Particle Interactions
| Home | | Pharmaceutical Technology |Chapter: Pharmaceutical Engineering: Powders
The attraction between particles or between particles and a containing boundary influences the flow and packing of powders.
PARTICLE INTERACTIONS
The
attraction between particles or between particles and a containing boundary
influences the flow and packing of powders. If two particles are placed
together, the cohesive bond is normally very much weaker than the mechanical
strength of the particles themselves. This may be due to the distortion of the
crystal lattice, which prevents the correct adlineation of the atoms or the
adsorption of surface films. These prevent contact of the surfaces and usually
but not always decrease cohesion. Low cohesion is also the result of small area
of contact between the surfaces. On a molecular scale, surfaces are very rough,
and the real area of contact will be very much smaller than the apparent area.
Finally, the structure of the surface may differ from the interior structure of
the particle. Nevertheless, the cohesion and adhesion that occur with all
particles are appreciable. It is normally ascribed to nonspecific Van der
Waal’s forces, although, in moist materials, a moisture layer can confer
cohesiveness by the action of surface tension at the points of contact. For
this reason, an increase in humidity can produce a sudden increase in
cohesiveness and the complete loss of mobility in a powder that ceases to flow
and pour. The acquisition of an electric charge by frictional movement between
particles is another mechanism by which particles cohere together or adhere to
containers.
These
effects depend on both the chemical and physical forms of the powder. They
normally oppose the gravitational and momentum forces acting on a particle
during flow and therefore become more effective as the weight or size of the
particle decreases. Cohesion and adhesion increase as the size decreases
because the number of points in contact in a given area of apparent contact
increases. The effects of cohesion will often predominate at sizes less than
100 μm and powders will
not pass through quite large orifices, and vertical walls of a limited height
appear in a free surface. The magnitude of cohesion also increases as the bulk
density of the powder increases.
Cohesion
also depends on the time for which contact is made. This is not fully
understood but may be due to the gradual squeezing of air and adsorbed gases
from between the approaching surfaces. The result, however, is that a system
that flows under certain conditions may cease to flow when these con-ditions
are restored after interruption. This is of great importance in the storage and
intermittent delivery of powders. Fluctuating humidity can also destroy flow
properties if a water-soluble component is present in the powder. The
alternating processes of dissolution and crystallization can produce very
strong bonds between particles, which cement the mass together.
Measurement of the Effects of
Cohesion and Adhesion
The
measurement of the cohesion between two particles or the adhesion of a particle
to a boundary is difficult, although several methods can be used. More
commonly, these effects are assessed by studying an assembly of particles in
the form of a bed or a heap. Flow and other properties of the powder are then
predicted from these studies (Crowder and Hickey, 2000).
The
most commonly observed and measured property of a heap is the maximum angle at
which a free powder surface can be inclined to the
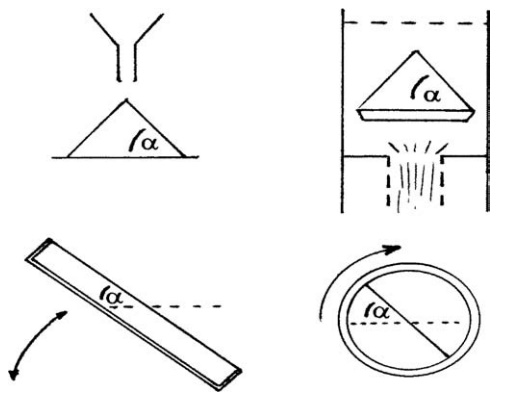
FIGURE 5.1 Measurement of the angle of repose, α.
horizontal.
This is the angle of repose, and it can be measured in a number of ways, four
of which are shown in Figure 5.1. The angle depends to some extent on the
method chosen and the size of the heap. Minimum angles are about 25○,
and powders with repose angles of less than 40○ flow well. If the
angle is over 50○, the powder flows with difficulty or does not flow
at all.
The
angle, which is related to the tensile strength of a powder bed, increases as
the particle shape departs from sphericity and as the bulk density increases.
Above 100 μm, it is independent
of particle size, but below this value, it increases sharply. The effect of
humidity on cohesion and flow is reflected in the repose angle. Moist powders
form an irregular heap with repose angles of up to 90○.
A
more fundamental measure is the tensile stress necessary to divide a powder
bed. The powder may be dredged on to a split plate or, in a more refined
apparatus, contained within a split cylinder and carefully consolidated. The
stress is found from the force required to break the bed and the area of the
divided surface. The principles of this method are shown in Figure 5.2A, and
stresses of up to 100 N/m2 are necessary to divide a bed of fine
powders. Values increase as the bulk density increases. Changes in cohesiveness
with time and the severe changes in the flow properties of some powders that
occur when the relative humidity exceeds 80% can be assessed with this
apparatus.
Apparatus
for shearing a bed of powder is shown diagrammatically in Figure 5.2B. The
shear stress at failure is measured while the bed is constrained under a normal
stress. The latter can be varied. The relation between these stresses, a
subject fully explored in the science of soil mechanics, is used in the design
of bins and hoppers for the storage and delivery of powders.
The
adhesion of particles to surfaces can be studied in a number of ways.
Measurement of the size of the particles retained on an upturned plate is a
useful qualitative test. A common method measures the angle of inclination at
which a powder bed slides on a surface, the bed itself remaining coherent.
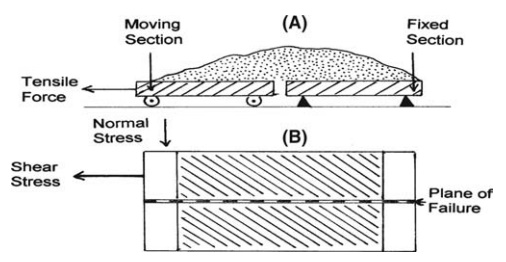
FIGURE 5.2 Measuring the (A)
tensile and (B) shear strength of a powder bed.
