluid in laminar flow occurs by molecular diffusion.
MOLECULAR DIFFUSION IN GASES
Transport of material in stagnant fluids or across the streamlines of a fluid in laminar flow occurs by molecular diffusion. In Figure 4.1, two adjacent compartments, separated by a partition, are drawn. Each compartment contains a pure gas, A or B. Random movement of all molecules occurs so that after a period of time molecules are found quite remote from their original positions. If the partition is removed, some molecules of A will move toward the region occupied by B, their number depending on the number of molecules at the point considered. Concurrently, molecules of B diffuse toward regions formerly occupied by pure A.
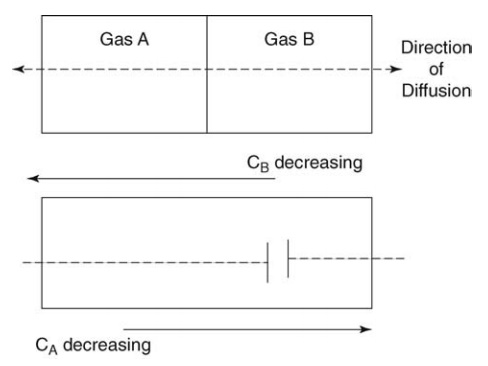
FIGURE 4.1 Molecular diffusion of gases A and B.
Ultimately,
complete mixing will occur. Before this point in time, a gradual variation in
the concentration of A will exist along an axis, designated x, which joins the original
compartments. This variation, expressed mathematically, is -dCA/dx, where CA is the concentration of A. The negative sign
arises because the concentration of A decreases as the distance x increases.
Similarly, the variation in the concentration of gas B is -dCB/dx. These
expressions, which describe the change in the number of molecules of A or B
over some small distance in the direction indicated, are concentration
gradients. The rate of diffusion of A, NA, depends on the
concentration gradient and on the average velocity with which the molecules of
A move in the x direction. Fick’s law
expresses this relationship.
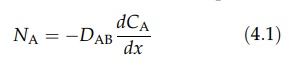
where
D is the diffusivity of A in B. It is a property proportional to the average
molecular velocity and is, therefore, dependent on the temperature and pressure
of the gases. The rate of diffusion, NA, is usually expressed as the
number of moles diffusing across unit area in unit time. In the SI system,
which is used frequently for mass transfer, NA would be expressed as
moles per square meter per second. The unit of diffusivity then becomes m2/sec.
As with the basic equations of heat transfer, equation (4.1) indicates that the
rate of a process is directly proportional to a driving force, which, in this
context, is a concentration gradient.
This
basic equation can be applied to a number of situations. Restricting discussion
exclusively to steady-state conditions, in which neither dCA/dx nor dCB/dx changes with time, equimolecular
counterdiffusion is considered first.
Related Topics
